Abstract
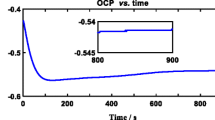
The dissolution behaviour of the anodized niobium electrode in NaOH solutions was investigated as a function of alkali concentration, formation voltage, formation current density and temperature using potential and impedance measurements. The rate of dissolution is dependent on the alkali concentration. In dilute NaOH solutions (1 N) the anodic oxide film formed in 0.5 M H2SO4 is reasonably stable. On the other hand, at higher concentrations of NaOH (≥2 M), the anodized electrode is subject to continuous dissolution depending on the alkali concentration. Also, the dissolution process is considerably affected by temperature; at temperatures greater than 320 K the oxide film is destroyed in less than 30 min. The results show that the current density used during the formation of the oxide film has no effect on its dissolution rate.
Similar content being viewed by others
References
O. De Nora,Chem. Ing. Tech. 42 (1970) 222.
S. Trasatti, ‘Electrodes of Conductive Metallic Oxides’, Part B, Elsevier, Amsterdam (1980) Chap. 11.
L. Young, ‘Anodic Oxide Films’, Academic Press, London (1961).
M. S. El-Basiouny and A. M. Bekheet,Br. Corros. J. 15 (1980) 89.
M. J. Joncich and L. S. Stewart,J. Electrochem. Soc. 112 (1965) 717.
M. Pourbaix, ‘Atlas of Electrochemical Equilibria in Aqueous Solutions’, Pergamon Press (1966).
D. Piron and Ken Nobe,Corros. NACE 25 (1969) 67.
K. E. Z. Heusler,Metalik 61 (1970) 828.
A. D. Davydov, V. D. Kashcheev and A. N. Kamkin,Elektrokhimiya 8 (1972) 282.
S. Iseki, K. Ohashi and S. Nagaura,Electrochim. Acta 17 (1972) 2239.
O. A. Omel'Chenko and Ya. S. Gorodetskii,Zasch. Met. 11 (1975) 466.
L. A. Kamel, N. Shawky and H. M. Sammour,Egypt Acad. Sci. 29 (1976) 1.
29 (1976) 7.
R. Badar, G. Bouyssoux and M. Romand,Mat. Res. Bull. 11 (1976) 525.
A. Ya. Shatalov, T. P. Bondareva and L. E. Tsygankous,J. Appl. Chem. USSR 36 (1963) 561.
M. S. El-Basiouny, A. M. Bekheet and A. G. Gad-Allah,Corros. NACE 40 (1984) 116.
J. J. Randall, W. J. Bernard and R. R. Wilkinson,Electrochim. Acta 10 (1965) 183.
D. M. Lakhiani and L. L. Shreir,Nature 188 (1960) 49.
D. Stützle and K. E. Heusler,Z. Phys. Chem. (Frankfurt am Main) 65 (1969) 201.
M. S. El-Basiouny, M. M. El-Kot and M. M. Hefny,Br. Corros. J. 14 (1979) 51.
M. S. El-Basiouny, S. A. Hassan and M. M. Hefny,Corros. Sci. 20 (1980) 909.
A. G. Gad-Allah, A. A. Mazhar and M. A. Ameer, 12th Egyptian Chemical Conference, Cairo, 7–10 April 1984, p. 140 (Abstract).
O. P. Agnihotri and B. K. Gupta, ‘Solar Selective Surfaces’, John Wiley and Sons, New York (1981).
L. Young, ‘Anodic Oxide Films’, Academic Press, London (1980) p. 49.
W. A. Badawy, M. S. El-Basiouny and M. M. Ibrahim,Indian J. Tech. 24 (1986) 1.
W. A. Badawy, M. M. Ibrahim, M. M. Abou-Romia and M. S. El-Basiouny,Corros. NACE 42 (1986) 324.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Badawy, W.A., Gad-Allah, A.G. & Rehan, H.H. Kinetics of the dissolution behaviour of anodic oxide films on niobium in NaOH solutions. J Appl Electrochem 17, 559–566 (1987). https://doi.org/10.1007/BF01084130
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01084130