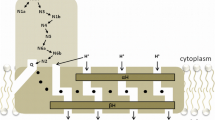
Abstract
In the presence of effectors variations in the two recognized activities of the plasma membrane NADH-oxidoreductase system were studied in separate, specificin vitro assays. We report here that ubiquinone analogues that contain a short, less hydrophobic side chain than coenzyme Q-10 dramatically stimulate the NADH-oxidase activity of isolated rat liver plasma membranes whereas they show no effect on the reductase activity of isolated membranes. If measured in assays of the NADH∶ferricyanide reductase of living cultured cells these compounds have only a limited effect; the oxidase activity of whole cells is not measurable in our hands. We have furthermore identified selective inhibitors of both enzyme activities. In particular, the NADH-oxidase activity can be significantly inhibited by structural analogues of ubiquinone, such as capsaicin and resiniferatoxin. The NADH∶ferricyanide reductase, on the other hand, is particularly sensitive to pCMBS, indicating the presence of a sulfhydryl group or groups at its active site. The identification of these specific effectors of the different enzyme activities of the PMOR yields further insights into the function of this system.
Similar content being viewed by others
Abbreviations
- DCIP:
-
2,6-dichlorophenol-indophenol
- DTNB:
-
5,5′-dithio-bis(2-nitrobenzoic acid)
- FAC:
-
ferric ammonium citrate
- pCMBS:
-
p-chloromercuriphenylsulfonic acid
- PMOR:
-
plasma membrane NADH-oxidoreductase
- coenzyme Q-0:
-
2,3-dimethoxy-5-methyl-1,4-benzoquinone
- SH:
-
sulfhydryl group
References
Afanas'ev, I. B., Korkina, L. G., Suslova, T. B., and Soodaeva, S. K. (1990).Arch. Biochem. Biophys. 281, 245–250.
Aledort, L. M., Troup, S. B., and Weed, R. I. (1968).Blood 31, 471–479.
Beaufay, H., Amar-Costesec, A., Feytmans, E., Thinès-Sempoux, D., Wibo, M., Robbi, M., and Berthet, J. (1974).J. Cell Biol. 61, 188–200.
Bérczi, A., Sizensky, J. A., Crane, F. L., and Faulk, W. P. (1991).Biochim. Biophys. Acta 1073, 562–570.
Bradford, M. M. (1976).Anal. Biochem. 72, 248–254.
Brandt, U., and Trumpower, B. (1994).Crit. Rev. Biochem. Mol. Biol. 29, 165–197.
Braun, B. S., Benbow, U., Lloyd-Williams, P., Bruce, J. M., and Dutton, P. L. (1986).Methods Enzymol. 125, 119–129.
Brightman, A. O., Wang, J., Miu, R. K.-m., Sun, I. L., Barr, R., Crane, F. L., and Morré, D. J. (1992).Biochim. Biophys. Acta 1105, 109–117.
Clark, M. G., Partick, E. J., Patten, G. S., Crane, F. L., Löw, H., and Grebing, C. (1981).Biochem. J. 200, 565–572.
Crane, F. L., Roberts, H., Linnane, A. W., and Löw, H. (1982).J. Bioenerg. Biomembr. 14, 191–205.
Crane, F. L., Sun, I. L., Clark, M. G., Grebing, C., and Löw, H. (1985).Biochim. Biophys. Acta 811, 233–264.
Crane, F. L., Sun, I. L., Barr, R., and Löw, H. (1991).J. Bioenerg. Biomembr. 23, 773–803.
Crane, F. L., Sun, I. L., and Sun, E. E. (1993).Clin. Invest. 71, S55-S59.
Desjardins, P., Frost, E., and Morais, R. (1985).Mol. Cell. Biol. 5, 1163–1169.
Ellem, K. A. O., and Kay, G. F. (1983).Biochem. Biophys. Res. Commun. 112, 183–190.
Goldenberg, H., Crane, F. L., and Morré, D. J. (1979).J. Biol. Chem. 254, 2491–2498.
Hochstein, P. (1983).Fundam. Appl. Toxicol. 3, 215–217.
Inman, R. S., and Wessling-Resnick, M. (1993).J. Biol. Chem. 268, 8521–8528.
King, M. P., and Attardi, G. (1989).Science 246, 500–503.
Larm, J. A., Vaillant, F., Linnane, A. W., and Lawen, A. (1994).J. Biol. Chem. 269, 30097–30100.
Larm, J. A., Wolvetang, E. J., Vaillant, F., Martinus, R. D., Lawen, A., and Linnane, A. W. (1995).Protoplasma 184, 173–180.
Lawen, A., Martinus, R. D., McMullen, G. L., Nagley, P., Vaillant, F., Wolvetang, E. J., and Linnane, A. W. (1994).Mol. Aspects Med. 15, s13-s27.
Lenaz, G., Pasquali, P., Bertoli, E., and Parenti-Castelli, G. (1975).Arch. Biochem. Biophys. 169, 217–226.
Löw, H., Grebing, C., Lindgren, A., Tally, M., Sun, I. L., and Crane, F. L. (1987).J. Bioenerg. Biomembr. 19, 535–549.
Luft, R. (1994).Proc. Natl. Acad. Sci. USA 91, 8731–8738.
Mahler, H. R. (1955).Methods Enzymol. 2, 688–693.
Martinus, R. D., Linnane, A. W., and Nagley, P. (1993).Biochem. Mol. Biol. Int. 31, 997–1005.
Morré, D. J., and Brightman, A. O. (1991).J. Bioenerg. Biomembr. 23, 469–489.
Morré, D. J., and Morré, D. M. (1989).BioTechniques 7, 946–958.
Nagley, P., Zhang, C., Martinus, R. D., Vaillant, F., and Linnane, A. W. (1993). InMitochondrial DNA in Human Pathology (DiMauro, S., and Wallace, D. C., eds.), Raven Press, New York, pp. 137–157.
Naruta, Y. (1980).J. Am. Chem. Soc. 102, 3774–3783.
Navas, P., Estévez, A., Burón, M. I., Villalba, J. M., and Crane, F. L. (1988).Biochem. Biophys. Res. Commun. 154, 1029–1033.
Navas, P., Nowack, D. D., and Morré, D. J. (1989).Cancer Res. 49, 2147–2156.
Nishinaka, Y., Aramaki, Y., Yoshida, H., Masuya, H., Sugawara, T., and Ichimori, Y. (1993).Biochem. Biophys. Res. Commun. 193, 554–559.
Nyormoi, O., Klein, G., Adams, A., and Dombos, L. (1973).Int. J. Cancer 12, 396–408.
Sottocasa, G. L., Kuylenstierna, B., Ernster, L., and Bergstrand, A. (1967).J. Cell Biol. 32, 415–438.
Sun, I. L., Toole-Simms, W., Crane, F. L., Golub, E. S., Díaz de Pagán, T., Morré, D. J., and Löw, H. (1987).Biochem. Biophys. Res. Commun. 146, 976–982.
Sun, I. L., Crane, F. L., Grebing, C., and Löw, H. (1984a).J. Bioenerg. Biomembr. 16, 583–595.
Sun, I. L., Crane, F. L., Löw, H., and Grebing, C. (1984b).J. Bioenerg. Biomembr. 16, 209–221.
Sun, I. L., Sun, E. E., and Crane, F. L. (1992a).Biochem. Biophys. Res. Commun. 189, 8–13.
Sun, I. L., Sun, E. E., Crane, F. L., Morré, D. J., Lindgren, A., and Löw, H. (1992b).Proc. Natl. Acad. Sci. USA 89, 11126–11130.
Sun, I. L., Sun, E. E., and Crane, F. L. (1995).Protoplasma 184, 214–219.
Vaillant, F., and Nagley, P. (1995).Hum. Mol. Genet. 4, 903–914.
Vaillant, F., Loveland, B. E., Nagley, P., and Linnane, A. W. (1991).Biochem. Int. 23, 571–580.
van Iwaarden, P. R., Driessen, A. J. M., and Konings, W. N. (1992).Biochim. Biophys. Acta 1113, 161–170.
VanSteveninck, J., Weed, R. I., and Rothstein, A. (1965).J. Gen. Physiol. 48, 617–632.
Villalba, J. M., Navarro, F., Córdoba, F., Serrano, A., Arroyo, A., Crane, F. L., and Navas, P. (1995).Proc. Natl. Acad. Sci. USA 92, 4887–4891.
Wan, Y.-R, Williams, R. H., Folkers, K., Leung, K. H., and Racker, E. (1975).Biochem. Biophys. Res. Commun. 63, 11–15.
Wanders, R. J. A., Kos, M., Roest, B., Meijer, A. J., Schrakamp, G., Heymans, H. S. A., Tegelaers, W. H. H., van den Bosch, H., Schutgens, R. B. H., and Tager, J. M. (1984).Biochem. Biophys. Res. Commun. 123, 1054–1061.
Wolvetang, E. J., Johnson, K. L., Krauer, K., Ralph, S. J., and Linnane, A. W. (1994).FEBS Lett. 339, 40–44.
Wolvetang, E. J., Larm, J. A., Mousoulas, P. and Lawen, A. (1996).Cell Growth Differ. 7, in press.
Wróblewski, F., and LaDue, J. S. (1955).Proc. Soc. Exp. Biol. Med. 90, 210–213.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vaillant, F., Larm, J.A., McMullen, G.L. et al. Effectors of the mammalian plasma membrane NADH-oxidoreductase system. Short-chain ubiquinone analogues as potent stimulators. J Bioenerg Biomembr 28, 531–540 (1996). https://doi.org/10.1007/BF02110443
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02110443