Summary
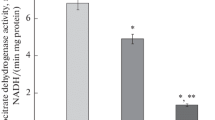
Influence of insulin on liver glycogen metabolism and on lipolysis appears to be mediated by a decreased intracellular 3′,5′-AMP concentration. Reduced formation of 3′,5′-AMP had been shown in adipose tissue incubated with insulin. The influence of insulin on 3′,5′-AMP degradation has been investigated. — 3′,5′-AMP phosphodiesterase (PDE) activity was reduced in liver, adipose tissue and, insignificantly, in skeletal muscle of insulin deficient, i.e. alloxan diabetic or starved rats. I.V. injection of a low dose of insulin (0.5 U/kg) or stimulation of endogenous insulin secretion by injection of glucose led to a rapid increase of PDE activity in these tissues. 15 min after insulin injection liver PDE activity was increased. The maximal effect occurred after 30–45 min. Renal PDE activity was not decreased in alloxan diabetes, insulin injection has been found ineffective. —In vitro, there was an activating effect of crystalline insulin on PDE purified from beef heart. Insulin concentration required for duplication of enzyme activity was of the order of 2 · 10−5 M. Treatment with actinomycin D nearly prevented stimulation of liver PDE by insulin. This may indicate that the action of insulin on PDE activity is essentially based on an increased enzyme synthesis. — Owing to the influence of insulin secretion on liver and adipose tissue 3′,5′-AMP concentration, glycogen metabolism and lipolysis can be quickly adapted to food intake.
Résumé
L'influence de l'insuline sur le métabolisme du glycogène hépatique et sur la lipolyse semble s'exercer par l'intermédiaire d'une diminution de la concentration de 3,′5′-AMP intracellulaire. Onamontré une diminution de la formation de 3′5′-AMP dans le tissu adipeux incubé avec de l'insuline. L'influence de l'insuline sur la dégradation du 3,′5′-AMP est étudiée. — L'activité de la 3,′5′-AMP-phos-phodiestérase (PDE) est diminuée dans le foie, le tissu adipeux et, de façon non-significative, dans le muscle strié des rats qui manquent d'insuline, c-à-d les rats rendus diabétiques par l'alloxane ou les rats privés de nourriture. L'injection intraveineuse d'une faible dose d'insuline (0.5 U/kg) ou la stimulation de la sécrétion d'insuline endogène par une injection de glucose provoquent une augmentation rapide de l'activité de la phosphodiestérase dans ces tissus. 15 min après l'injection d'insuline, l'activité de la phosphodiesterase du foie est augmentée. L'effet maximum est atteint après 30–45 min. L'activité de la phosphodiestérase rénale n'est pas diminuée dans le diabète alloxanique, l'injection d'insuline s'est avérée inefficace.In vitro, l'insuline cristalline a un effet activant sur la phosphodiestérase purifiée du coeur de boeuf. La concentration d'insuline requise pour doubler l'activité de l'enzyme est de l'ordre de 2 · 10−5 M. Le traitement avec actinomycin D empêche la stimulation par l'insuline de la PDE dans le foie. Ceci peut indiquer que l'action de l'insuline sur l'activité de la phosphodiestérase est essentiellement basée sur une synthèse accrue de l'enzyme. A cause de l'influence de la sécrétion d'insuline sur la concentration en 3,′5′-AMP du foie et du tissu adipeux, le métabolisme du glycogène et la lipolyse peuvent s'adapter rapidement à la prise de nourriture.
Zusammenfassung
An der Steigerung der Glykogensynthese der Leber und der Verminderung der Lipolyse durch Insulin ist eine Abnahme der 3′,5′-AMP-Konzentration wesentlich beteiligt. Die 3′,5′-AMP-Bildung ist in Fettgewebe, das mit Insulin inkubiert wird, vermindert. Insulin beeinflußt jedoch auch den 3′,5′-AMP-Abbau. -Die 3′,5′-AMP-Phosphodiesterase (PDE)-Aktivität des Fettgewebes, der Leber und, in geringerem Grade, der Skeletmuskulatur ist im Insulinmangel vermindert, d.h. bei alloxandiabetischen oder hungernden Ratten. I.v. Injektion von 0,5 E/kg Insulin oder eine erhöhte Abgabe von Insulin aus dem Pankreas nach Glucoseinjektion führen in diesen Geweben zu einem raschen Anstieg der PDE-Aktivität. Dieser ist in der Leber schon 15 min nach Insulingabe nachweisbar und erreicht nach 30–45 min sein Maximum. In der Niere ist kein Einfluß von Insulin auf die PDE-Aktivität nachweisbar. — Aus Rinderherz isolierte PDE wirdin vitro durch Insulin aktiviert, jedoch werden2 · 10−5 M zur Verdopplung der Aktivität benötigt. Actinomycin D verhindert die Steigerung der Leber-PDE-Aktivität nach Insulininjektion. So kann die Wirkung des Hormons im wesentlichen auf eine gesteigerte PDE-Synthese zurückgeführt werden. — Durch diesen Einfluß der Insulininkretion auf die 3′,5′-AMP-Konzentration in Leber und Fettgewebe können Glykogenstoffwechsel und Lipolyse rasch an die Nahrungsaufnahme angepaßt werden.
Article PDF
Similar content being viewed by others
Abbreviations
- G 6 P:
-
Glucose-6-phosphate
- UDPG:
-
UDP-glucose
- FFA:
-
non-esterifled, free fatty acids
- 3′,5′-AMP:
-
cyclic adenosine-3′,5′-monophosphate
- PDE:
-
3′,5′-AMP phosphodiesterase
References
Appleman, M.M., L. Birnbaumer, andH.N. Tores: Factors affecting the activity of muscle glyeogen synthetase. III. The reaction with adenosine triphosphate, Mg++, and cyclic 3′,5′-adenosine monophosphate. Arch. biochem.116, 39–43 (1966).
Bishop, J.S., andJ. Larner: Rapid activation-inactivation of liver uridine diphosphate glucose-glycogen transferase and phosphorylase by insulin and glucagon in vivo. J. biol. Chem.242, 1354–1356 (1967).
Blecher, M., N.S. Merlino, andJ.T. Ro'ane: Control of metabolism and lipolytic effects of cyclic 3′,5′-adenosine monophosphate in adipose tissue by insulin, methyl xanthines, and nicotinic acid. J. biol. Chem.243, 3973–3977 (1968).
Butcher, R.W., J.G.T. Sneyd, C.R. Park, andE.W. Sutherland, jr.: Effect of insulin on adenosine 3′,5′-monophosphate in the rat epididymal fat pad. J. biol. Chem.241, 1651–1653 (1966).
—,C.E. Baird, andE.W. Sutherland: Effects of lipolytic and antilipolytic substances on adenosine 3′,5′-monophosphate levels in isolated fat cells. J. biol. Chem.243, 1705–1712 (1968).
—,R.J. Ho, H.C. Meng, andE.W. Sutherland: Adenosine 3′,5′-monophosphate in biological materials. II. The measurement of adenosine 3′,5′-monophosphate in tissues and the role of the cyclic nucleotide in the lipolytic response of fat to epinephrine. J. biol. Chem.240, 4515–4523 (1965).
Butcher, R.W., andE.W. Sutherland: The effects of the catecholamines, adrenergic blocking agents, prostaglandin E1, and insulin on cyclic AMP levels in the rat epididymal fat pad in vitro. Ann. N. Y. Acad. Sci.139, 849–859 (1967).
Butcher, R.W., andE.W. Sutherland: Adenosine 3′,5′-phosphate in biological materials. I. Purification and properties of cyclic 3′,5′-nucleotide phosphodiesterase and use of this enzyme to characterize adenosine 3′,5′-phosphate in human urine. J. biol. Chem.237, 1244–1250 (1962).
Cerasi, E., andR. Luft: The plasma insulin response to glucose infusion in healthy subjects and in diabetes mellitus. Acta Endocrin. (Kbh.)55, 278–304 (1967).
Corbin, J.D., J.G.T. Sneyd, andR.W. Butcher: Control of lipolysis in fat tissne by cyclic adenylate. Fed. Proc.27, 649 (1968).
Craig, J.W., andJ. Larner: Influence of epinephrine and insulin on uridine diphosphate glucose-α-glucan transferase and phosphorylase in muscle. Nature (Lond.)202, 971–973 (1964).
Danforth, W.H.: Glyeogen synthetase activity in skeletal muscle. Interconversion of two forms and control of glyeogen synthesis. J. biol. Chem.240, 588–593 (1965).
De Wulf, H., andH.G. Hers: The stimulation of glyeogen synthesis and of glyeogen synthetase in the liver by the administration of glucose. Europ. J. Biochem.2, 50–56 (1967).
Exton, J.H., L.S. Jefferson, jr.,R.W. Butcher, andC.R. Park: Gluconeogenesis in the perfused liver. The effects of fasting, alloxan diabetes, glucagon, epinephrine, adenosine 3′,5′-monophosphate and insulin. Amer. J. Med.40, 709–715 (1966).
Friedman, D.L., andJ. Larner: Studies on uridine diphosphate glucose: α 1,4-glucan α-4-glucosyltransferase. VIII. Catalysis of the phosphorylation of muscle phosphorylase and transferase by separate enzyme?. Biochemistry4, 2261–2264 (1965).
Giinsmann, W.H., andG.E. Mortimore: Influence of glucagon and 3′,5′-AMP on insulin responsiveness of the perfused rat liver. J. biol. Chem.215, 553–559 (1968).
Goldberg, I.H., M. Rabinowitz, andE. Reich: Basis of actinomycin action. I. DNA binding and inhibition of RNA-polymerase synthetic reactions by actinomycin. Proc. nat. Acad. Sci (Wash.)48, 2094–2101 (1962).
Goldberg, N.D., C. Villar-Palasi, H. Sasko, andJ. Larner: Effects of insulin treatment on muscle 3′,5′-cyclic adenylate levels in vivo and in vitro. Biochim. biophys. Acta148, 665–672 (1967).
Goodmann, H.M., andE. Knobil: Some endocrine factors in regulation of fatty acid mobilization during fasting. Amer. J. Physiol.201, 1–3 (1961).
Gordon, R.S., jr.: Unesterified fatty acid in human blood plasma. II. The transport function of unesterified fatty acid. J. clin. Invest.36, 810–815 (1957).
Hales, C.N., andP.J. Randle: Immunoassay of insulin with insulin-antibody precipitate. Biochem. J.88, 137–146 (1963).
Hardman, J.G., J.W. Davis, andE.W. Sutherland: Measurement of guanosine 3′,5′-monophosphate and other cyclic nucleotides. Variations in urinary excretion with hormonal state of the rat. J. biol. Chem.241, 4812–4815 (1966).
Henning, H.V., B. Stumpf, B. Ohly, andW. Seubert: On the mechanism of gluconeogenesis and its regulation. III. The gluconeogenic capacity and the activities of pyruvate carboxylase and PEP-carboxylase of rat kidney and rat liver after cortisol treatment and starvation. Biochem. Z.344, 274–288 (1966).
Hizukuri, S., andJ. Larner: Studies on UDPG: α-1,4-glucan α-4-glucosyltransferase. VII. Conversion of the enzyme from glucose-6-phosphate-dependent to independent form in liver. Biochemistry3, 1783–1788 (1964).
Hoffmann, M.: Einfluß von Hormonen und Pharmaka auf die Bildung und den Abbau von cyclischem Adenosin-3′,5′-Monophosphat in der Rattenniere. Inaug.-Diss. Math.-nat. Fak. Freie Universität Berlin, 1967.
Huggett, A.S.G., andD.A. Nixon: Use of glucose oxidase, peroxidase, and o-dianisine in determination of blood and urinary glucose. Lancet1957 II, 368–370.
Huijing, F., andJ. Larner: On the mechanism of action of adenosine 3′,5′-cyclophosphate. Proc. nat. Acad. Sci. (Wash.)56, 647–653 (1966).
Jefferson, L.S., J.H. Exton, R.W. Butcher, E.W. Sutherland, andC.R. Park: Role of adenosine 3′,5′-monophosphate in the effects of insulin and antiinsulin serum on liver metabolism. J. biol. Chem.243, 1031–1038 (1068).
Jungas, R.L.: Role of cyelie-3′,5′-AMP in the response of adipose tissue to insulin. Proc. nat. Acad. Sci. (Wash.)56, 757–763 (1966).
Jungas, R.L., andE.G. Ball: Studies on the metabolism of adipose tissue.XII, The effects of insulin and epinephrine on free fatty acid and glycerol production in the presence and absence of glucose. Biochemistry2, 383–388 (1963).
Korner, A., andM.S. Raben: Effect of aminonucleoside and puromycin on insulin and epinephrine control of fatty acid release from adipose tissue. Nature (Lond.)203, 1287–1289 (1964).
Kovacev, V.P., andR.O. Scow: Effect of hormones on fatty acid release by rat adipose tissue in vivo. Amer. J. Physiol.210, 1199–1208 (1966).
Krebs, E.G., D.S. Love, G.E. Bratvold, K.A. Trayser, W.L. Meyer, andE.H. Fischer: Purification and properties of rabbit skeletal muscle phosphorylase b kinase. Biochemistry3, 1022–1033 (1964).
Larner, J.: Hormonal and nonhormonal control of glycogen metabolism. Trans. N.Y. Acad. Sci.29, 192–209 (1967).
Lineweaver, H., andD. Burk: The determination of enzyme dissociation constants. J. Amer. chem. Soc.56, 658–666 (1934).
Lowry, O.H., N.J. Rosenbrough, A.L. Rarr, andR.J. Randall: Protein measurement with the Folin phenol reagent. J. biol. Chem.193, 265–275 (1951).
Maddaiah, V.T., andN.B. Madsen: Kinetics of purified liver phosphorylase. J. biol. Chem.241, 3873–3881 (1966).
Madsen, N.B.: The inhibition of glycogen phosphorylase by uridine diphosphate glucose. Biochem. biophys. Res. Comm.6, 310–315 (1961).
Möllering, H., andH.U. Bergmeyer: In Bergmeyer, H.U.: Methoden der enzymatischen Analyse. Weinheim: Verlag Chemie 1962.
Müller-Oerlinghausen, B., U. Schwabe, A. Hasselblatt, andF.H. Schmidt: Activity of 3′,5′-AMP phosphodiesterase in liver and adipose tissue of normal and diabetic rats. Life Sciences7, part II, 593–598 (1968).
Neubert, D., H. Helge, andR. Bass: Einbau von Thymidin in die Deoxyribonueleinsäure von Mitochondrien. Naunyn-Schmiedeberg's Arch. exp. Path. Pharmak.252, 258–268 (1965).
Pätau, K.: Zur statistischen Beurteilung von Messungsreihen. (Eine t-Tafel). Biol. Zbl.63, 152–168 (1943).
Rall, T.W., J.W. Craig, andJ. Larner: unpublished observations, cited by (29).
Rizack, M.A.: Activation of an epinephrine-sensitive lipolytic activity from adipose tissue by adenosine 3′,5′-phosphate. J. biol. Chem.239, 392–395 (1964).
—: An epinephrine-sensitive lipolytic activity in adipose tissue. J. biol. Chem.236, 657–662 (1961).
Rodbell, M., andA.B. Jones: Metabolism of isolated fat cells. III. The similar inhibitory action of phospholipase C (clostridium perfringens α toxin) and of insulin on lipolysis stimulated by lipolytic hormones and theophylline. J. biol. Chem.241, 140–142 (1966).
- The actions of insulin and catabolic hormones on the plasma membrane of fat cells. Biochem. J.105, 2P (1967).
Schultz, G., G. Senft, andK. Munske: Der Einfluß von Insulin auf die enzymatische Regulation der Glycogenolyse. Naturwissenschaften53, 529 (1966).
— — —: Die Abhängigkeit glykogenauf- und-abbauender enzymatischer Reaktionen von der Insulininkretion. Naunyn-Schmiedebergs Arch. Pharmak. exp. Path257, 62–63 (1967).
Senft, G., G. Schultz, K. Munske, andM. Hoffmann: Effects of glucocortieoids and insulin on 3′,5′-AMP phosphodiesterase activity in adrenalectomized rats, Diabetologia4, 330–335 (1968)
—,W. Losert, G. Schultz, R. Sitt, andH.K. Bartelheimer: Ursachen der Störungen im Kohlenhydratstoffwechsel unter dem Einfluß sulfonamidierter Diuretica. Naunyn-Schmiedebergs Arch. Pharmak. exp. Path.255, 369–382 (1966).
Seltzer, H.S., E.W. Allen, A.L. Herron, jr., andM.T. Brennan: Insulin secretion in response to glycemic Stimulus: Relation of delayed initial release to carbohydrate intolerance in mild diabetes mellitus. J. clin. Invest.46, 323–335 (1967).
Shimazu, T.: Glycogen synthetase activity in liver: Regulation by the autonomic nerves. Science156, 1256–1257 (1967).
Sitt, R., W. Losert, G. Schultz, H. Kaess, andG. Senft: Der Einfluß von Insulin auf den Kaliumtransport in der Skeletmuskulatur. Naunyn-Schmiedebergs Arch. Pharmak. exp. Path.255, 398–406 (1966).
Søvik, O.: The action of insulin on glycogen synthesis in rat diaphragm. Acta physiol. scand.68, 246–254 (1966).
—:, andO. Walaas: Insulin stimulation of glycogen synthesis in the isolated rat diaphragm in the absence and in the presence of puromycin and actinomycin D. Nature (Lond.)202, 396–397 (1964).
Steiner, D.F., andJ. King: Induced synthesis of hepatic uridine diphosphate glucose-glycogen glucosyltransferase after administration of insulin to alloxan-diabetic rats. J. biol. Chem.239, 1292–1298 (1964).
—V. Rauda, andR.H. Williams: Effects of insulin, glucagon, and glucocortieoids upon hepatic glycogen synthesis from uridine diphosphate glucose. J. biol. Chem.236, 299–304 (1961).
Sutherland, E.W., R.W. Butcher, G.A. Robison, andJ.G. Hardman: The role of adenosine 3′,5′-monophosphate in hormone action. In: Wirkungsmechanismen der Hormone, 18. Colloqu. Ges. Physiol. Chem., Mosbach 1967, p. 1, ed. byP. Karlson. Berlin-Heidelberg-New York: Springer 1967.
Torres, H.N., L.R. Marechal, E. Bernard, andE. Belocopitow: Control of muscle glycogen phosphorylase activity by insulin. Biochim. biophys. Acta156, 206–209 (1968).
Vaughan, M., J.E. Berger, andD. Steinberg: Hormone-sensitive lipase and monoglyoeride lipase activities in adipose tissue. J. biol. Chem.239, 401–409 (1964).
Villar-Palasi, C., andJ. Larner: Levels of activity of the enzymes of the glycogen cycle in rat tissues. Arch, biochem.86, 270–273 (1960).
—, andJ.I. Wenger: In vivo effect of insulin on muscle glycogen synthetase. Identification of the action pathway. Fed. Proc.26, 563 (1967).
Weber, G.: Study and evaluation of regulation of enzyme activity and synthesis in mammalian liver. In: Advances in Enzyme Regulation, Vol. I, p. 1, ed. by G. Weber. Oxford, London, New York, Paris: Pergamon Press 1963.
Wool, I.G., andPh. Cavicchi: Protein synthesis by skeletal muscle ribosomes. Effect of diabetes and insulin. Biochemistry6, 1231–1242 (1967).
Author information
Authors and Affiliations
Additional information
This study was supported by the Deutsche Forschungsgemeinschaft.
Deceased October 31, 1967.
Rights and permissions
About this article
Cite this article
Senft, G., Schultz, G., Munske, K. et al. Influence of insulin on cyclic 3′,5′-AMP phosphodiesterase activity in liver, skeletal muscle, adipose tissue, and kidney. Diabetologia 4, 322–329 (1968). https://doi.org/10.1007/BF01211766
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01211766