Summary
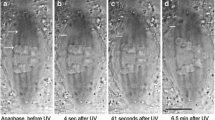
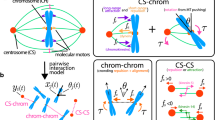
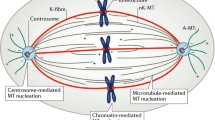
The “Pac-Man” model for explaining chromosome movement is based on three main tenets: (i) the force that moves chromosomes is generated at the kinetochore; (ii) disassembly of the microtubules (MTs) of the kinetochore fibre generates poleward movement; and (iii) the energy required for this movement comes from MT disassembly. We show that these tenets are not valid in some and perhaps many situations. Thus, the Pac-Man model is inadequate and misleading as the central basis for explaining chromosomal motion generally. We argue that multiple mechanisms are involved in mitotic function and that a contractile/elastic spindle matrix is likely involved not only in anchoring kinetochore fibres, but also by exerting force on them. This view of the spindle matrix shares some features with the “tensegrity” model already formulated as a basis for understanding interphase cell behaviour.
Similar content being viewed by others
References
Ault JG, Nicklas RB (1989) Tension, microtubule rearrangements, and the proper distribution of chromosomes in mitosis. Chromosoma 98: 33–39
Beckerle MC, Porter KR (1983) Analysis of the role of microtubules and actin in erythrophore intracellular motility. J Cell Biol 96: 354–362
Czaban BB, Forer A (1992) Rhodamine-labelled phalloidin stains components in the chromosomal spindle fibres of crane-fly spermatocytes andHaemanthus endosperm cells. Biochem Cell Biol 70: 664–676
Darnell J, Lodish H, Baltimore D (1990) Molecular cell biology, 2nd edn. Scientific American Books/WH Freeman, New York
Forer A (1965) Local reduction of spindle fiber birefringence in livingNephrotoma suturalis (Loew) spermatocytes induced by ultraviolet microbeam irradiation. J Cell Biol 25: 95–117
— (1988) Do anaphase chromosomes chew their way to the pole or are they pulled by actin? J Cell Sci 91: 449–453
—, Wilson P (1994) A model for chromosome movement during mitosis. Protoplasma 179: 95–105
Goldstein LSB (1993) Functional redundancy in mitotic force generation. J Cell Biol 120: 1–3
Gorbsky GJ, Sammak PJ, Borisy GG (1988) Microtubule dynamics and chromosome motion visualized in living anaphase cells. J Cell Biol 106: 1185–1192
Hepler PK, Palevitz BA (1986) Metabolic inhibitors block anaphase A in vivo. J Cell Biol 102: 1995–2005
—, Wolniak SM (1984) Membranes in the mitotic apparatus: their structure and function. Int Rev Cytol 90: 169–238
Ingber DE (1993) Cellular tensegrity: defining new rules of biological design that govern the cytoskeleton. J Cell Sci 104: 613–627
Inoue S, Salmon ED (1995) Force generation by microtubule assembly/disassembly in mitosis and related movements. Mol Biol Cell 6: 1619–1640
Koshland D (1994) Mitosis: back to the basics. Cell 77: 951–954
—, Mitchison TM, Kirschner MW (1988) Polewards chromosome movement driven by microtubule depolymerization in vitro. Nature 331: 499–504
Luby KJ, Porter KR (1980) The control of pigment migration in isolated erythrophores ofHolocentrus ascensionis (Osbeck). I. Energy requirements. Cell 21: 13–23
McNeill PA, Berns MW (1981) Chromosome behaviour after laser microbeam irradiation of a single kinetochore in mitotic PtK2 cells. J Cell Biol 88: 543–553
Mitchison TJ, Salmon ED (1992) Poleward kinetochores fibre movement occurs during both metaphase and anaphase-A in newt lung cell mitosis. J Cell Biol 119: 569–582
Mole-Bajer J, Bajer A, Owczarzak A (1975) Chromosome movements in prometaphase and aster transport in the newt. Cytobios 13: 45–65
Nicklas RB (1989) The motor for poleward chromosome movement in anaphase is in or near the kinetochore. J Cell Biol 109: 2245–2255
Östergren G (1951) The mechanism of co-orientation in bivalents and multivalents. The theory of orientation by pulling. Hereditas 37: 85–156
Pickett-Heaps JD (1991) Cell division in diatoms. Int Rev Cytol 128: 63–108
—, Bajer A (1978) Mitosis: an argument for multiple mechanisms achieving chromosomal movement. Cytobios 19: 171–180
—, Spurck TP (1982) Studies on kinetochore function in mitosis. II. The effects of metabolic inhibitors on mitosis and cytokinesis in the diatom.Hantzschia amphioxys. Eur J Cell Biol 28: 83–91
—, Tippit DH, Porter KR (1982) Rethinking mitosis. Cell 29: 729–744
—, Spurck TP, Tippit DH (1984) Chromosome motion and the spindle matrix. J Cell Biol 99: 137s-143s
Porter KR (1976) Motility in cells. In: Goldman CR, Pollard T, Rosenbaum J (eds) Cell motility. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY, pp 1–26
—, McNiven MA (1982) The cytoplast: a unit structure in chromatophores. Cell 29: 23–32
—, Beckerle M, McNiven M (1983) The cytoplasmic matrix. Mod Cell Biol 2: 259–302
Rieder CL (1991) Mitosis: towards a molecular understanding of chromosome behavior. Curr Opin Cell Biol 3: 59–66
—, Alexander SP (1990) Kinetochores are transported polewards along a single astral microtubule during chromosome attachment to the spindle in newt lung cells. J Cell Biol 110: 81–85
—, Salmon ED (1994) Motile kinetochores and polar ejection forces dictate chromosome position on the vertebrate mitotic spindle. J Cell Biol 124: 223–233
Salmon ED (1989) Microtubule dynamics and chromosome movement. In: Hyams JS, Brinkley BR (eds) Mitosis: molecules and mechanisms. Academic Press, New York, pp 119–182
Sampson K, Pickett-Heaps JD, Forer A (1996) Cytochalasin D blocks chromosomal attachment to the spindle in the green algaOedogonium. Protoplasma (in press)
Schibler MJ, Pickett-Heaps JD (1980) Mitosis inOedogonium: spindle microfilaments and the origin of the kinetochore fibre. Eur J Cell Biol 22: 687–698
— — (1987) The kinetochore fibre structure in the acentric spindles ofOedogonium. Protoplasma 137: 29–44
Schrader F (1944) Mitosis. Columbia University Press, New York
Spurck T, Pickett-Heaps JD (1987) On the mechanism of anaphase A: evidence that ATP is needed for microtubule disassembly and not generation of polewards force. J Cell Biol 105: 1691–1705
— —, Klymkowsky MW (1986a) Metabolic inhibitors and mitosis. I. Effects of dinitrophenol/deoxyglucose and nocodazole on the live spindle. Protoplasma 131: 47–59
— — — (1986b) Metabolic inhibitors and mitosis. II. Effects of dinitrophenol/deoxyglucose and nocodazole on the microtubule cytoskeleton. Protoplasma 131: 60–74
Tippit DH, Pickett-Heaps JD, Leslie R (1980) Cell division in two large pennate diatomsHantzschia andNitzschia. III. A new proposal for kinetochore function during prometaphase. J Cell Biol 86: 402–416
Wadsworth P, Salmon ED (1988) Spindle microtubule dynamics: modulation by metabolic inhibitors. Cell Motil Cytoskeleton 11: 97–105
Wilson PJ, Forer A (1988) Ultraviolet microbeam irradiation of chromosomal spindle fibres shears microtubules and permits study of new free ends in vivo. J Cell Sci 91: 455–468
— —, Leggiadro C (1994) Evidence that kinetochore microtubules in crane-fly spermatocytes disassemble during anaphase primarily at the poleward end. J Cell Sci 107: 3015–3027
Zar JH (1984) Biostatistical analysis, 2nd edn. Prentice Hall, Englewood Cliffs
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pickett-Heaps, J.D., Forer, A. & Spurck, T. Rethinking anaphase: where “Pac-Man” fails and why a role for the spindle matrix is likely. Protoplasma 192, 1–10 (1996). https://doi.org/10.1007/BF01273239
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01273239