Abstract
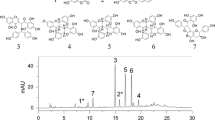
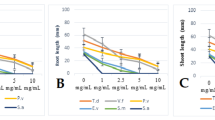
The allelopathic potential of fescue (Festuca arundinacea Shreb.) was investigated under field and laboratory conditions. In the field, incorporation of fescue aboveground biomass into the soil provided an effective weed control as it significantly reduced both the number and the dry weight of all weed species. The effect on weeds was species specific. The reduction of the most abundant species, Picris echioides, with respect to the number of individuals and their dry weight was 67 and 45%. Fescue aboveground biomass, harvested immediately before soil incorporation, was utilized in a bioassay-oriented fractionation, aimed at identifying the compounds in the toxic extracts of the plant. While phytotoxic activity was detected for the n-butanol fraction, no activity was found for the aqueous one. Germination of an indicator species, lettuce (Lactuca sativa L.), was reduced by 33% at the higher extract concentration of 20 mg/mL, owing to the increase of the number of abnormal seedlings. The LC/MS profile of the extract revealed the presence of two alkaloids, seven flavonol glycosides, and two flavonol aglyca. Quercetin 3-O-rutinoside, isorhamnetin 3-O-rutinoside, and kaempferol 3-O-rutinoside were reported in fescue for the first time.







Similar content being viewed by others
References
Belz R, Hurle K (2004) A novel laboratory screening bioassay for crop seedling allelopathy. J Chem Ecol 30:175–198
Bertin C, Paul RN, Duke SO, Weston LA (2003) Laboratory assessment of the allelopathic potential of fine leaf fescues (Festuca rubra L.). J Chem Ecol 8:1919–1937
Buer CS, Imin N, Djordjevic MA (2010) Flavonoids: new roles for old molecules. J Integr Plant Biol 52:98–111
Buta JG, Spaulding DW (1989) Allelochemicals in tall fescue-abscisic and phenolic acids. J Chem Ecol 15:1629–1636
Cuyckens F, Claeys M (2004) Mass spectrometry in the structural analysis of flavonoids. J Mass Spectrom 39:1–15
Elakovich SD, Wooten JW (1991) Allelopathic potential of Nuphar lutea (L.) Sibth. & SM. (Nymphaeaceae). J Chem Ecol 17:707–715
Gilmore AR (1977) How fescue Festuca arundinacea inhibits growth of sweetgum trees Liquidambar styraciflua. Ill Res 19:8–9
Inderjit, Weston LA (2000) Are laboratory bioassays for allelopathy suitable for prediction of field response? J Chem Ecol 26:2111–2118
Koulman A, Seeliger C, Edwards PJB, Fraser K, Simpson W, Johnson L, Cao M, Rasmussen S, Lane GA (2008) E/Z-thesinine-O-4′-α-rhamnoside, pyrrolizidine conjugates produced by grasses (Poaceae). Phytochemistry 69:1927–1932
Lovett JV, Jessor RS (1982) Effects of residues of crop plants on germination and early growth of wheat. Aust J Agric Res 33:909–916
Lovett JV, Ryuntyu M (1992) Allelopathy: broadnening the context. In: Rizvi JSH, Rizvi V (eds) Allelopathy: basic and applied aspects. Chapman & Hall, London, pp 11–18
Ma YL, Li QM, Van den Heuvel H, Claeys M (1997) Characterization of flavone and flavonol aglycones by collision-induced dissociation tandem mass spectrometry. Rapid Commun Mass Spectrom 11:1357–1364
Macias FA, Castellano D, Molinillo JMG (2000) Search for a standard phytotoxic bioassay for allelochemicals. Selection of standard target species. J Agric Food Chem 48:2512–2521
McCalla TM (1971) Studies on phytotoxic substances from microorganisms and crop residues at Lincoln, Nebraska. In: Biochemical interactions among Plants. National Academy of Sciences, Washington DC, pp 39–43
Oyun MB (2006) Allelopathic potentialities of Gliciridia sepium and Acacia auriculiformis on the germination and seedling vigour of maize (Zea mays L.). Am J Agric Biol Sci 1:44–47
Peters EJ, Lun KT (1985) Allelopathy in tall fescue. In: Thompson AC (ed) The chemistry of allelopathy. Symposium Series 268 American Chemical Society, Washington DC, pp 273–283
Rice EL (1984) Allelopathy, 2nd edn. Academic Press, New York
Rizvi SJH, Rizvi V (1992) Allelopathy: basic and applied aspects. Chapman & Hall, London
Sanchez-Moreiras AM, Weiss OA, Reigosa-Roger MJ (2004) Allelophatic evidence in the Poaceae. Bot Rev 69:300–319
Smith AE, Martin LD (1994) Allelopathic characteristics of three cool-season grass species in the forage ecosystem. Agron J 86:243–246
Snedecor GW, Cochran WG (1989) Statistical methods, 8th edn. University Press, Iowa State
Springer TL (1996) Allelopathic effects on germination and seedling growth of clovers by endophyte-free and -infected tall fescue. Crop Sci 36:1639–1642
Steel RGD, Torrie JH (1980) Principles and procedures of statistics: a biometrical approach. McGraw-Hill, New York
TA IS (2005) International rules for seed testing. The International Seed Testing Association, Basserdorf
Taylor LP, Grotewold E (2005) Flavonoids as developmental regulators. Curr Opin Plant Biol 8:317–323
Wollenweber E (1982) Flavones and flavonols. In: Harborne JB, Mabry TJ (eds) The flavonoids—advances in researches. Chapman and Hall, London, pp 240–242
Wong E (1980) Flavonol glycosides of tall fescue. NZ J Sci 23:39–42
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Res 14:415–421
Author information
Authors and Affiliations
Corresponding author
Additional information
C. de Bertoldi and M. De Leo contributed equally to this work.
Rights and permissions
About this article
Cite this article
de Bertoldi, C., De Leo, M., Ercoli, L. et al. Chemical profile of Festuca arundinacea extract showing allelochemical activity. Chemoecology 22, 13–21 (2012). https://doi.org/10.1007/s00049-011-0092-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-011-0092-4