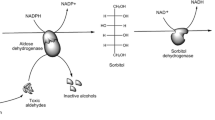
Abstract
A series of flavanone derivatives have been synthesized from 2-hydroxy acetophenone and benzaldehyde using fused calcium chloride in good to moderate yields, and their in vitro aldose reductase inhibitory activity has been tested on aldose reductase purified enzyme from Bovine lens. Most of the synthesized compounds exhibited potent aldose reductase inhibitory activity, and the obtained results are supported by the docking studies. Among the tested derivatives, 2, 3, 4-methoxy derivative 19 (IC50 5.88 ± 0.03 µM) exhibited the highest inhibitory activity whereas 2-methoxy derivative 12 showed the lowest, and the remaining compounds exhibited moderate activity with IC50 in the range of 6.09–7.89 µM. The spatial configuration of the most active derivative 19 was compared with pharmacophore requirements of the aldose reductase inhibitor site using a molecular modeling system.






Similar content being viewed by others
References
Aherne SA, O’Brien NM (2002) Nutrition 18:75–81
Akcok I, Çağır A (2010) Bioorg Chem 38:139–143
Bhatia VK, Krishnamurthy HG, Madhav R, Seshadri TR (1968) Tetrahedron Lett 9:3859–3862
Cabrera M, Simoens M, Falchi G, Lavaggi ML, Piro OE, Castellano EE, Vidal A, Azqueta A, Monge A, Lopez de Cerain A, Sagrera G, Seoane G, Cerecetto H, Gonzalez M (2007) Bioorg Med Chem 15:3356–3367
Chandrasekhar S, Vijeender K, Reddy KV (2005) Tetrahedron Lett 46:6991–6993
Chen PY, Wang TP, Chiang MY, Huang KS, Tzeng CC, Chen YL, Wang EC (2011) Tetrahedron 67:415–416
Chimenti F, Fioravanti R, Bolasco A, Chimenti P, Secci D, Rossi F, Yanez M, Orallo F, Ortuso F, Alcaro S, Cirilli R, Ferretti R, Sanna ML (2010) Bioorg Med Chem 18:1273–1279
Choudary BM, Ranganath K, Yadav J, Kantam M (2005) Tetrahedron Lett 46:1369–1372
Costantino L, Rastelli G, Gamberini M, Barlocco D (2000) Expert Opin Ther Pat 10:1245–1262
Dubrovskiy A, Larock R (2010) Org Lett 12:3117–3119
Gabbay K, Merola L, Field R (1996) Science 151:209–210
Ghahary A, Chakrabarti S, Sima A, Murphy L (1991) Diabetes 40:1391–1396
Hamada Y, Nakamura J, Naruse K, Komori T, Kato K, Kasuya Y, Nagai R, Horiuchi S, Hotta N (2000) Diabetes Care 23:1539–1544
Haraguchi H, Mochida Y, Sakai S, Masuda H, Tamura Y, Izutani K (1996a) Biosci Biotechnol Biochem 60:945–948
Haraguchi H, Ohmi I, Masuda H, Tamura Y, Mizutani K, Tanaka O (1996b) Experientia 52:564–567
Izumi T, Hino T, Kasahara A (1992) J Chem Soc Perkin Trans 1:1265–1267
Joglekar S, Samant S (1988) Tetrahedron Lett 29:241–244
Jung H, Islam M, Kwon Y, Jin S, Son Y, Park J (2011) Food Chem Toxicol 49:376–384
Kagawa H, Shigematsu A, Ohta S, Harigaya Y (2005) Chem Pharm Bull 53:547–554
Kavala V, Lin C, Kuo C, Fang H, Yao C (2012) Tetrahedron 68:1321–1329
Kigarashi K, Uchida Y, Murakami N, Mizutani K, Masuda H (1996) Biosci Biotechnol Biochem 60:513–515
Kulkarni P, Totawar B, Zubaidha PK (2012) Monatsh Chem 143:625–629
Kumar D, Patel G, Mishra B, Varma R (2008) Tetrahedron Lett 49:6974–6976
Kumar A, Sharma S, Tripathi V, Srivastava S (2010) Tetrahedron 66:9445–9449
Lee J, Jung M, Jung H (2007) Bull Korean Chem Soc 28:859–862
Maki Y, Shimada K, Sako M, Hirota K (1988) Tetrahedron 44:3187–3194
Matsushima R, Kageyama H (1985) J Chem Soc Perkin Trans 2:743–748
Middleton E, Kandaswami C, Theoharides T (2000) Pharmacol Rev 529:673–751
Miyamoto S (2002) Chem Bio Inform J 2:74–85
Monserrat J, Chabot G, Hamon L, Quentin L, Scherman D, Jaouen G, Hillard E (2010) Chem Commun 46:5145–5147
Obrosova L, Chung S, Kador P (2010) Diabetes Metab Res Rev 26:172–180
Pandey G, Krishna A, Kumaraswamy G (1987) Tetrahedron Lett 28:4615–4516
Po-Yuan C, Tzu-Pin W, Michael Y, Keng-Shian H, Cherng-Chyi T, Yi-Long C, Eng-Chi W (2011) Tetrahedron 67:4155–4160
Sanicanin Z, Tabakovic I (1986) Tetrahedron Lett 27:407–408
Sing O (1993) Org Prep Proced Int 25:693–695
Soni L, Gupta A, Kaskhedikar S (2008) Med Chem Res 17:258–266
Subramanian RS, Balasubramanian KK (1990) J Chem Soc Chem Commun 21:1469–1470
Sun W, Lee H, Park J, Kim S, Yu J, Kim J (2001) J Antibiot 54:827–830
Wang L, Liu X, Dong Z, Fu X, Feng X (2008) Angew Chem Int Ed 47:8670–8673
Wild S, Roglic A, Green R, Sicree H (2004) Diabetes Care 27:1047–1053
Wirasathien L, Pengsuparp T, Suttisri R, Ueda H, Moriyasu M, Kawanishi K (2007) Phytomedicine 14:546–550
Acknowledgements
We wish to express our sincere gratitude to the Department of Science and Technology, New Delhi, INDIA for providing research fellowship to Mr. Dasharath D. Kondhare under INSPIRE Program for the pursuit of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Rights and permissions
About this article
Cite this article
Kondhare, D., Gyananath, G., Tamboli, Y. et al. An efficient synthesis of flavanones and their docking studies with aldose reductase. Med Chem Res 26, 987–998 (2017). https://doi.org/10.1007/s00044-017-1813-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-017-1813-1