Abstract
Purpose
There are an increasing number of anecdotal reports and trials of recombinant activated factor VII (rFVIIa) for bleeding during surgery. The reports of rFVIIa during cardiac surgery are limited. We report our experience using rFVIIa, in the operating room; to treat bleeding that prevented chest closure, despite appropriate conventional treatment, following complex cardiac surgery.
Methods
Retrospective chart review, at an Australian University hospital and associated private hospital, of cardiac surgery patients given rFVIIa (usual dose 90 μg·kg-1). We used rFVIIa for bleeding that prevented closure of the chest despite administration of blood products, protamine, and surgical attempts to secure hemostasis.
Results
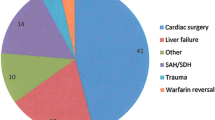
Recombinant activated factor VII was administered on 55 occasions to 53 patients. Most patients had complex aortic or valve surgery. Median bypass time was 266 min. Before administering rFVIIa, patients received (median): packed red cells four units; platelets 15 units; fresh frozen plasma eight units; and cryoprecipitate ten units. After administering rFVIIa the median doses of donor blood products up to 12 hr after intensive care unit admission were: packed red cells one unit; platelets zero units; fresh frozen plasma zero units; and cryoprecipitate zero units. The decrease in doses of all blood products was significant (P < 0.001). We could not determine if rFVIIa played a role in significant mortality (19%) and morbidity (17%).
Conclusion
Use of rFVIIa in cardiac surgery may be effective, but definitive clinical trials are needed to clarify its role in clinical practice and safety. We present an rFVIIa guideline developed during the audit period.
Résumé
Objectif
Un nombre croissant d’articles et d’essais isolés traitent du facteur VII recombinant activé (rFVIIa) utilisé contre les saignements chirurgicaux. Peu d’articles mentionnent le rFVIIa pendant la chirurgie cardiaque. Nous présentons son utilisation en salle d’opération pour contrer les saignements qui entravent la fermeture du thorax malgré le traitement approprié habituel suivant une opération cardiaque complexe.
Méthode
Une revue rétrospective a été faite des dossiers des patients de chirurgie cardiaque, d’un hôpital universitaire australien et d’un hôpital privé associé, qui ont reçu une dose habituelle de 90 μg·kg-1 de rFVIIa. L’usage du rFVIIa visait à contrer les saignements qui surviennent malgré les produits sanguins et la protamine administrés et des tentatives chirurgicales pour assurer l’hémostase.
Résultats
Le rFVIIa a été administré à 55 reprises chez 53 patients qui ont subi, en majorité, une opération aortique ou valvulaire complexe avec une CEC d’une durée moyenne de 266 min. Les patients ont d’abord reçu en moyenne: quatre unités de culots globulaires, 15 unités de plaquettes, huit unités de plasma frais congelé et dix unités de cryoprécipité. Après le rFVIIa, la moyennes des produits sanguins administrés jusqu’à 12 h après l’arrivée à l’unité des soins intensifs a été de: une unité de culot globulaire, aucune unité de plaquettes, de plasma frais congelé et de cryoprécipité, donc une diminution significative (P < 0,001). Un rôle possible du rFVIIa dans les taux significatifs de mortalité (19%) et de morbidité (17%) n’a pu être déterminé.
Conclusion
Le rFVIIa en chirurgie cardiaque peut être efficace, mais des études cliniques approfondies devront clarifier son rôle dans une pratique clinique sûre. Nous présentons une ligne de conduite concernant le rFVIIa, élaborée pendant la période d’évaluation.
Article PDF
Similar content being viewed by others
References
Franchini M, Zaffanello M, Veneri D. Recombinant factor VIIa. An update on its clinical use. Thromb Haemost 2005; 93: 1027–35.
Levi M, Peters M, Buller HR. Efficacy and safety of recombinant factor VIIa for treatment of severe bleeding: a systematic review. Crit Care Med 2005; 33: 883–90.
Raivio P, Suojaranta-Ylinen R, Kuitunen AH. Recombinant factor VIIa in the treatment of postoperative hemorrhage after cardiac surgery. Ann Thorac Surg 2005; 80: 66–71.
Hyllner M, Houltz E, Jeppsson A. Recombinant activated factor VII in the management of life-threatening bleeding in cardiac surgery. Eur J Cardiothorac Surg 2005; 28: 254–8.
Karkouti K, Beattie WS, Wijeysundera DN, et al. Recombinant factor VIIa for intractable blood loss after cardiac surgery: a propensity score-matched case-control analysis. Transfusion 2005; 45: 26–34.
Aggarwal A, Malkovska V, Catlett JP, Alcorn K. Recombinant activated factor VII (rFVIIa) as salvage treatment for intractable hemorrhage. Thromb J 2004; 2: 9.
von Heymann C, Redlich U, Jain U, et al. Recombinant activated factor VII for refractory bleeding after cardiac surgery-a retrospective analysis of safety and efficacy. Crit Care Med 2005; 33: 2241–6.
Diprose P, Herbertson MJ, O’Shaughnessy D, Gill RS. Activated recombinant factor VII after cardiopulmonary bypass reduces allogeneic transfusion in complex non-coronary cardiac surgery: randomized doubleblind placebo-controlled pilot study. Br J Anaesth 2005; 95: 596–602.
von Heymann C, Hotz H, Konertz W, Kox WJ, Spies C. Successful treatment of refractory bleeding with recombinant factor VIIa after redo coronary artery bypass graft surgery. J Cardiothorac Vasc Anesth 2002; 16: 615–6.
Despotis G, Avidan M, Lublin DM. Off-label use of recombinant factor VIIA concentrates after cardiac surgery. Ann Thorac Surg 2005; 80: 3–5.
Spiess BD. Transfusion of blood products affects outcome in cardiac surgery. Semin Cardiothorac Vasc Anesth 2004; 8: 267–81.
Karkouti K, Wijeysundera DN, Yau TM, et al. The independent association of massive blood loss with mortality in cardiac surgery. Transfusion 2004; 44: 1453–62.
Martinowitz U, Michaelson M; The Israeli Multidisciplinary rFVIIa Task Force. Guidelines for the use of recombinant activated factor VII (rFVIIa) in uncontrolled bleeding: a report by the Israeli Multidisciplinary rFVIIa Task Force. J Thromb Haemost 2005; 3: 640–8.
National Health and Medical Research Council. Clinical Practice Guidelines on the Use of Blood Components (red blood cells, platelets, fresh frozen plasma, cryoprecipitate). Canberra: Commonwealth of Australia, 2002. Available from URL; www.nhmrc.gov. au.
Bevan DH. Cardiac bypass haemostasis: putting blood through the mill. Br J Haematol 1999; 104: 208–19.
Blome M, Isgro F, Kiessling AH, et al. Relationship between factor XIII activity, fibrinogen, haemostasis screening tests and postoperative bleeding in cardiopulmonary bypass surgery. Thromb Haemost 2005; 93: 1101–7.
Paparella D, Brister SJ, Buchanan MR. Coagulation disorders of cardiopulmonary bypass: a review. Intensive Care Med 2004; 30: 1873–81.
French CJ, Bellomo R, Angus P. Cryoprecipitate for the correction of coagulopathy associated with liver disease. Anaesth Intensive Care 2003; 31: 357–61.
British Committee for Standards in Haematology, Blood Transfusion Task Force. Guidelines for the use of platelet transfusions. Br J Haematol 2003; 122: 10–23.
O’Shaughnessy DF, Atterbury C, Bolton Maggs P, et al.; British Committee for Standards in Haematology, Blood Transfusion Task Force. Guidelines for the use of freshfrozen plasma, cryoprecipitate and cryosupernatant. Br J Haematol 2004; 126: 11–28.
Thompson AR. When all else fails to stop massive bleeding from trauma. J Thromb Haemost 2005; 3: 638–9.
Engoren MC, Habib RH, Zacharias A, Schwann TA, Riordan CJ, Durham SJ. Effect of blood transfusion on long-term survival after cardiac operation. Ann Thorac Surg 2002; 74: 1180–6.
Clark AD, Gordon WC, Walker ID, Tait RC. ’Lastditch’ use of recombinant factor VIIa in patients with massive haemorrhage is ineffective. Vox Sang 2004; 86: 120–4.
Hardy JF. Managing uncontrolled hemorrhage in trauma and surgery: a novel and promising approach. Can J Anesth 2002; 49(Suppl 2): S4–6.
Hall TS, Brevetti GR, Skoultchi AJ, Sines JC, Gregory P, Spotnitz AJ. Re-exploration for hemorrhage following open heart surgery differentiation on the causes of bleeding and the impact on patient outcomes. Ann Thorac Cardiovasc Surg 2001; 7: 352–7.
Boffard KD, Riou B, Warren B, et al.; NovoSeven Trauma Study Group. Recombinant factor VIIa as adjunctive therapy for bleeding control in severely injured trauma patients: two parallel randomized, placebo-controlled, double-blind clinical trials. J Trauma 2005; 59: 8–15; discussion 15-8.
Enomoto TM, Thorborg P. Emerging off-label uses for recombinant activated factor VII: grading the evidence. Crit Care Clin 2005; 21: 611–32.
de Jong E, Friederich PW, Vlasuk GP, et al. Activation of coagulation by administration of recombinant factor VIIa elicits interleukin 6 (IL-6) and IL-8 release in healthy human subjects. Clin Diag Lab Immunol 2003; 10: 495–7.
Spahn DR, Tucci MA, Makris M. Is recombinant FVIIa the magic bullet in the treatment of major bleeding? Br J Anaesth 2005; 94: 553–5.
Goodnough LT, Lublin DM, Zhang L, Despotis G, Eby C. Transfusion medicine service policies for recombinant factor VIIa administration. Transfusion 2004; 44: 1325–31.
Author information
Authors and Affiliations
Corresponding author
Additional information
Funding and conflict-of-interest statement: The Austin Department of Anaesthesia Research Fund funded this audit. The Austin Department of Anaesthesia Fund has received a non-conditional educational grant from Novo Nordisk. Peter McCall is a member of a Cardiac Surgery Advisory Committee for Novo Nordisk and has presented at company sponsored educational seminars. David Story and Darshi Karapillai have no other potential conflicts-of-interest with this study.
An erratum to this article is available at http://dx.doi.org/10.1007/BF03021596.
Rights and permissions
About this article
Cite this article
McCall, P., Story, D.A. & Karapillai, D. Audit of factor VIIa for bleeding resistant to conventional therapy following complex cardiac surgery. Can J Anesth 53, 926–933 (2006). https://doi.org/10.1007/BF03022836
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03022836