Summary
Intake of the anticonvulsant drug valproic acid, or its sodium salt, has been associated with occasional instances of severe and sometimes fatal hepatotoxicity. Probably at least 80 cases have occurred worldwide. The syndrome affects perhaps 1 in 10,000 persons taking the drug, and usually develops in the early weeks or months of therapy. Most instances have involved children, usually those receiving more than 1 anticonvulsant. Multiple cases have occurred in 2 families. The typical presentation is of worsening epilepsy, increasing depression of consciousness, and progressive clinical and biochemical evidence of liver failure. The liver has sometimes shown hepatocyte necrosis, and on other occasions widespread microvesicular steatosis, while cholestatic changes have also occurred. The appearances are interpreted as consistent with a drug toxicity reaction.
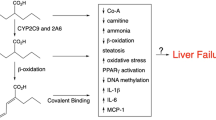
During the hepatotoxicity increased amounts of unsaturated metabolites of valproate, notably 4-en-valproate, have been found in blood and urine. In 4 cases there has been evidence of impaired β-oxidation of valproate with, in 1 case, accumulation of isomers of valproate glucuronide caused by intramolecular rearrangement of the conjugate. There are molecular structural similarities between 4-en-valproate and 2 known hepatotoxins (4-en-pentanoate and methylenecyclopropylacetic acid, the latter being responsible for hypoglycin poisoning). There are also clinical and histopathological similarities between valproate hepatotoxicity and both hypoglycin poisoning and certain spontaneous disorders of isoleucine metabolism (one pathway ofvalproate metabolism is analogous to oxidative degradation of isoleucine). Unsaturated metabolites of valproate, in particular 4-en-valproate, may contribute to the hepatotoxicity of the drug. However, since the hepatotoxicity appears to involve an element of idiosyncrasy, the primary defect in some cases may be an inherited or acquired deficiency in the drug’s β-oxidation. This defect may divert valproate metabolism towards ω-oxidation, with increased formation of the toxin 4-en-valproate, but may also allow increased formation of a toxic metabolite derived from isoleucine, since β-oxidation of isoleucine derivatives will also be impaired.
Similar content being viewed by others
References
Abbott FS, Kassam J, Orr JM, Farrell K. The effect of aspirin on valproic acid metabolism. Clinical Pharmacology and Therapeutics 40: 94–110, 1986
Acheampong A, Abbott FS. Synthesis and stereochemical determination of diunsaturated valproic acid analogs including its major diunsaturated metabolite. Journal of Lipid Research 26: 1002–1008, 1985
Allen RJ, Coulter DL. Valproic acid induced pancreatitis in children. Pediatrics 65: 1194–1195, 1980
Batalden PB, Van Dyne BJ, Lloyd J. Pancreatitis associated with valproic acid therapy. Pediatrics 64: 520–522, 1979
Becker C-M, Harris RA. Influence of valproic acid on hepatic carbohydrate and lipid metabolism. Archives of Biochemistry and Biophysics 223: 381–392, 1983
Benavides J, Martin A, Ugarte M, Valdivieso F. Inhibition by valproic acid of pyruvate uptake by brain mitochondria. Biochemical Pharmacology 31: 1633–1636, 1982
Bjorge SM, Baillie TA. Inhibition of medium-chain fatty acid β-oxidation in vitroby valproic acid and its unsaturated metabolite, 2-n-propyl-4-pentenoic acid. Biochemical and Biophysical Research Communications 132: 245–252, 1985
Böhles H, Richter K, Wagner-Thiessen E, Schäfer H. Decreased serum carnitine in valproate-induced Reye syndrome. European Journal of Paediatrics 139: 185–186, 1982
Camfield PR, Bagnell P, Camfield CS, Tibbies JAR. Pancreatitis due to valproic acid. Lancet 1: 1198–1199, 1979
Chapman A, Keane PE, Meldrum BS, Simiand J, Vernieres JC. Mechanism of anticonvulsant action of valproate. Progress in Neurobiology 19: 315–359, 1982
Coude FX, Grimber G, Pelet A, Benoit Y. Action of the anti-epileptic drug, valproic acid, on fatty acid oxidation in isolated rat hepatocytes. Biochemical and Biophysical Research Communications 115: 730–736, 1983
Coulter DL. Carnitine deficiency: a possible mechanism for valproate hepatotoxicity. Lancet 1: 689, 1984
Coulter DL, Allen RJ. Secondary hyperammonaemia: a possible mechanism for valproate encephalopathy. Lancet 1: 1310–1311, 1980
Coulter DL, Allen RJ. Hyperammonemia with valproic acid therapy. Journal of Pediatrics 99: 317–319, 1981
Dickinson RG, Bassett ML, Searle J, Tyrer JH, Eadie MJ. Valproate hepatotoxicity: a review and report of two instances in adults. Clinical and Experimental Neurology 21: 79–91, 1985a
Dickinson RG, Harland RC, Ilias AM, Rodgers RM, et al. Disposition of valproic acid in the rat: dose-dependent metabolism, distribution, enterohepatic recirculation and choleretic effect. Journal of Pharmacology and Experimental Therapeutics 211: 583–595, 1979
Dickinson RG, Hooper WD, Eadie MJ. pH-Dependent rearrangement of the biosynthetic ester glucuronide of yalproic acid to β-glucuronidase-resistant forms. Drug Metabolism and Disposition 12: 247–252, 1984
Dickinson RG, Kluck RM, Hooper WD, Patterson M, Chalk JB, et al. Rearrangement of valproate glucuronide in a patient with drug-associated hepatobiliary and renal dysfunction. Epilepsia 26: 589–593, 1985b
Dreifuss FE, Santilli N, Langer DH, Sweeney KP, Moline KA, et al. Valproic acid hepatic fatalities: a retrospective review. Neurology 37: 379–385, 1987
Ferrandes B, Eymard P. Metabolism of valproate sodium in rabbit, rat, dog and man. Epilepsia 18: 169–182, 1977
Gastaut H, Noel P. A case of fatal toxic hepatitis: recommendations for the administration of sodium valproate. Epilepsia 22: 711–713, 1981
Gerber N, Dickinson RG, Harland RC, Lynn RK, Houghton D, et al. Reye like syndrome associated with valproic acid therapy. Journal of Pediatrics 95: 142–144, 1979
Glasgow AM, Chase HP. Production of the features of Reye’s syndrome in rats with 4-pentenoic acid. Pediatric Research 9: 133–138, 1975
Gompertz D, Tippett P, Bartlett K, Baillie T. Identification of urinary metabolites of sodium dipropylacetate in man: potential sources of interference in organic acid screening procedures. Clinica Chimica Acta 74: 153–160, 1977
Gram L. Hepatic toxicity of valproate: reflections on the pathogenesis and proposal for an international collaborative registration. In Oxley et al. (Eds) Chronic toxicity of antiepileptic drugs, pp. 69–78, Raven Press, New York, 1983
Granneman GR, Marriott TB, Wang SI, Sennello LT, Hagen NS, et al. Aspects of the dose-dependent metabolism of valproic acid. In Levy et al. (Eds) Metabolism of antiepileptic drugs, pp. 97–104, Raven Press, New York, 1984a
Granneman GR, Wang S-I, Kesterson JW, Machinist JM. The hepatotoxicity of valproic acid and its metabolites in rats, II: intermediary and valproic acid metabolism. Hepatology 4: 1153–1158, 1984b
Granneman GR, Wang SI, Machinist JM, Kesterson JW. Aspects of the metabolism of valproic acid. Xenobiotica 14: 375–387, 1984c
Gugler R, von Unruh GE. Clinical pharmacokinetics of valproic acid. Clinical Pharmacokinetics 5: 67–83, 1980
Haas R, Stumpf DA, Bergen BJ, Parks JK, Eguren L. Inhibition of oxidative phosphorylation by sodium valproate. Neurology 30: 420, 1980
Haas R, Stumpf DA, Parks JK, Eguren L. Inhibitory effects of sodium valproate on oxidative phosphorylation. Neurology 31: 1473–1476, 1981
Haidukewych D, John G. Chronic valproic acid and coepileptic drug therapy and incidence of increases in serum liver enzymes. Therapeutic Drug Monitoring 8: 407–410, 1986
Hayasaka K, Takahashi I, Kobayashi Y, Iinuma K, Narisawa K, et al. Effects of valproate on biogenesis and function of liver mitochondria. Neurology 36: 351–356, 1986
Heinemeyer G, Nau H, Hildebrand AG, Roots I. Oxidation and glucuronidation of valproic acid in male rats — influence of phenobarbital, 3-methylcholanthrene, β-naphthoflavone and clofibrate. Biochemical Pharmacology 34: 133–139, 1985
Hjelm M, De Silva LVK, Seakins JWT, Oberholzer VG, Rolles CJ. Evidence of inherited urea cycle defect in a case of fatal valproate toxicity. British Medical Journal 292: 23–24, 1986a
Hjelm M, Oberholzer V, Seakins J, Thomas S, Kay JDS. Valproate-induced inhibition of urea synthesis and hyperammonaemia in healthy subjects. Lancet 2: 859, 1986b
Horie S, Suga T. Enhancement of peroxisomal β-oxidation in the liver of rats and mice treated with valproic acid. Biochemical Pharmacology 34: 1357–1362, 1985
Isom JB. On the toxicity of valproic acid. American Journal of Diseases of Children 138: 901–903, 1984
Isom JB, Gerber N, Lynn RK, Harland RC, Kaufman SN, et al. Pharmacokinetic studies of valproic acid: serum half-life and tissue distribution in 2 children dying in acute hepatic failure. Annals of Neurology 6: 178, 1979
Jakobs C, Loscher W. Identification of metabolites of valproic acid in serum of humans, dog, rat and mouse. Epilepsia 19: 591–602, 1978
Jeavons PM. Hepatotoxicity of antiepileptic drugs. In Oxley et al. (Eds) Chronic toxicity of antiepileptic drugs, pp. 1–45, Raven Press, New York, 1983
Jeavons PM. Non-dose-related side effects of valproate. Epilepsia 25 (Suppl. 1): S5O–S55, 1984
Karlsen RL, Kett K, Henriksen OE. Intoxication with sodium valproate. Acta Medica Scandinavica 213: 405–406, 1983
Kay JDS, Hilton-Jones D, Hyman N. Valproate toxicity and ornithine carbamoyltransferase deficiency. Lancet 2: 1283–1284, 1986
Keene D, Humphreys P, Carpenter B, Reicher J. Poster presentation at Epilepsy International Congress, Kyoto, 1981. Cited by Jeavons PM. Hepatotoxicity of antiepileptic drugs. In Oxley et al. (Eds) Chronic toxicity of antiepileptic drugs, Raven Press, New York, 1983
Kerwin RW, Taberner PV. The mechanism of action of sodium valproate. General Pharmacology 12: 71–75, 1981
Kesterson JW, Granneman GR, Machinist JM. The hepatotoxicity of valproic acid and its metabolites in rats. I. Toxicologie, biochemical and histopathologic studies. Hepatology 4: 1143–1152, 1984
Keulen FP, Kochen W. Hepatotoxizitat unter Valproinsaure behandlung. Klinische Pädiatrie 197: 431–436, 1985
Kochen W, Imbeck H, Jakobs C. Untersuchungen uber die Ausscheidung von Metaboliten de Valproinsaure in Urin der Ratte und das Menschen. Arzneimittel Forschung 27: 1090–1099, 1977
Kochen W, Scheffner H. On unsaturated metabolites of the valproic acid (VPA) in serum of epileptic children. In Johannessen et al. (Eds) Antiepileptic therapy: advances in drug monitoring, pp. 111–120, Raven Press, New York, 1980
Kochen W, Schneider A, Ritz A. Abnormal metabolism of valproic acid in fatal hepatic failure. European Journal of Pediatrics 141: 30–35, 1983
Kochen W, Sprunck HP, Tauscher B, Klemens M. Five doubly unsaturated metabolites of valproic acid in urine and plasma of patients on valproic acid therapy. Journal of Clinical Chemistry and Clinical Biochemistry 22: 309–317, 1984
Kochi H, Hayasaka K, Hiraga K, Kikuchi G. Reduction of the level of the glycine cleavage system in the rat liver resulting from administration of dipropylacetic acid: an experimental approach to hyperglycinemia. Archives of Biochemistry and Biophysics 198: 589–597, 1979
Kuhara T, Hirakata Y, Yamada S, Matsumoto I. Metabolism of sodium dipropylacetate in humans. European Journal of Drug Metabolism and Pharmacokinetics 3: 171–177, 1978
Kuhara T, Inoue Y, Matsumoto M, Shinka T, Matsumoto I, et al. Altered metabolic profiles of valproic acid in a patient with Reye’s syndrome. Clinica Chimica Acta 145: 135–142, 1985
Kuhara T, Matsumoto I. Metabolism of branched medium chain length fatty acid, I: w-oxidation of sodium dipropylacetate in rats. Biomedical Mass Spectrometry 1: 291–294, 1974
Laub MC. Nutritional influence of serum ammonia in young patients receiving sodium valproate. Epilepsia 27: 55–59, 1986
Laub MC, Paetzke-Brunner I, Jaeger G. Serum carnitine during valproic acid therapy. Epilepsia 27: 559–562, 1986
Lewis JH, Zimmerman HJ, Garrett CT, Rosenberg E. Valproate-induced hepatic steatogenesis in rats. Hepatology 2: 870–873, 1982
Loscher W. Concentration of metabolites of valproic acid in plasma of epileptic patients. Epilepsia 22: 169–178, 1981
Loscher W, Bohme G, Schafer H, Kochen W. Effect of metabolites of valproic acid on the metabolism of G ABA in brain and brain nerve endings. Neuropharmacology 20: 1187–1192, 1981
Loscher W, Schmidt D. Increase of human plasma GABA by sodium valproate. Epilepsia 21: 611–615, 1980
Martin-Gallardo A, Rodriguez P, Lopez M, Benavides J, Ugarte M. Effect of dipropylacetate on the glycine cleavage enzyme system and glycine levels: a possible experimental approach to non-ketotic hyperglycinemia. Biochemical Pharmacology 16: 2877–2882, 1983
Matsumoto I, Kuhara T, Inoue Y, Matsumoto M, Ohkura T. Differential diagnosis for hepatopathy in five patients with valproate therapy. Proceedings of the 34th Annual Conference on Mass Spectrometry and Allied Topics, June 8–13, 1986, Cincinnati, Ohio, pp. 925–926, 1986
Matsumoto I, Kuhara T, Shinka T, Inoue Y, Matsumoto M. Effect of valproic acid on amino acid and organic acid metabolism. In Levy et al. (Eds) Metabolism of antiepileptic drugs, pp. 169–174, Raven Press, New York, 1984
Matsumoto I, Kuhara T, Yoshino M. Metabolism of branched chain medium length fatty acid II: β-oxidation of sodium dipropylacetate in rats. Biomedical Mass Spectrometry 3: 235–240, 1976a
Matsumoto I, Kuhara T, Yoshino M. The effect of isoleucine on the metabolism of sodium dipropylacetate in rats. Advances in Mass Spectrometry in Biochemistry and Medicine 1: 17–26, 1976b
Millington DS, Bohan TP, Roe CP, Yergey AL, Liberato DJ. Valproylcarnitine: a novel drug metabolite identified by fast atom bombardment and thermospray liquid chromatography-mass spectrometry. Clinica Chimica Acta 145: 69–76, 1985
Moore JR, Williams THC, Talbot IC, Tanner MS. Heterozygous α1-antitrypsin deficiency and valproate hepatotoxicity. Lancet 1: 221, 1984
Mortensen PB. Inhibition of fatty acid oxidation by valproate. Lancet 2: 856–857, 1980
Mortensen PB, Gregersen N, Kocvraa S, Christensen E. The occurrence of C6-C10-dicarboxylic acids in urine from patients and rats treated with dipropylacetate. Biochemical Medicine 24: 153–161, 1980a
Mortensen PB, Kolvraa S, Christensen E. Inhibition of the glycine cleavage system: hyperglycinemia and hyperglycinuria caused by valproic acid. Epilepsia 21: 563–569, 1980b
Nau H, Merker H-J, Brendel K, Gansan C, Hauser I, et al. Disposition, embryotoxicity and teratogenicity of valproic acid in the mouse as related to man. In Levy et al. (Eds) Metabolism of antiepileptic drugs, pp. 85–93, Raven Press, New York, 1984
Nau H, Loscher W. Valproic acid and metabolites: pharmacological and toxicological studies. Epilepsia 25 (Suppl. 1): S14–S22, 1984
Nau H, Wittfoht W, Schafer H, Jakobs C, Rating D, et al. Valproic acid and several metabolites: quantitative determination in serum, urine, breast milk and tissue by gas chromatographymass spectrometry using selected ion monitoring. Journal of Chromatography 226: 69–78, 1981
Ohtani Y, Endo F, Matsuda I. Carnitine deficiency and hyper-ammonemia associated with valproic acid therapy. Journal of Pediatrics 101: 782–785, 1982
Orr JM, Abbott FS, Farrell K, Ferguson S, Sheppard I, et al. Interaction between valproic acid and aspirin in epileptic children: serum protein binding and metabolic effects. Clinical Pharmacology and Therapeutics 31: 642–649, 1982
Panesar S, Orr J, Burton R, Farrell K. Effect of carbamazepine on valproic acid metabolism. Epilepsia 27: 591, 1986
Powell-Jackson PR, Tredger JM, Williams R. Hepatotoxicity to sodium valproate: a review. Gut 25: 673–681, 1984
Prickett KS, Baillie TA. Metabolism of valproic acid by hepatic microsomal cytochrome P450. Biochemical and Biophysical Research Communications 122: 1166–1173, 1984
Prickett KS, Baillie TA. Metabolism of unsaturated derivatives of valproic acid in rat liver microsomes and destruction of cytochrome P-450. Drug Metabolism and Disposition 14: 221–229, 1986
Rawat S, Borkowski WJ, Swick HM. Valproic acid and secondary hyperammonemia. Neurology 31: 1173–1174, 1981
Rettenmeier AW, Gordon WP, Prickett KS, Levy RH, Baillie TA. Biotransformation and pharmacokinetics in the rhesus monkey of 2-n-propyl-4-pentenoic acid, a toxic metabolite of valproic acid. Drug Metabolism and Disposition 14: 454–464, 1986a
Rettenmeier AW, Gordon WP, Prickett KS, Levy RH, Lockard JS, et al. Metabolic fate of valproic acid in the rhesus monkey: formation on a toxic metabolite, 2-n-propyl-4-pentenoic acid. Drug Metabolism and Disposition 14: 443–453, 1986b
Rettenmeier AW, Prickett KS, Gordon WP, Bjorge SM, Chang S-L, et al. Studies on the biotransformation in the perfused rat liver on 2-n-propyl-4-pentenoic acid, a metabolite of the antiepileptic drug valproic acid. Drug Metabolism and Disposition 15: 81–96, 1985
Rettie AE, Rettenmeier AW, Bayer BK, Baillie TA, Juchau MR. Valproate hydroxylation by human fetal tissues and embryotoxicity of metabolites. Clinical Pharmacology and Therapeutics 40: 172–177, 1986
Rettie AE, Rettenmeier AW, Howald WN, Baillie TA. Cytochrome P-450-catalysed formation of Δ4-VPA, a toxic metabolite of valproic acid. Science 235: 890–893, 1987
Rogiers A, Vandenberghe Y, Vercruysse A. Inhibition of gluconeogenesis by sodium valproate and its metabolites in isolated rat hepatocytes. Xenobiotica 15: 759–765, 1985
Rosenberg E. Disorders of propionate and methylmalonate metabolism. In Stanbury et al. (Eds) The metabolic basis of inherited disease, 5th ed., pp. 474–497, McGraw-Hill, New York, 1983
Rothner AD. Valproic acid: a review of 23 fatal cases. Annals of Neurology 10: 287, 1981
Rumbach L, Warter JM, Rendon A, Marescaux C, Micheletti G, et al. Inhibition of oxidative phosphorylation in hepatic and cerebral mitochondria of sodium valproate-treated rats. Journal of the Neurological Sciences 61: 417–423, 1983
Sawaya MCB, Horton RW, Meldrum BS. Effects of anticonvulsant drugs on the cerebral enzymes metabolising GABA. Epilepsia 16: 649–655, 1975
Schafer H, Luhrs R. Gas Chromatographie determination of the valproic acid metabolite artifacts, heptanone-3, 4- and 5-hydroxyvalproic acid lactone. Arzneimittel-Forschung 28: 657–662, 1978
Schafer H, Luhrs R. Responsibility of the metabolic pattern for potential side-effects in the rat being treated with valproic acid, 2-propylpenten-2-oic acid, and 2-propylpenten-4-oic acid. In Levy et al. (Eds) Metabolism of antiepileptic drugs, pp. 73–81, Raven Press, New York, 1984a
Schafer H, Luhrs R. Determination of 4,5-dihydroxy valproate, a target molecule in a potentially pathological valproate metabolism. Ninth European Workshop on Drug Metabolism, Pont-a-Mousson, 1984b
Scheffner D. Fatal liver failure in children on valproate. Lancet 2: 511, 1986
Schmid RD. Propionic acid and dipropylacetic acid in the urine of patients treated with dipropylacetic acid. Clinica Chimica Acta 74: 39–42, 1977
Schnabel R, Rambeck B, Janssen F. Fatal intoxication with sodium valproate. Lancet 1: 221–222, 1984
Sherratt HSA, Osmundsen H. On the mechanisms of some pharmacological actions of the hypoglycemic toxins hypoglycin and pent-4-enoic acid: a way out of the confusion. Biochemical Pharmacology 25: 743–750, 1976
Simila S, Von Wendt L, Linna S-L. Dipropylacetate and aminoaciduria. Journal of the Neurological Sciences 45: 83–86, 1980
Simon D, Penry JK. Sodium di-n-propylacetate (DPA) in the treatment of epilepsy. Epilepsia 16: 549–573, 1975
Stumpf DA, Parker Jr WD, Angelini C. Carnitine deficiency, organic acidemias and Reye’s syndrome. Neurology 35: 1041–1045, 1985
Stricker BHC, Spoelstra P. Drug-induced hepatic injury, pp. 84–87, Elsevier, Amsterdam, 1985
Suchy FJ, Balistreri WF, Buchino JJ, Sondheimer JM, Bates SR, et al. Acute hepatic failure associated with the use of sodium valproate. New England Journal of Medicine 300: 962–966, 1979
Sussman NM, McLain LW. A direct hepatotoxic effect of valproic acid. Journal of the American Medical Association 242: 1173–1174, 1979
Tanaka K, Rosenberg E. Disorders of branched chain amino acid and organic acid metabolism. In Stanbury et al. (Eds) The metabolic basis of inherited disease, 5th ed., pp. 440–473, McGraw Hill, New York, 1983
Thayer WS. Inhibition of mitochondrial fatty acid oxidation in pentenoic acid — induced fatty liver: a possible model for Reye’s syndrome. Biochemical Pharmacology 33: 1187–1194, 1984
Tripp JH, Hargreaves T, Anthony PP, Searle JF, Miller P, et al. Sodium valproate and ornithine carboxyl transferase deficiency. Lancet 1: 1165–1166, 1981
Turnbull DM, Bone AJ, Bartlett K, Koundakjian PP, Sherratt HSA. The effect of valproate on intermediary metabolism of isolated rat hepatocytes and intact rats. Biochemical Pharmacology 32: 1887–1892, 1983
Turnbull DM, Dick DJ, Wilson L, Sherratt HSA, Alberti KGMM. Valproate causes metabolic disturbance in normal man. Journal of Neurology, Neurosurgery and Psychiatry 49: 405–410, 1986
Van der Branden C, Roels F. Perioxisomal β-oxidation and sodium valproate. Biochemical Pharmacology 34: 2147–2149, 1985
Vree TB, van der Kleijn E. Pharmacokinetics and renal excretion of 2-n-propyl pentanoate (Depakine) in man, dog and Rhesus monkey. Pharmaceutisch Weekblad 112: 290–292, 1977
Warter JM, Brandt C, Marescaux C, Rumbach L, Micheletti G, et al. The renal origin of sodium valproate-induced hyperammonemia in fasting humans. Neurology 33: 1136–1140, 1983a
Warter JM, Imler M, Marescaux C, Chabrier G, Rumbach L, et al. Sodium valproate-induced hyperammonemia in the rat: role of the kidney. European Journal of Pharmacology 87: 177–182, 1983b
Williams CA, Tiefenbach S, McReynolds JW. Valproic acid-induced hyperammonemia in mentally retarded adults. Neurology 34: 550–553, 1984
Willmore LJ, Wilder BJ, Bruni J, Villarreal, HJ. Effect of valproic acid on hepatic function. Neurology 28: 961–964, 1978
Wolf B, Paulsen EP, Dreifuss FE. Valproate in the treatment of seizures associated with propionic acidemia. Pediatrics 67: 162–163, 1981
Zaccara G, Campostrini R, Paganini M, Moroni F, Valenza T, et al. Acute changes of blood ammonia may predict short-term adverse effects of valproic acid. Neurology 34: 1519–1521, 1984
Zafrani ES, Berthelot P. Sodium valproate in the induction of unusual hepatotoxicity. Hepatology 2: 648–649, 1982
Zimmerman HJ, Ishak KG. Valproate-induced hepatic injury: analyses of 23 fatal cases. Hepatology 2: 591–597, 1982
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eadie, M.J., Hooper, W.D. & Dickinson, R.G. Valproate-Associated Hepatotoxicity and its Biochemical Mechanisms. Dis-Manage-Health-Outcomes 3, 85–106 (1988). https://doi.org/10.1007/BF03259935
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03259935