Abstract
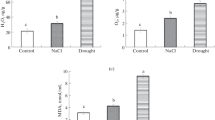
Being a considerable abiotic stress factor particularly for arid and semi-arid regions, salt stress may significantly limit the plant growth and yield. Plant’s response to salt stress involves secondary metabolites and especially phenylpropanoids that significantly contribute to the antioxidant activity of plant tissues. In addition to their important role in the control of cancer, phenylpropanoid compounds act as quenchers of singlet oxygen formation, free radical scavengers and reducing agents. One of the important gateway enzymes in the secondary metabolic pathway leading to the synthesis of phenylpropanoids is phenylalanine ammonia lyase (PAL). The aim of this study is to determine the phenolic acid composition, antioxidant capacity and antiproliferative effect associated with PAL activity in some plants that grow naturally under salt stress. The PAL activities of Salsola nitraria, Salvia halophila and Cyathobasis fruticulosa were evaluated. The antioxidant content of the extracts was studied and they were evaluated for their antioxidant activity. MTT assay was used to determine the antiproliferative effects of the extracts on HT-29 cells. Also, phenolic acids in extracts, namely p-coumaric acid, vanillic acid, gallic acid, caffeic acid, chlorogenic acid and syringic acid were screened using LC-MS/MS. Considering all results, C. fruticulosa with its highest PAL activity (62.85 μmol.min−1.mg−1 protein) has become prominent among the three plants. C. fruticulosa extract exhibited the highest antioxidant content with total phenolic content (120.36 mg/g) as the major antioxidant component. It was also found to be the plant extract richest in p-coumaric acid, vanillic acid, gallic acid, caffeic acid, chlorogenic acid and syringic acid as phenolic acids. Besides its marked antiproliferative activity against HT-29 cells, C. fruticulosa extract had the highest antioxidant activity compared with other extracts. In conclusion, the antioxidant and anticancer properties of plants naturally growing under salt stress may partly arise from their high PAL activities. Therefore, compounds obtained through plants exhibiting high levels of PAL activities could be used in the development of new pharmaceuticals as an antioxidant and anticancer agent.


Similar content being viewed by others
References
Acton A (2011) Protective agents: advances in research and application. A Scholarly Editions, Atlanta
Ajanal M, Gundkalle MB, Nayak SU (2012) Estimation of total alkaloid in Chitrakadivati by UV-Spectrophotometer. Anc Sci Life 31(4):198
Albayrak S, Aksoy A, Hamzaoğlu E (2008) Determination of antimicrobial and antioxidant activities of Turkish endemic Salvia halophila Hedge. Turk J Biol 32(4):265–270
Andjelković M, Van Camp J, De Meulenaer B, Depaemelaere G, Socaciu C, Verloo M, Verhe R (2006) Iron-chelation properties of phenolic acids bearing catechol and galloyl groups. Food Chem 98(1):23–31
Araújo JR, Gonçalves P, Martel F (2011) Chemopreventive effect of dietary polyphenols in colorectal cancer cell lines. Nutr Res 31(2):77–87
Asan-Ozusaglam M, Erzengin M, Darilmaz DO, Erkul SK, Teksen M, Karakoca K (2015) Antimicrobial and antioxidant activity of various solvent extracts of Salsola stenoptera Wagenitz and Petrosimonia nigdeensis Aellen (Chenopodiaceae) plants. Chiang Mai J Sci 42(1):156–172
Barcelo JM, Guieb M, Ventura A, Nacino A, Pinasen H, Viernes L, Binwag T (2014) Antibacterial, prooxidative and genotoxic activities of gallic acid and its copper and iron complexes against Escherichia coli. Asia Pac J Multidiscip Res 2(6):44–56
Blois MS (1958) Antioxidant determinations by the use of a stable free radical. Nature 181(4617):1199
Boz H (2015) p‐Coumaric acid in cereals: presence, antioxidant and antimicrobial effects. Int J Food Ccience & Technol 50(11):2323–2328
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1-2):248–254
Caverzan A, Passaia G, Rosa SB, Ribeiro CW, Lazzarotto F, Margis-Pinheiro M (2012) Plant responses to stresses: role of ascorbate peroxidase in the antioxidant protection. Genet Mol Biol 35(4):1011–1019
Chou TH, Ding HY, Hung WJ, Liang CH (2010) Antioxidative characteristics and inhibition of α‐melanocyte‐stimulating hormone‐stimulated melanogenesis of vanillin and vanillic acid from Origanum vulgare. Exp Dermatol 19(8):742–750
Choubey S, Varughese LR, Kumar V, Beniwal V (2015) Medicinal importance of gallic acid and its ester derivatives: a patent review. Pharm Pat Anal 4(4):305–315
Colla G, Rouphael Y, Cardarelli M, Svecova E, Rea E, Lucini L (2013) Effects of saline stress on mineral composition, phenolic acids and flavonoids in leaves of artichoke and cardoon genotypes grown in floating system. J Sci Food Agric 93(5):1119–1127
Corrêa NS, Bandeira JDM, Marini P, ICGD Borba, Lopes NF, Moraes DMD (2013) Salt stress: antioxidant activity as a physiological adaptation of onion cultivars. Acta Bot Bras 27(2):394–399
Danis TCP, Madeira VMC, Almeida MLM (1994) Action of phenolic derivates (acetoaminophen, salycilate and 5-amino salycilate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch Biochem Biophy 315:161–169
Decker EA, Elias R, McClements DJ (2010) Oxidation in foods and beverages and antioxidant applications. Woodhead Publishing, Cambridge
Decker EA, Welch B (1990) Role of ferritin as a lipid oxidation catalyst in muscle food. J Agric Food Chem 38(3):674–677
Farhoudi R, Hussain M, Dong-Jin L (2012) Modulation of enzymatic antioxidants improves the salinity resistance in canola (Brassica napus). International Journal of Agriculture and Biology 14(3):465–468
Fazel S, Hamidreza M, Rouhollah G, Verdian-rizi M (2010) Spectrophotometric determination of total alkaloids in some Iranian medicinal plants. Soc Adv Hortic 12(1):69–70
Finefrock AE, Bush AI, Doraiswamy PM (2003) Current status of metals as therapeutic targets in Alzheimer’s disease. J Am Geriatr Soc 51(8):1143–1148
Gao S, Ouyang C, Wang S, Xu Y, Tang L, Chen F (2008) Effects of salt stress on growth, antioxidant enzyme and phenylalanine ammonia-lyase activities in Jatropha curcas L. seedlings. Plant Soil Environ 54(9):374–381
Ghasemzadeh A, Ghasemzadeh N (2011) Flavonoids and phenolic acids: Role and biochemical activity in plants and human. J Med Plants Res 5(31):6697–6703
Gholizadeh A, Kohnehrouz BB (2010) Activation of phenylalanine ammonia lyase as a key component of the antioxidative system of salt-challenged maize leaves. Braz J Plant Physiol 22(4):217–223
Goldson A, Lam M, Scaman CH, Clemens S, Kermode A (2008) Screening of phenylalanine ammonia lyase in plant tissues, and retention of activity during dehydration. J Sci Food Agric 88(4):619–625
Güven M, Aras AB, Topaloğlu N, Özkan A, Şen HM, Kalkan Y, Çoşar M (2015) The protective effect of syringic acid on ischemia injury in rat brain. Turk J Med Sci 45(1):233–240
Halliwell B, Gutteridge JMC (2015) Free radicals in biology and medicine. Oxford University Press, Oxford
Hemmati S (2015) Phenylalanine ammonia-lyase through evolution: a bioinformatic approach. Trends Pharm Sci 1(1):10–14
Hsieh LS, Yeh CS, Pan HC, Cheng CY, Yang CC, Lee PD (2010) Cloning and expression of a phenylalanine ammonia-lyase gene (BoPAL2) from Bambusa oldhamii in Escherichia coli and Pichia pastoris. Protein Expr Purif 71(2):224–230
Kim SJ, Kim MC, Um JY, Hong SH (2010) The beneficial effect of vanillic acid on ulcerative colitis. Molecules 15(10):7208–7217
Klein BP, Perry AK (1982) Ascorbic acid and vitamin A activity in selected vegetables from different geographical areas of the United States. J Food Sci 47(3):941–945
Ksouri R, Megdiche W, Debez A, Falleh H, Grignon C, Abdelly C (2007) Salinity effects on polyphenol content and antioxidant activities in leaves of the halophyte Cakile maritima. Plant Physiol Biochem 45(3-4):244–249
Küçükboyacı N, Küpeli Akkol E, Suntar İ, Çalış İ (2016) In vivo anti-inflammatory and antinociceptiveactivities of the extracts and chemical constituents of an endemic Turkish Plant, Salsola grandis. Records of Natural Products 10(3):369–379
Lim JH, Park KJ, Kim BK, Jeong JW, Kim HJ (2012) Effect of salinity stress on phenolic compounds and carotenoids in buckwheat (Fagopyrum esculentum M.) sprout. Food Chem 135(3):1065–1070
Liu RH (2004) Potential synergy of phytochemicals in cancer prevention: mechanism of action. J Nutr 134(12):3479S–3485S
Locatelli C, Filippin-Monteiro FB, Creczynski-Pasa TB (2013) Alkyl esters of gallic acid as anticancer agents: a review. Eur J Med Chem 60:233–239
MacDonald MJ, D’Cunha GB (2007) A modern view of phenylalanine ammonia lyase. Biochem Cell Biol 85(3):273–282
Magnani C, Isaac VLB, Correa MA, Salgado HRN (2014) Caffeic acid: a review of its potential use in medications and cosmetics. Anal Methods 6(10):3203–3210
Miller NJ, Sampson J, Candeias LP, Bramley PM, Rice-Evans CA (1996) Antioxidant activities of carotenes and xanthophylls. FEBS Lett 384(3):240–242
Mohsen SM, Ammar AS (2009) Total phenolic contents and antioxidant activity of corn tassel extracts. Food Chem 112(3):595–598
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. Journal of Immunological Methods 65(1–2):55–63
Murtaza G, Sajjad A, Mehmood Z, Shah SH, Siddiqi AR (2015) Possible molecular targets for therapeutic applications of caffeic acid phenethyl ester in inflammation and cancer. Journal of Food and Drug Analysis 23(1):11–18
Nagata M, Yamashita I (1992) Simple method for simultaneous determination of chlorophyll and carotenoids in tomato fruit. Nippon Shokuhin Kogyo Gakkaishi 39(10):925–928
Nichenametla SN, Taruscio TG, Barney DL, Exon JH (2006) A review of the effects and mechanisms of polyphenolics in cancer. Crit Rev Food Sci Nutr 46(2):161–183
Ning L, Hua-Feng L, Zun-Chi L, Zheng-Xia C, Qiao-Mei L, Tong L (2015) Specificity of germination of heteromorphic seeds in four annuals (Salsola L.) at different temperatures in the Junggar Basin. Pak J Bot 47(3):867–876
Ohnishi M, Morishita H, Iwahashi H, Toda S, Shirataki Y, Kimura M, Kido R (1994) Inhibitory effects of chlorogenic acids on linoleic acid peroxidation and haemolysis. Phytochemistry 36(3):579–583
Pei K, Ou J, Huang J, Ou S (2016) p‐Coumaric acid and its conjugates: dietary sources, pharmacokinetic properties and biological activities. J Sci Food Agric 96(9):2952–2962
Reichert AI, He XZ, Dixon RA (2009) Phenylalanine ammonia-lyase (PAL) from tobacco (Nicotiana tabacum): characterization of the four tobacco PAL genes and active heterotetrameric enzymes. Biochem J 424(2):233–242
Rivas FJ, Beltran FJ, Frades J, Buxeda P (2001) Oxidation of p-hydroxybenzoic acid by Fenton’s reagent. Water Res 35(2):387–396
Rodriguez-Martinez MA, Ruiz-Torres A (1992) Homeostasis between lipid peroxidation and antioxidant enzyme activities in healthy human aging. Mech Ageing Dev 66(2):213–222
Ryu DS, Kim SH, Lee DS (2009) Anti-proliferative effect of polysaccharides from Salicornia herbacea on induction of G2/M arrest and apoptosis in human colon cancer cells. Journal of Microbiology and Biotechnology 19(11):1482–1489
Sairam RK, Srivastava GC, Agarwal S, Meena RC (2005) Differences in antioxidant activity in response to salinity stress in tolerant and susceptible wheat genotypes. Biol Plant 49(1):85
Sawa T, Nakao M, Akaike T, Ono K, Maeda H (1999) Alkylperoxyl radical-scavenging activity of various flavonoids and other phenolic compounds: implications for the anti-tumor-promoter effect of vegetables. J Agric Food Chem 47(2):397–402
Sharma I, Ching E, Saini S, Bhardwaj R, Pati PK (2013) Exogenous application of brassinosteroid offers tolerance to salinity by altering stress responses in rice variety Pusa Basmati-1. Plant Physiol Biochem 69:17–26
Shehab NG, Abu-Gharbieh E (2014) Phenolic profiling and evaluation of contraceptive effect of the ethanolic extract of Salsola imbricata Forssk in male albino rats. Evidence-Based Complementary and Alternative Medicine 2014, 1–8
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16(3):144–158
Stankovic MS, Niciforovic N, Mihailovic V, Topuzovic M, Solujic S (2012) Antioxidant activity, total phenolic content and flavonoid concentrations of different plant parts of Teucrium polium L. subsp. polium. Acta Societatis Botanicorum Poloniae 81(2):117–122
Stanojević L, Stanković M, Nikolić V, Nikolić L, Ristić D, Čanadanovic-Brunet J, Tumbas V (2009) Antioxidant activity and total phenolic and flavonoid contents of Hieracium pilosella L. extracts. Sensors 9(7):5702–5714
Şirin S, Aydaş SB, Aslım B (2016) Biochemical evaluation of phenylalanine ammonia lyase from endemic plant cyathobasis fruticulosa (Bunge) Aellen. for the dietary treatment of phenylketonuria. Food Technol Biotechnol 54(3):296
Tai A, Sawano T, Ito H (2012) Antioxidative properties of vanillic acid esters in multiple antioxidant assays. Biosci Biotechnol Biochem 76(2):314–318
Teixeira J, Gaspar A, Garrido EM, Garrido J, Borges F (2013) Hydroxycinnamic acid antioxidants: an electrochemical overview. BioMed Research International 2013, 1–10
Tohge TR, Fernie A (2017) An overview of compounds derived from the shikimate and phenylpropanoid pathways and their medicinal importance. Mini Rev Med Chem 17(12):1013–1027
Tovar MJ, Romero MP, Girona J, Motilva MJ (2002) l‐Phenylalanine ammonia‐lyase activity and concentration of phenolics in developing olive (Olea europaea L cv Arbequina) fruit grown under different irrigation regimes. J Sci Food Agric 82(8):892–898
Vogt T (2010) Phenylpropanoid biosynthesis. Mol Plant 3(1):2–20
Watson RR, Preedy VR (2013) Polyphenols in human health and disease. Academic Press, Elsevier, San Diego
Xiang Z, Ning Z (2008) Scavenging and antioxidant properties of compound derived from chlorogenic acid in South-China honeysuckle. LWT-Food Sci Technol 41(7):1189–1203
Yaltirak T, Aslim B, Ozturk S, Alli H (2009) Antimicrobial and antioxidant activities of Russula delica Fr. Food Chem Toxicol 47(8):2052–2056
Yuan G, Wang X, Guo R, Wang Q (2010) Effect of salt stress on phenolic compounds, glucosinolates, myrosinase and antioxidant activity in radish sprouts. Food Chem 121(4):1014–1019
Acknowledgements
We would like to thank Gazi University for funding the 46/2011-03 coded project including this study and declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Şirin, S., Aslım, B. Determination of antioxidant capacity, phenolic acid composition and antiproliferative effect associated with phenylalanine ammonia lyase (PAL) activity in some plants naturally growing under salt stress. Med Chem Res 28, 229–238 (2019). https://doi.org/10.1007/s00044-018-2278-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-018-2278-6