Abstract
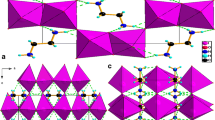
The crystal structure of the title compound has been determined from diffractometer data and refined with isotropic thermal parameters to finalR w factors ofR=0.058 andR w =0.056. It crystallizes in the orthorhombic space groupp212121 (No. 19), witha=9.706(7),b=18.826(6), andc=25.258(6) Å. The asymmetric unit contains four nitroprusside ions, eight potassium ions, and five water molecules, one of which shows disorder. The infrared spectra of the polycrystalline compound, both normal and with different degrees of deuteration at temperatures ranging from liquid nitrogen to room temperature, were obtained. The vibrational spectra present an unusual number of fundamental and first overtone NO stretching bands and a rich structure in the water bending region, in accordance with the existence of inequivalent nitroprusside ions and water molecules.
Similar content being viewed by others
References
Amalvy, J. I., and Aymonino, P. J. (1986), to be published.
Bates, J. B., and Khanna, R. K. (1970)Inorg. Chem. 9, 1376.
Berglund, B., Lindgren, J., and Tegenfeldt, J. (1978a)J. Mol. Struct. 43, 169.
Berglund, B., Lindgren, J., and Tegenfeldt, J. (1978b)J. Mol. Struct. 43, 179.
Brink, G., and Falk, M. (1970)Can. J. Chem. 48, 2096.
Chamberlain, N. M., and Green, A. F., Jr. (1963)J. Inorg. Nucl. Chem. 25, 1471.
Cromer, D. T., and Ibers, J. A. (1974).International Tables for X-Ray Crystallography, (Kynoch Press, Birmingham), Vol. IV, p. 149.
Cromer, D. T., and Waber, J. T. (1974).International Tables for X-Ray Crystallography (Kynoch Press, Birmingham), Vol. IV, pp. 71–98.
Delia Védova, C. O., Lesk, J. H., Varetti, E. L., Aymonino, P. J., Piro, O. E., Rivero, B. E., and Castellano, E. E. (1981)J. Mol. Struct. 70, 241.
Falk, M. (1984)Spectrochim Acta A 40, 43.
Friedman, H. L., and Lewis L. (1976)J. Solution Chem. 5, 445.
Gans, P., Sabatini, A., and Sacconi, L. (1966)Coord. Chem. Rev. 1, 187.
Garg, A. N., and Goel, P. S. (1971)Inorg. Chem. 10, 1345.
Gentil, L. A. (1973) Doctoral Thesis, Facultad de Ciencias Exactas, Universidad Nacional de La Plata, R. Argentina.
Gentil, L. A., Olabe, J. A., Baran, E. J., and Aymonino, P. J. (1975)Therm. Anal. 7, 279.
González, S. R., Aymonino, P. J., and Piro, O. E. (1984)J. Chem. Phys. 81, 625.
Holzbecher, M., Knop, O., and Falk, M. (1971)Can. J. Chem. 49, 1413.
Khanna, R. K., Brown, C. W., and Jones, L. H. (1969)Inorg. Chem. 8, 2195.
Knop, O., Westerhaus, W. C., and Falk, M. (1980)Can. J. Chem. 58, 867.
Long, L., Thomas, J. L., and Lombardi, J. C. (1978)J. Inorg. Nucl. Chem. 40, 1627.
Main, P., Fise, S. J., Hull, S. E., Lessinger, L., Germain, G., Declerq, J. P., and Woolfson, M. M. (1980) A System of Programs for the Automatic Solution of Crystal Structures from X-ray Diffraction Data (Universities of York, England and Louvain La Neuve, Belgium.
Paliani, G., Poletti, A., and Santucci, A. (1971)J. Mol. Struct. 8, 63.
Punte, G., Rigotti, G., Rivero, B. E., Podjarny, A. D., and Castellano, E. E. (1980)Acta Cryst. B 26, 1472.
Rigotti, G., Aymonino, P. J., and Varetti, E. L. (1984)J. Crystallogr. Spectrosc. Res. 14, 517.
Sabatini, A. (1967)Inorg. Chem. 6, 1756.
Sheldrick, G. H. (1976)Shelx A Program for Crystal Structure Determination (University of Cambridge, England).
Swanson, B. I., and Jones, L. H. (1971)J. Chem. Phys. 55, 4174.
Swanson, B. I., and Jones, L. H. (1974)Inorg. Chem. 13, 313.
Tables of Wave Numbers for the Calibration of Infrared Spectrometers (1961)Pure Appl. Chem. 1, 537.
Tosi, L. (1973)Compt. Rend. C 277, 335.
Varetti, E. L., and Aymonino, P. J. (1973)Inorg. Chim. Acta 7, 597.
Varetti, E. L., and Aymonino, P. J. (1982)J. Mol. Struct. 79, 281.
Author information
Authors and Affiliations
Additional information
This work is part of a doctoral dissertation, performed while a fellow of CONICET (Consejo Nacional de Investigaciones Científicas y Técnicas), República Argentina.
Rights and permissions
About this article
Cite this article
Amalvy, J.I., Varetti, E.L., Aymonino, P.J. et al. DTA-TGA, crystal and molecular structure determination and vibrational studies of potassium nitroprusside sesquiquarterhydrate, K2[Fe(CN)5NO]·1.25H2O. Journal of Crystallographic and Spectroscopic Research 16, 537–555 (1986). https://doi.org/10.1007/BF01161042
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01161042