Summary
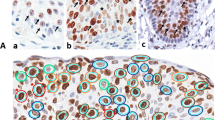
The cell cycle-dependent distribution of the proliferation-associated Ki-67 antigen has been evaluated immunocytochemically in L-132 human fetal lung cells. The cells were synchronized and cell cycle phases were determined: G1 = 6.7 h, S =5.4 h, G2 = 8.5 h and mitosis = 1.3 h. The Ki-67 patterns were strictly correlated with the cell cycle phases. In late G1-phase, Ki-67 antigen was present only in the perinucleolar region. In the S-phase, Ki-67 staining was found homogeneously in the karyoplasm and in the perinucleolar region. G2-phase cells contained a finely granular Ki-67 staining in the karyoplasm with Ki-67-positive specks and perinucleolar staining. In early mitotic cells (pro-and metaphase) an intense perichromosomal Ki-67 staining was observed in addition to a homogeneously stained karyoplasm in prophase, and cytoplasm in metaphase. During ana-and telophase the Ki-67 antigen disappeared rapidly. In resting cells there was no Ki-67 staining.
Similar content being viewed by others
References
Beckstead JH (1985) Optimal antigen localization in human tissues using aldehyde fixated plasitc-embedded sections. J Histochem Cytochem 33:954–958
Bouteille M, Bouvier D, Seve AP (1983) Heterogeneity and teritorial organization of the nuclear matrix and related structures. Int Rev Cytol 83:135–182
Bradbury EM, Matthews HR (1982) Chromatin structure, histone modifications and the cell cycle. In: Nicolini C (ed) Cell growth: NATO Advanced Study Institutes Series A. Life Sci, vol 38. Plenum Press, New York, pp 411–454
Bravo R, Frank R, Blundell PA, McDonald-Bravo H (1987) Cyclin/PCNA is the auxiliary protein of DNA polymerase-δ. Nature 326:515–517
Burger PC, Shibata T, Kleihues P (1986) The use of the monoclonal antibody Ki-67 in the identification of proliferating cells: application to surgical neuropathology. Am J Surg Pathol 10:611–617
Busch H, Smetana K (1970) The nucleolus, vol 1. Academic Press, London
Chaly N, Bladon T, Setterfield G, Little JE, Kaplan FG, Brown DL (1984) Changes in distribution of nuclear matrix antigens during mitotic cell cycle. J Cell Biol 99:661–671
Clevenger CV, Epstein AL (1984) Identification of a nuclear protein component of interchromatin granules using a monoclonal antibody and immunogold electron microscopy. Exp Cell Res 151:194–207
Cooper S (1982) The continuum model: application to G1-arrest and G0. In: Nicolini C (ed) Cell growth: NATO advanced study institutes series A, Life sci, vol 38, Plenum Press, New York, pp 315–336
Daskal Y, Smetana K, Busch H (1980) Evidence from studies on segregated nucleoli that nucleolar silver staining proteins C23 and B23 are in the fibrillar component. Exp Cell Res 127:285–291
Davis FM, Wegner R-D, Rao PN (1987) Monoclonal antibody with specificity to mitotic chromosomes of primates. Exp Cell Res 170:417–427
Epifanova OI, Abuladze MK, Zosimovskaya AI (1975) Effects of low concentrations of actinomycin D on the initiation of DNA synthesis in rapidly proliferating and stimulated cell cultures. Exp Cell Res 92:23–30
Escande M-L, Gas N, Stevens BJ (1985) Immunolocalization of the 100 k nucleolar protein in CHO cells. Biol Cell 53:99–110
Franke WW, Schmid E, Wellsteed J, Grund C, Gigi O, Geiger B (1983) Change of cytokeratin filament organization during the cell cycle: Selective masking of an immunologie determinant in interphase PtK2 cells. J Cell Biol 97:1255–1260
Franklin WA, Bibbo M, Doria MI, Dytch HE, Toth J, De-Sombre E, Wied GL (1987) Quantitation of estrogen receptor content and Ki-67 staining in breast carcinoma by microticas image analysis system. Anal Quant Cytol Histol 9:279–285
Gallee MP, van Steenbrugge GJ, ten Kate FJ, Schroeder FH, van der Kwast TH (1987) Determination of the proliferative fraction of a transplantable, hormone-dependent, human prostatic carcinoma (PC-82) by monoclonal antibody Ki-67: potential application for hormone therapy monitoring. J Natl Cancer Inst 79:1333–1340
Gerdes J, Lemke H, Baische H, Wacker HH, Schwab U (1984) Cell cycle analysis of a cell proliferation associated human nuclear antigen defined by the monoclonal antibody Ki-67. J Immunol 133:1710–1715
Gerdes J, Schwab U, Lemke H, Stein H (1983) Production of a mouse monoclonal antibody reactive with a human antigen associated with cell proliferation. Int J Cancer 31:13–20
Gerdes J, Stein H, Pileri S, Rivano MT, Gobbi M, Ralfkiaer E, Nielsen KM, Pallesen G, Bartels H, Palestro G, Delsol G (1987) Prognostic relevance of tumor-cell growth fraction in malignant non-Hodgkin’s lymphomas. Lancet 2:448–449
Giangaspero F, Doglioni C, Rivano MT, Pileri S, Gerdes J, Stein H (1987) Growth fraction in human brain tumors defined by the monoclonal antibody Ki-67. Acta Neurop 74:179–182
Hancock R, Boulikas T (1982) Functional organization in the nucleus. Int Rev Cytol 79:165–214
Hügle B, Hazan R, Scheer U, Franke WW (1985) Localization of ribosomal protein S1 in the granular component of the interphase nucleolus and its distribution during mitosis. J Cell Biol 100:873–886
Kaul S, Pfeifer GP, Drahovsky D (1984) Preparation of monoclonal antibodies against DNA-cytosine-5′-methyltransferase from human-placenta. Eur J Cell Biol 34:330–335
Koepsel RR, Khan SA (1986) Static and initiator-enhanced bending of DNA at a replication origin. Science 233:1316–1318
Koepsel RR, Murray RW, Khan SA (1986) Sequence-specific interaction between the replication initiator protein of the plasmid pT181 and its origin of the replication. Proc Natl Acad Sci USA 83:5484–5488
Lalor PA, Mapp PI, Hall PA, Revell PA (1987) Proliferative activity of cells in the synovium as demonstrated by a monoclonal antibody, Ki-67. Rheumatol Int 7:183–186
Lelle RJ, Heidenreich W, Stauch G, Wecke I, Gerdes J (1987) Determination of growth fractions in benign breast disease (BBD) with monoclonal antibody Ki-67. J Cancer Res Clin Oncol 113:73–77
Lloyd R, Wilson BS, Vrani J, Gaur PK, Moline S, Makari J (1985) Immunocytochemical characterization of a monoclonal antibody that recognizes mitosing cells. Am J Pathol 121:275–283
Matsuhashi S, Watanabe T, Hori K (1987) An antigen expressed in proliferating cells at late G1-S phase. Exp Cell Res 170:351–362
McGurrin JF, Doria MI Jr, Dawson PJ, Karrison T, Stein HO, Franklin WA (1987) Assessment of tumor cell kinetics by immunohistochemistry in carcinoma of breast. Cancer 59:1744–1750
McKeon FD, Tuffanelli DL, Kobayashi S, Kirschner MW (1984) The redistribution of a conserved nuclear envelope protein during the cell cycle suggests a pathway for chromosome condensation. Cell 36:83–92
Olson MOJ, Guetzow K, Busch H (1981) Localization of phosphoprotein C23 in nucleoli by immunological methods. Exp Cell Res 135:259–265
Palutke M, Kakuruga D, Tabaczka P (1987) A flow cytometric method for measuring lymphocyte proliferation directly from tissue culture plates using Ki-67 and propidium iodide. J Immunol Methods 105:97–105
Pastolero GC, Mori S (1987) Expression of proliferation-associated nuclear antigen in human malignancies. Jpn J Exp Med 57:193–198
Pileri S, Gerdes J, Rivano M, Tazzari PL, Magnani M, Gobbi M, Stein H (1987) Immunohistochemical determination of growth-fractions in human permanent cell lines and lymphoid tumors: a critical comparision of monoclonal antibodies OKT9 and Ki-67. Br J Haematol 65:271–276
Prelich G, Kostura M, Marhak DR, Mathews MB, Stillman B (1987a) The cell cycle regulated proliferating cell nuclear antigen is required for SV40 DNA replication in vitro. Nature 326:471–475
Prelich G, Tan CK, Kostura M, Mathews MB, So AG, Downey KM, Stillman B (1987b) Functional identity of proliferating cell nuclear antigen and a DNA polymerase delta auxiliary protein. Nature 326:517–520
Puck TT (1964) Studies of the life cycle of mammalian cells. Cold Spring Harbor 29:167–176
Puck TT, Steffen J (1963) Life cycle analysis of mammalian cells. Biophysic J 3:379–397
Ralfkiaer E, Stein H, Bosq J, Gatter KC, Ralfkiaer N, Wantzin GL, Mason DY (1986) Expression of a cell-cycle-associated nuclear antigen (Ki-67) in cutaneous lymphoid infiltrates. Am J Dermatopathol 8:37–43
Rao P, Engelberg J (1966) Effects of temperature on the mitotic cycle of normal and synchronized mammalian cells. In: Cameron IL, Padilla GM (eds) Cell synchrony, vol 1. Academic Press, New York, pp 332–351
Roggendorf W, Schuster T, Pfeifer J (1987) Proliferative potential of menigiomas determined with the monoclonal antibody Ki-67. Acta Neuropathol 73:361–364
Sasaki K, Murakami T, Kawasaki M, Takahashi M (1987) The cell cycle associated change of the Ki-67 reactive nuclear antigen expression. J Cell Physiol 133:579–584
Schmidt-Zachmann MS, Hügle B, Scheer U, Franke WW (1984) Identification and localization of a novel nucleolar protein of high molecular weight by a monoclonal antibody. Exp Cell Res 153:327–346
Schwarting R, Gerdes J, Niehus J, Jaeschke J, Stein H (1986) Determination of growth fraction in cell suspensions by flow cytometry using the monoclonal antibody Ki-67. J Immunol Methods 90:65–70
Stein H, Gerdes J, Schwab U, Lemke H, Mason DY, Ziegler A, Schienle W, Diehl V (1982) Identification of Hodgkin and Sternberg-Reed cells as a unique cell type derived from a newly-detected small-cell poulation. Int J Cancer 30:445–459
Taylor EW (1965) Control of DNA synthesis in mammalian cells in culture. Exp Cell Res 40:316–332
Terasima T, Tolmach LJ (1963) Growth and nucleic acid synthesis in synchronously dividing populations of HeLa cells. Exp Cell Res 30:347–362
Turner BM, Franchi L (1987) Identification of protein antigens associated with the nuclear matrix and with clusters of interchromatin granules in both interphase and mitotic cells. J Cell Sci 87:269–282
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Braun, N., Papadopoulos, T. & Müller-Hermelink, H.K. Cell cycle dependent distribution of the proliferation-associated Ki-67 antigen in human embryonic lung cells. Virchows Archiv B Cell Pathol 56, 25–33 (1988). https://doi.org/10.1007/BF02889998
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02889998