Summary
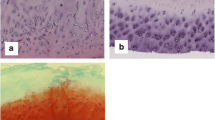
Very thin slices of the superficial and deep layers of osteophytic and apparently normal articular cartilage from six human osteoarthritic femoral heads were digested with papain. The digests were analyzed for keratan sulfate content using an enzyme-linked immunosorbent assay (ELISA) with inhibition step, and for chondroitin sulfate and collagen contents using biochemical assays. Although there were marked differences in Safranin-O staining of the superficial and deep layers of osteophytic cartilage, these two layers had identical high ratios of chondroitin sulfate/collagen. Keratan sulfate was present only in small amounts in osteophytic cartilage. However, the deeper layer contained significantly more of this glycosaminoglycan. The deeper layer of articular cartilage contained approximately twice as much chondroitin sulfate and six times more keratan sulfate relative to collagen than the superficial layer. The results of this study showed that this new sensitive approach, which requires as little as 200 μg wet cartilage as starting material, provides important qualitative and quantitative information about the major constituents of the matrix.
Similar content being viewed by others
References
Sokoloff L (1969) The biology of degenerative joint disease. University of Chicago Press, Chicago
McCarty DJ (1979) Arthritis and allied conditions, 9th ed., Lea and Febiger, Philadelphia
Jeffery AK (1975) Osteophytes and the osteoarthritic femoral head. J Bone Joint Surg [Am] 57B:314–324
Goldberg VM, Norby DP, Sachs BL, Moskowitz RW, Malemud CJ (1984) Correlation of histopathology and sulfated proteoglycans in human osteoarthritic hip cartilage. J Orthop Res 1:302–312
Sachs BL, Goldberg VM, Getzy LL, Moskowitz RW, Malemud CJ (1982) A histopathologic differentiation of tissue types in human osteoarthritic cartilage. J Rheumatol 9:210–216
Malemud CJ, Moskowitz RW, Goldberg VM (1979) DNA synthesis and cell proliferation by topographically distinct components of human osteoarthritic cartilage. Arthritis Rheum 22:637
Sweet MBE, Thonar EJ-MA, Immelman AR, Solomon L (1977) Biochemical changes in progressive osteoarthritis. Ann Rheum Dis 36:387–398
Malemud CJ, Goldberg VM, Moskowitz RW, Getzy LL, Papay RS, Norby DP (1982) Biosynthesis of proteoglycan in vitro by cartilage from human osteochondrophytic spurs. Biochem J 206:329–341
Malemud CJ, Moskowitz RW, Goldberg VM (1984) Biosynthesis of sulfated proteoglycan in vitro by cells derived from human osteochondrophytic spurs of the femoral head. Connective Tissue Res 12:319–355
Caterson B, Calabro T, Donohue PJ, Jahnke MR (1986) Monoclonal antibodies against cartilage proteoglycan and link protein. In: Kuettner KE, Schleyerbach R, Hascall VC (eds) Articular cartilage biochemistry. Raven Press, New York, p 59
Thonar EJ-MA, Lenz ME, Klintworth GK, Caterson B, Pachman LM, Glickman P, Katz R, Huff J, Kuettner KE (1985) Quantification of keratan sulfate in blood as a marker of cartilage catabolism. Arthritis Rheum 28:1367–1376
Solomon L, Schnitzler CM, Browett JP (1982) Osteoarthritis of the hip: the patient behind the disease. Ann Rheum Dis 41:118–125
Meachim G (1972) Light microscopy of India ink preparations of fibrillated cartilage. Ann Rheum Dis 31:457–464
Rosenberg L (1971) Chemical basis for the histological use of Safranin-O in the study of articular cartilage. J Bone Joint Surg [Am] 53A:69–82
Immelman AR, Sweet MBE, Thonar EJ-MA (1978) Automated procedure for the quantitative determination of hexuronic acid. S Afr Lab Tech 24:35–36
Bitter T, Muir H (1962) A modified uronic acid carbazole reaction. Anal Biochem 4:330–340
Schwartz DE, Choi Y, Sandell LJ, Hansen WR (1985) Quantitative analysis of collagen, protein and DNA in fixed, paraffin-embedded and sectioned tissue. Histochem J 17:655–663
Schenk RK, Eggli PS, Hunziker EB (1986) Articular cartilage morphology. In: Kuettner KE, Schleyerbach R, Hascall VC (eds) Articular cartilage biochemistry. Raven Press, New York, p 3
Maroudas A, Evans H, Almeida L (1973) Cartilages of the hip joint. Topographical variations of glycosaminoglycan content in normal and fibrillated tissues. Ann Rheum Dis 32:1–9
Maroudas A, Muir H, Wingham J (1969) The correlation of fixed negative charge with glycosaminoglycan content of human articular cartilage. Biochim Biophys Acta 177:492–500
Stockwell RP, Scott JE (1967) Distribution of acid glycosaminoglycans in human articular cartilage. Nature 215:1376–1378
Bayliss MT, Venn M, Maroudas A, Ali SY (1983) Structure of proteoglycans from different layers of human articular cartilage. Biochem J 209:387–400
Aydelotte MB, Kuettner KE (1984) A histochemical study of distribution of proteoglycans in cultures of adult bovine chondrocytes in agarose gel. Orthop Trans 9:117
Zanetti M, Ratcliffe A, Watt FM (1985) Two subpopulations of differentiated chondrocytes identified with a monoclonal antibody to keratan sulfate. J Cell Biol 101:53–59
Aydelotte MB, Kuettner KE (1986) A comparative study of cultures of chondrocytes isolated from different depths of articular cartilage. Orthop Trans 10:102
Aydelotte MB, Kuettner KE (1986) Morphological differences between cultured chondrocytes derived from superficial and deep zones of articular cartilage. Orthop Trans 11:404
Aydelotte MB, Schleyerbach R, Zeck BJ, Kuettner KE (1986) Articular chondrocytes cultured in agarose gel for study of chondrocytic chondrolysis. In: Kuettner KE, Schleyerbach R, Hascall VC (eds) Articular cartilage biochemistry, Raven Press, New York, p 235
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Williams, J.M., Katz, R.J., Childs, D. et al. Keratan sulfate content in the superficial and deep layers of osteophytic and nonfibrillated human articular cartilage in osteoarthritis. Calcif Tissue Int 42, 162–166 (1988). https://doi.org/10.1007/BF02556329
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02556329