Abstract
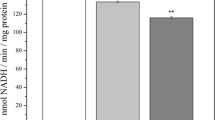
Oxidative damage on biological molecules has been proposed as a major cause of alterations observed in aging brain as well as in neurodegenerative diseases. In this study, we measured membrane fluidity in mitochondria extracted from three cerebral regions and cerebellum of Alzheimer disease (AD) patients and age-matched controls by means of fluorescence polarization technique. A significant reduction of mitochondrial membrane fluidity was found in AD, except in cerebellum. In controls, a decrease of membrane fluidity was observed along with age, and it was also related to the content of the oxidized nucleoside 8-hydroxy-2′-deoxyguanosine (OH8dG) in mitochondrial DNA (mtDNA). Alteration in membrane fluidity seems to be a result of lipid peroxidation, since it dramatically decreased when mitochondria were exposed to FeCl2 and H2O2. The parallel increase of viscosity in mitochondrial membrane and the amount of OH8dG in mtDNA is suggestive of a relationship between these biological markers of oxidative stress. These results provide further evidence that oxidative stress may play a role in the pathogenesis of AD.
Similar content being viewed by others
References
Ando S., Kon K., Aino K., and Totani Y. (1990) Increased levels of lipid peroxides in aged rat brain as revealed by direct assay of peroxide values.Neurosci. Lett. 133, 199–204.
Beal M. F., Mattson W. R., and Swartz K. J. (1990) Kynurenine pathway measurements in Huntington’s disease striatum: evidence for reduced formation of kynurenic acid.J. Neurochem. 55, 1327–1339.
Benzi G. and Moretti A. (1995) Are reactive oxygen species involved in Alzheimer’s disease?Neurobiol. Aging 16, 661–674.
Blass J. P. (1993) Pathophysiology of the Alzheimer syndrome.Neurology 43, 525–538.
Chen J. J. and Yu B. P. (1994) Alterations in mitochondrial membrane fluidity by lipid peroxidation products.Free Radical Biol. Med. 17, 411–418.
Dyrks T., Dyrks E., Hartmann T., Masters C., and Beyreuther K. (1992) Amyloidogenicity of βA4 and βA4-bearing amyloid protein precursor fragments by metal-catalyzed oxidation.J. Biol Chem. 267, 18,210–18,217.
Dyrks T., Dyrks E., Masters C. L., and Beyreuther K. (1993) Amyloidogenicity of rodent and human βA4 sequences.FEBS Lett. 324, 231–236.
Fraga C. G., Shigenaga M. K., and Park J-W. (1990) Oxidative damage to DNA during aging: 8-hydroxy-2′ deoxyguanosine in rat organ DNA and urine.Proc. Natl. Acad. Sci. USA 87, 4533–4537.
Gabuzda D., Busciglio J., Chen L. B., Matsudaira C., and Yankner B. A. (1994) Inhibition of energy metabolism alters the processing of amyloid precursor protein and induces a potentially amyloidogenic derivative.J. Biol. Chem. 6, 13,623–13,628.
Ghosh C., Dick R. M., and Ali S. F. (1993) Iron/ascorbate-induced lipid peroxidation changes membrane fluidity and muscarinic cholinergic receptor binding in rat frontal cortex.Neurochem. Int. 23, 479–484.
Hajimohammadreza I. and Brammer M. (1990) Brain membrane fluidity and lipid peroxidation in Alzheimer’s disease.Neurosci. Lett. 112, 333–337.
Hamm M. W., Winick M., and Schachter D. (1985) Macrophage phagocytosis and membrane fluidity in mice: the effect of age and dietary protein.Mech. Age Dev. 32, 11–20.
Hayakawa M., Torii K., and Sugiyama S. (1991) Age-associated accumulation of 8-hydroxy-2′-deoxyguanosine in mitochondrial DNA of human diaphragm.Biochem. Biophys. Res. Commun. 179, 1023–1029.
Hayakawa M., Hattori K., Sugiyama S., and Ozawa T. (1992) Age-associated oxygen damage and mutations in mitochondrial DNA of human hearts.Biochem. Biophys. Res. Commun. 189, 979–985.
Hruszkewycz A. M. (1992) Lipid peroxidation and mtDNA degeneration. A hypothesis.Mutat. Res. 275, 243–248.
Hruszkewycz A. M. and Bergtold D. S. (1990) The 8-hydroxyguanine content of isolated mitochondria increases with lipid peroxidation.Mutat. Res. 244, 123–128.
Irazu C. E., Rajagopalan P. R., Orak J. K., Fitts C. T., and Singh I. (1990) Mitochondrial membrane fluidity changes in renal ischemia.J. Exp. Pathol. 5, 1–6.
Kasai H. and Nashimura S. (1984) Hydroxylation of deoxyguanosine at the C-8 position by ascorbic acid and other reducing agents.Nucleic Acids Res. 12, 2137–2145.
Kish S. J., Bergeron C., Rajput A., Doric S., Mastrogiacomo F., Chang L. J., Wilson D. M., Di Stefano L. M., and Nobrega J. N. (1992) Brain cytochrome oxidase in Alzheimer’s disease.J. Neurochem. 59, 776–779.
Mecocci P., MacGarvey U., Kaufaman A. E., Koontz D., Shoffner J. M., Wallace D., and Beal M. F. (1993) Oxidative damage to mitochondrial DNA shows marked age-dependent increases in human brain.Ann. Neurol. 34, 609–616.
Mecocci P., MacGarvey U., and Beal M. F. (1994) Oxidative damage to mitochondrial DNA is increased in Alzheimer’s disease.Ann. Neurol. 36, 747–751.
Mehrotra S., Kakkar P., and Viswanathan P. N. (1991) Mitochondrial damage by active oxygen species in vitro.Free Radical Biol. Med. 10, 277–285.
Miquel J. (1991) An integrated theory as the result of mitochondrial DNA mutation in differentiated cells.Arch. Gerontol. Geriatr. 12, 99–117.
Mutisya E. M., Bowling A. C., and Beal M. F. (1994) Cortical cytochrome activity is reduced in Alzheimer’s disease.J. Neurochem. 63, 2179–2184.
Nohl H. (1986) Oxygen radical release in mitochondria: influence of age, inFree Radicals, Aging and Degenerative Diseases. (Johnson J. E., Jr. Walford R., Harman D., and Miquel J., eds.), Alan R. Liss, New York, pp. 77–97.
Nohl H. and Hegner D. (1978) Do mitochondria produce oxygen radicals in vivo?Eur. J. Biochem. 82, 563–567.
Orr W. C. and Sohal R. S. (1994) Extension of life-span by overexpression of superoxide dismutase and catalase inDrosophila melanogaster.Science 263, 1128–1130.
Palmer A. M. and Burns M. A. (1994) Selective increase in lipid peroxidation in the inferior temporal cortex in Alzheimer diseases.Brain Res. 645, 338–342.
Paker W. D., Jr. Filley C. M., and Parks J. K. (1990) Cytochrome oxidase deficiency in Alzheimer’s disease.Neurology 40, 1302, 1303.
Parker Jr. W. D., Mahr N. J., Filley C. M., Parks J. K., Hughes D., Young D. A., and Cullum C. A. (1994a) Reduced platelet cytochrome C oxidase activity in Alzheimer’s disease.Neurology 44, 1086–1090.
Parker W. D., Jr., Parks J., Filley C. M., and Kleinschmidt-De Masters B. K. (1994b) Electron transport chain defects in Alzheimer disease brain.Neurology 44, 1090–1996.
Partridge R. S., Monroe S. M., Parks J. K., Johnson K., Parker W. D., Eaton G. R., and Eaton S. S. (1994) Spin trapping of azidyl and hydroxyl radicals in azide-inhibited rat brain submitochondrial particles.Arch. Biochem. Biophys. 310, 210–217.
Pryor W. A. (1986) Oxy-radicals and related species: their formation, lifetimes and reactions.Annu. Rev. Physiol. 48, 657–667.
Richter C., Park L-W., and Ames B. N. (1988) Normal oxidative damage to mitochondrial and nuclear DNA is extensive.Proc. Natl. Acad. Sci. USA 85, 6465–6467.
Saido T. C., Yokota M., Maruyama K., Yamao-Harigaya W., Tani E., Ihara Y., and Kawashima S. (1994) Spatial resolution of the primary β-amyloidogenic process induced in postischemic hippocampus.J. Biol. Chem. 269, 15,253–15,257.
Saraiva A. A., Borges M. M., Madeira M. D., Tavares M. A., and Paula-Barbosa M. M. (1985) Mitochondrial abnormalities in cortical dendrites from patients with Alzheimer’s disease.J. Submicrosc. Cytol. 17, 459–464.
Shinitzky M. and Barenholz Y. (1978) Fluidity parameters of lipid regions determined by fluorescence polarization.Biochim. Biophys. Acta 515, 367–394.
Smith C. D., Carney J. M., Starke-Reed P. E., Oliver C. N., Stadtman E. R., Floyd R. A., and Markesbery W. R. (1991) Excess brain protein oxidation and enzyme dysfunction in normal aging and Alzheimer’s disease.Proc. Natl. Acad. Sci. USA 88, 10,540–10,543.
Sohal R. S. (1993) Aging, cytochrome oxidase activity and hydrogen peroxide release by mitochondria.Free Radical Biol. Med. 14, 583–588.
Sohal R. S., Arnold L. A., and Sohal B. H. (1990) Age-related changes in antioxidant enzymes and prooxidant generation in tissues of the rat with special reference to parameters in two insect species.Free Radical Biol. Med. 10, 495–500.
Sohal R. S., Ku H-H., Agarwal S., Forster M. J., and Lal H. (1994) Oxidative damage, mitochondrial oxidant generation and antioxidant defenses during aging and in response to food restriction in the mouse.Mech. Age Dev. 74, 121–133.
Subbarao K. V., Richardson J. S., and Ang L. C. (1990) Autopsy samples of Alzheimer’s cortex show increased peroxidation in vitro.J. Neurochem. 55, 342–345.
Tappel A. L. and Zalkin H. (1959) Lipid peroxidation in isolated mitochondria.Arch. Biochem. Biophys. 80, 326–332.
Troncoso J. C., Costello A., Watson A. L., Jr., and Johnson G. V. W. (1993) In vitro polymerization of oxidazed tau into filaments.Brain Res. 613, 313–316.
Wahnon R., Mokady S., and Cogan U. (1989) Age and membrane fluidity.Mech. Age Dev. 50, 249–255.
Wells K., Farooqui A. A., Liss L., and Horrocks L. A. (1995) Neural membrane phospholipids in Alzheimer disease.Neurochem. Res. 20, 1329–1333.
Yu B. P. (1993) Oxidative damage by free radicals and lipid peroxidation in aging, inFree Radicals in Aging (Yu B. P., ed.), CRC, Boca Raton, FL, pp. 57–88.
Yu B. P., Suescon E. A., and Yang S. Y. (1992) Effect of age-related lipid peroxidation on membrane fluidity and phospholipase A2: modulation by dietary restriction.Mech. Age Dev. 65, 17–33.
Zhang J-R., Andrus P. K., and Hall E. D. (1991) Age-related phospholipid hydroperoxide levels in gerbil brain measured by HPLC-chemiluminescence and their relation to hydroxyl radical stress.Brain Res. 639, 275–282.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mecocci, P., Beal, M.F., Cecchetti, R. et al. Mitochondrial membrane fluidity and oxidative damage to mitochondrial DNA in aged and AD human brain. Molecular and Chemical Neuropathology 31, 53–64 (1997). https://doi.org/10.1007/BF02815160
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02815160