Summary
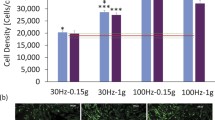
A tissue culture system has been developed which can mechanically stimulate cells growing on a highly elastic plastic substratum in a 24-well cell growth chamber. The collagen-coated substratum to which the cells attach and grow in the Mechanical Cell Stimulator (Model I) can be repetitively stretched and relaxed by stepper motor with linear accuracy of 30 μm. The activity controlling unit is an Apple IIe computer interfaced with the cell growth chamber via optical data links and is capable of simulating many of the mechanical activity patterns that cells are subjected to in vivo. Primary avian skeletal myoblasts proliferate and fuse into multinucleated myotubes in this set-up in a manner similar to normal tissue culture dishes. Under static culture conditions, the muscle cells differentiate into networks of myotubes which show little orientation. Growing the proliferating muscle cells on a unidirectional stretching substratum causes the developing myotubes to orient parallel to the direction of movement. In contrast, growing the cells on a substratum undergoing continuous stretch-relaxation cycling orients the developing myotubes perpendicular to the direction of movement. Neither type of mechanical activity significantly affects the rate of cell proliferation of the rate of myoblast fusion into myotubes. These results indicate that during in vivo skeletal muscle organogenesis, when substantial mechanical stresses are placed on skeletal muscle cells by both continuous bone elongation and by spontaneous contractions, only bone elongation plays a significant role in proper fiber orientation for subsequent functional work.
Similar content being viewed by others
References
Belloussou, L. V.; Dorfman, J. G.; Cherdantzev, V. G. Mechanical stresses and morphological patterns in amphibian embryos. J. Embryol. Exp. Morphol. 34:559–574; 1975.
Blechschmidt, E.; Gaser, R. F. Biokinetics and biodynamics of human differentiation. Principles and applications. IL: Charles C. Thomas Publisher; 1897:362.
Bragina, E. E.; Vasiliev, J. M.; Gelfand, I. M. Formation of bundles of microfilaments during spreading of fibroblasts on the substrate. Exp. Cell Res. 97:241–248; 1976.
Bray, D. Axonal growth in response to experimentally applied mechanical tension. Dev. Biol. 102:379–389; 1984.
Buck, R. C. Reorientation response of cells to repeated stretch and recoil of the substratum. Exp. Cell Res. 127:470–474; 1980.
Burton, A. D. Relation of structure to function of the tissues of the wall of blood vessels. Physiol. Rev. 34:619–667; 1954.
Caterson, B.; Lowther, D. A. Changes in the metabolism of the proteoglycan from sheep articular cartilage in response to mechanical stress. Biochim. Biophys. Acta 540:412–423; 1979.
Dartsch, P. C.; Hammerle, H.; Betz, H. Orientation of cultured arterial smooth muscle cells growing on cyclically stretched substrates. Acta Anat. 125:108–113; 1986.
Dartsch, P. C.; Hammerle, H. Orientation response of arterial smooth muscle cells to mechanical stimulation. Eur. J. Cell Biol. 41:339–346; 1986.
De Witt, M. T.; Handley, C. J.; Oates, B. W., et al.In vitro response of chondrocytes to mechanical loading. The effects of short term mechanical tension. Conn. Tissue Res. 12:97–109; 1984.
Ede, D. A.; Gumpel-Pinot, M.; Flint, O. P. Orientated movement of myogenic cells in the avian limb bud and its dependence on presence of the apical ectodermal ridge. In: Kemp, R. B.; Hinchcliffe, U. R., eds. Matrices and cell differentiation. New York: Alan R. Liss; 1984:427–438.
Folkman, J.; Moscona, A. Role of cell shape in growth control. Nature 273:345–349; 1978.
Franke, R. P.; Grafe, M.; Schnittler, H., et al. Induction of human vascular endothelial stress fibers by fluid shear stress. Nature 307:648–649; 1984.
Fry, D. L. Acute vascular endothelial changes associated with increased blood velocity. Circ. Res. 22:165–197; 1968.
Gabbiani, G.; Gabbiani, F.; Lomardi, D., et al. Organization of actin cytoskeleton in normal and regenerating arterial endothelial cells. Proc. Natl. Acad. Sci. USA 80:2361–2364; 1983.
Goss, R. J. Regulation of organ and tissue growth. New York: Academic Press; 1972:365.
Holmes, L. B.; Trelstad, R. L. Cell polarity in precartilage mouse limb mesenchyme cells. Dev. Biol. 78:511–520; 1980.
Ives, C. L.; Eskin, S. G.; McIntire, L. V. Mechanical effects on endothelial cell morphology: In vitro assessment. In Vitro 22:500–507; 1986.
Konigsberg, I. R. Diffusion-mediated control of myoblast fusion. Dev. Biol. 26:133–152; 1971.
Landmesser, L. T.; O'Donovan, M. J. Activation patterns of embryonic chick hind limb muscles recordedin ovo and in an isolated spinal cord preparation. J. Physiol. (Lond.) 347:189–204; 1984.
Leung, D. Y. M.; Glagov, S.; Mathews, M. B. A newin vitro system for studying cell response to mechanical stimulation. Different effects of cyclic stretching and agitation of smooth muscle cell biosynthesis. Exp. Cell Res. 109:285–298; 1977.
Luther, P. W.; Peng, H. B.; Lin, J. J. C. Changes in cell shape and actin distribution induced by constant electric fields. Nature 303:61–64; 1983.
Moss, P. S.; Spector, D. H.; Glass, C. A., et al. Streptomycin retards the phenotypic maturation of chick myogenic cells. In Vitro 20:473–478; 1984.
Pitelka, D. R.; Taggart, B. N. Mechanical tension induces lateral movement of intramembrane components of the tight junction: Studies on mouse mammary cells in culture. J. Cell. Biol. 96:606–612; 1983.
Powell, J. A. Development of normal and genetically dystrophic mouse muscle in tissue culture. Prefusion and fusion activities of mouse cells: phase contrast and time-lapse studies. Exp. Cell Res. 80:251–264; 1973.
Sheehan, D. C.; Hrapchak, B. B. Theory and practice of histochemistry. 2nd ed. St. Louis, MO: C. V. Mosby Co.; 1980:143.
Stewart, D. The role of tension in muscle growth. In: Regulation of organ and tissue growth. Goss, R. J. ed. New York: Academic Press; 1972:77–100.
Takenchi, S. Wound healing in the cornea of the chick embryo. IV. Promotion of the migratory activity of isolated corneal epithelium in culture by the application of tension. Dev. Biol. 70:232–240; 1979.
Vandenburgh, H. H. Separation of plasma membrane markers by glycerol-induced blistering of muscle cells. Biochim. Biophys. Acta 466:302–314; 1977.
Vandenburgh, H. H.; Kaufman, S.In vitro model for stretch-induced hypertrophy of skeletal muscle. Science 203:265–268; 1979
Vandenburgh, H. H. Dynamic mechanical orientation of skeletal myofibersin vitro. Dev. Biol. 93:438–443; 1982.
Vandenburgh, H. H. Motion into mass: How does tension stimulate muscle growth? Med. Sci. Sports Exerc. 19:S142-S149; 1987.
Vandenburgh, H. H.; Karlisch, P.; Farr, L. Maintenance of highly contractile tissue-cultured avian skeletal myotubes in collagen gel. In Vitro 24:166–174; 1988.
Weiss, P. Functional adaptation and the role of ground substances in development. Am. Naturalist 118:389–407; 1933.
Zak, R.; Rabinowitz, M. Molecular aspects of cardian hypertrophy. Ann. Rev. Physiol. 41:539–552; 1979.
Author information
Authors and Affiliations
Additional information
Supported by grants NS16753, AR36266, and RR05818 from the National Institutes of Health, Bethesda, MD.
Rights and permissions
About this article
Cite this article
Vandenburgh, H.H. A computerized mechanical cell stimulator for tissue culture: Effects on skeletal muscle organogenesis. In Vitro Cell Dev Biol 24, 609–619 (1988). https://doi.org/10.1007/BF02623597
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02623597