Abstract
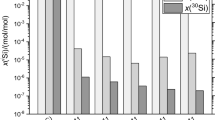
The solid solubility of the 3d metals Cr, Mn, Fe, Co and Ni in high-purity silicon was studied by INAA and electron paramagnetic resonance techniques in the temperature range of ≊600°C–1250°C. The solubility increases with atomic number from Cr to Mn and more distinctly from Co to Ni. For Mn, Fe and Co the solubilities are nearly the same within the experimental errors. An enthalpy of formation (ΔH) of about 2.8 eV was determined for Cr, Mn, Fe and Co, whereas for Ni 1.7 eV was derived. Relatively large diffusion coefficients were estimated from the rather short times, in which saturation of the solid solution was reached. From these observations and from the results of the EPR measurements it is concluded that the 3d metals occupy predominantly interstitial sites in the silicon lattice in thermal equilibrium.
Similar content being viewed by others
References
F. A. TRUMBORE, Bell Syst. Tech. J., 39 (1960) 205.
E. WEBER, H. G. RIOTTE, Appl. Phys. Letters, 33 (1978) 433.
R. N. H. HALL, J. H. RACETTE, J. Appl. Phys., 35 (1964) 379.
H. G. RIOTTE, U. HERPERS, E. WEBER, Radiochim. Acta, 27 (1980) 209.
E. WEBER, N. WIEHL, to be published.
E. WEBER, H. G. RIOTTE, J. Appl. Phys., 51 (1980) 1484.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wiehl, N., Herpers, U. & Weber, E. Study on the solid solubility of transition metals in high-purity silicon by instrumental neutron activation analysis and anticompton-spectrometry. J. Radioanal. Chem. 72, 69–78 (1982). https://doi.org/10.1007/BF02516775
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02516775