Abstract
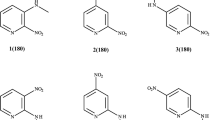
A series of 1,8-naphthalenedicarboximide derivatives containing substituents of different steric and electronic nature were studied by X-ray diffraction analysis.Ab initio quantumchemical calculations in the HF/3–21G approximation demonstrated the high conformational flexibility of the imide tetrahydro ring in the molecules of these compounds. The electronic nature of the substituents has no effect on the geometry and conformational flexibility of naphthalenedicarboximides due to weak conjugation between the imide and naphthalene fragments in the molecules. However, the steric effects of the bulky substituents noticeably affect the equilibrium geometry of the imide ring by increasing its conformational flexibility.
Similar content being viewed by others
References
I. Grabchev and T. Konstantinova,Dyes and Pigments, 1997,33, 197.
K. Wojciechowski,Dyes and Pigments, 1993,22, 239.
R. J. Cherney, S. G. Swartz, A. D. Patten, E. Akamike, and J. H. Sun,Bioorg. Med. Chem. Lett., 1997,7, 163.
M. F. Brana, J. M. Castellano, M. Moran, M. J. P. Devega, and D. Perron,Anti-cancer Drug Design, 1996,11, 297.
R. G. Baughman, S.-C. Chang, R. E. Utecht, and D. E. Lewis,Acta Crystallogr., 1995,C51, 1189.
I. Grabchev and T. Philipova,Ind. J. Chem., 1997,B36, 264.
T. Z. Filipova, I. Grabchev, and I. Petkov,J. Polym. Sci., 1997,A35, 1069.
P. N. Prasad and D. J. Williams,Introduction to Non-Linear Effects in Molecules and Polymers, Wiley Interscience, New York-London, 1990.
A. K. Agrawal and S. A. Jenekhe,Macromolecules, 1993,26, 895.
B. A. Reinhardt,TRIR, 1993,1, 4.
M. Langlois, J. L. Soulier, B. Bremont, S. Shen, V. Rampillon, and A. Giudice,Bioorg. Med. Chem. Lett., 1992,2, 691.
O. V. Shishkin and I. I. Ponomarev,Izv. Akad. Nauk, Ser. Khim., 1997, 64 [Russ. Chem. Bull., 1997,46, 59 (Engl. Transl.)].
B. M. Krasovitskii and N. I. Mal'tseva,Organicheskie lyuminofory na osnove naftalevoi kisloty [Organic Luminophores Based on Naphthalic Acid], Inst. Monokristallov, Kharkov, 1973, 61, (in Russian).
V. A. Strel'tsov and V. E. Zavodnik,Kristallografiya, 1989,34, 1369 [Sov. Phys.-Crystailogr., 1989,34 (Engl. Transl.)].
R. H. Blessing,J. Appl. Cryst., 1989,22, 396.
G. M. Sheldrick,SHELXTL PLUS. PC Version, A System of Computer Programs for the Determination of Crystal Structure from X-ray Diffraction Data, Rev. 5.02, Siemens Analytical X-ray Instruments Inc., Göttingen, (Germany), 1994.
M. W. Schmidt, K. K. Baldridge, J. A. Boatz, S. T. Elbert, M. S. Gordon, M. Dupois, and J. A. Montgomery,J. Comput. Chem., 1993,14, 1347.
G. R. Clark and S. B. Hall,Acta Crystallogr., 1989,C45, 67.
A. Houlton, D. M. P. Mingos, and D. J. Williams,Transition Met. Chem., 1994,19, 653.
Yu. V. Zefirov and P. M. Zorkii,Usp. Khim., 1989,58, 713 [Russ. Chem. Rev., 1989,58 (Engl. Transl.)].
C. J. Easton, J. M. Gulbis, B. F. Hoskins, I. M. Scharfbillig, and E. R. T. Tiekink,Z. Kristallogr., 1992,199, 249.
S. V. Lindeman, I. I. Ponomarev, and A. L. Rusanov,Acta Crystallogr., 1995,C51, 2157.
Y. Dromzee, J. Kossanyi, V. Wintgens, and P. Valat,Z. Kristallogr., 1995,210, 760.
R. A. Batchelor, C. A. Hunter, and J. Simpson,Acta Crystallogr., 1997,C53, 1117.
C. P. Brock and J. D. Dunitz,Acta Crystallogr., 1982,B38, 2218.
V. Bertolasi, P. Gilli, V. Ferretti, and G. Gilli,Acta Crystallogr., 1995,B51, 1004.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 1, pp. 68–73, January, 2000.
Rights and permissions
About this article
Cite this article
Kovalevsky, A.Y., Ponomarev, I.I., Antipin, M.Y. et al. Influence of steric and electronic effects of substituents on the molecular structures and conformational flexibility of 1,8-naphthalenedicarboximides. Russ Chem Bull 49, 70–76 (2000). https://doi.org/10.1007/BF02499068
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02499068