Abstract
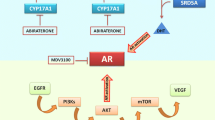
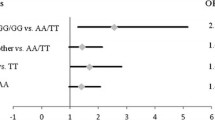
Since the tumor protein p53 (TP53), a transcription factor, plays a crucial role in prostate cancer development and progression, we hypothesized that sequence variants in TP53 binding sites might affect clinical outcomes in patients with prostate cancer. We systematically evaluated 41 single nucleotide polymorphisms (SNPs) within genome-wide predicted TP53 binding sites in a cohort of 1,024 prostate cancer patients. The associations of these SNPs with prostate cancer characteristics and clinical outcomes after radical prostatectomy for localized disease and after androgen-deprivation therapy (ADT) for advanced disease were assessed by Kaplan–Meier analysis and Cox regression model. ARAP2 rs1444377 and TRPS1 rs722740 were associated with advanced stage prostate cancer. FRK rs171866 remained as a significant predictor for disease progression; DAB2 rs268091 and EXOC4 rs1149558 remained as significant predictors for prostate cancer-specific mortality (PCSM); and EXOC4 rs1149558 remained as a significant predictor for all-cause mortality after ADT in multivariate models that included clinicopathologic predictors. In addition, the numbers of protective genotypes at DAB2 rs268091 and EXOC4 rs1149558 showed a cumulative effect on PCSM (P for trend = 0.002). Our results suggested that SNPs within TP53 binding sites might be valuable biomarkers for prostate cancer outcome prediction.


Similar content being viewed by others
Abbreviations
- SNP:
-
Single nucleotide polymorphism
- RP:
-
Radical prostatectomy
- ADT:
-
Androgen-deprivation therapy
- PCSM:
-
Prostate cancer-specific mortality
- ACM:
-
All-cause mortality
- PSA:
-
Prostate-specific antigen
- OR:
-
Odds ratio
- 95 % CI:
-
95 % confidence interval
- HR:
-
Hazard ratio
References
Amundson SA, Myers TG, Fornace AJ Jr (1998) Roles for p53 in growth arrest and apoptosis: putting on the brakes after genotoxic stress. Oncogene 17:3287–3299
Andriole GL, Crawford ED, Grubb RL 3rd et al (2009) Mortality results from a randomized prostate-cancer screening trial. N Engl J Med 360:1310–1319
Bao BY, Lin VC, Huang SH et al (2010a) Clinical significance of tumor necrosis factor receptor superfamily member 11b polymorphism in prostate cancer. Ann Surg Oncol 17:1675–1681
Bao BY, Pao JB, Lin VC et al (2010b) Individual and cumulative association of prostate cancer susceptibility variants with clinicopathologic characteristics of the disease. Clin Chim Acta 411:1232–1237
Bao BY, Pao JB, Huang CN et al (2011) Polymorphisms inside microRNAs and microRNA target sites predict clinical outcomes in prostate cancer patients receiving androgen-deprivation therapy. Clin Cancer Res 17:928–936
Bao BY, Pao JB, Huang CN et al (2012) Significant associations of prostate cancer susceptibility variants with survival in patients treated with androgen-deprivation therapy. Int J Cancer 130:876–884
Brauer PM, Tyner AL (2009) RAKing in AKT: a tumor suppressor function for the intracellular tyrosine kinase FRK. Cell Cycle 8:2728–2732
Bulyk ML (2003) Computational prediction of transcription-factor binding site locations. Genome Biol 5:201
Chang GT, Blok LJ, Steenbeek M, Veldscholte J, van Weerden WM, van Steenbrugge GJ, Brinkmann AO (1997) Differentially expressed genes in androgen-dependent and -independent prostate carcinomas. Cancer Res 57:4075–4081
Chang CF, Pao JB, Yu CC et al (2013) Common variants in IGF1 pathway genes and clinical outcomes after radical prostatectomy. Ann Surg Oncol 20:2446–2452
Dumont P, Leu JI, Della Pietra AC, George DL 3rd, Murphy M (2003) The codon 72 polymorphic variants of p53 have markedly different apoptotic potential. Nat Genet 33:357–365
Ecke TH, Schlechte HH, Hubsch A, Lenk SV, Schiemenz K, Rudolph BD, Miller K (2007) TP53 mutation in prostate needle biopsies—comparison with patients follow-up. Anticancer Res 27:4143–4148
Ferretti V, Poitras C, Bergeron D, Coulombe B, Robert F, Blanchette M (2007) PReMod: a database of genome-wide mammalian cis-regulatory module predictions. Nucl Acids Res 35:D122–D126
Freedland SJ, Sutter ME, Dorey F, Aronson WJ (2003) Defining the ideal cutpoint for determining PSA recurrence after radical prostatectomy. Prostate-specific antigen. Urology 61:365–369
Howell BW, Gertler FB, Cooper JA (1997) Mouse disabled (mDab1): a Src binding protein implicated in neuronal development. EMBO J 16:121–132
Huang SP, Huang CY, Wang JS et al (2007) Prognostic significance of p53 and X-ray repair cross-complementing group 1 polymorphisms on prostate-specific antigen recurrence in prostate cancer post radical prostatectomy. Clin Cancer Res 13:6632–6638
Huang SP, Huang LC, Ting WC et al (2009) Prognostic significance of prostate cancer susceptibility variants on prostate-specific antigen recurrence after radical prostatectomy. Cancer Epidemiol Biomarkers Prev 18:3068–3074
Huang SP, Ting WC, Chen LM et al (2010) Association analysis of Wnt pathway genes on prostate-specific antigen recurrence after radical prostatectomy. Ann Surg Oncol 17:312–322
Huang SP, Lan YH, Lu TL et al (2011) Clinical significance of runt-related transcription factor 1 polymorphism in prostate cancer. BJU Int 107:486–492
Huang CN, Huang SP, Pao JB et al (2012a) Genetic polymorphisms in androgen receptor-binding sites predict survival in prostate cancer patients receiving androgen-deprivation therapy. Ann Oncol 23:707–713
Huang CN, Huang SP, Pao JB et al (2012b) Genetic polymorphisms in oestrogen receptor-binding sites affect clinical outcomes in patients with prostate cancer receiving androgen-deprivation therapy. J Intern Med 271:499–509
Karolchik D, Hinrichs AS, Furey TS, Roskin KM, Sugnet CW, Haussler D, Kent WJ (2004) The UCSC table browser data retrieval tool. Nucl Acids Res 32:D493–D496
Li MS, Liu JL, Wu Y, Wang P, Teng H (2011) Meta-analysis demonstrates no association between p53 codon 72 polymorphism and prostate cancer risk. Genet Mol Res 10:2924–2933
Liu J, Yue P, Artym VV, Mueller SC, Guo W (2009) The role of the exocyst in matrix metalloproteinase secretion and actin dynamics during tumor cell invadopodia formation. Mol Biol Cell 20:3763–3771
Mok SC, Chan WY, Wong KK et al (1998) DOC-2, a candidate tumor suppressor gene in human epithelial ovarian cancer. Oncogene 16:2381–2387
Navone NM, Troncoso P, Pisters LL et al (1993) p53 protein accumulation and gene mutation in the progression of human prostate carcinoma. J Natl Cancer Inst 85:1657–1669
Pao JB, Yang YP, Huang CN et al (2013) Vitamin D receptor gene variants and clinical outcomes after androgen-deprivation therapy for prostate cancer. World J Urol 31:281–287
Roche PJ, Hoare SA, Parker MG (1992) A consensus DNA-binding site for the androgen receptor. Mol Endocrinol 6:2229–2235
Ross RW, Oh WK, Xie W et al (2008) Inherited variation in the androgen pathway is associated with the efficacy of androgen-deprivation therapy in men with prostate cancer. J Clin Oncol 26:842–847
Siegel R, Ward E, Brawley O, Jemal A (2011) Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin 61:212–236
Stacey SN, Sulem P, Jonasdottir A et al (2011) A germline variant in the TP53 polyadenylation signal confers cancer susceptibility. Nat Genet 43:1098–1103
Storey JD, Tibshirani R (2003) Statistical significance for genomewide studies. Proc Natl Acad Sci USA 100:9440–9445
Sun T, Lee GS, Oh WK et al (2010) Single-nucleotide polymorphisms in p53 pathway and aggressiveness of prostate cancer in a Caucasian population. Clin Cancer Res 16:5244–5251
Thorisson GA, Smith AV, Krishnan L, Stein LD (2005) The international HapMap project web site. Genome Res 15:1592–1593
Tseng CP, Ely BD, Li Y, Pong RC, Hsieh JT (1998) Regulation of rat DOC-2 gene during castration-induced rat ventral prostate degeneration and its growth inhibitory function in human prostatic carcinoma cells. Endocrinology 139:3542–3553
Visakorpi T, Kallioniemi AH, Syvanen AC et al (1995) Genetic changes in primary and recurrent prostate cancer by comparative genomic hybridization. Cancer Res 55:342–347
Xu B, Mi YY, Min ZC et al (2010) p53 codon 72 increased biochemical recurrence risk after radical prostatectomy in a southern Chinese population. Urol Int 85:401–405
Yang TP, Beazley C, Montgomery SB et al (2010) Genevar: a database and Java application for the analysis and visualization of SNP-gene associations in eQTL studies. Bioinformatics 26:2474–2476
Yoon HY, Miura K, Cuthbert EJ, Davis KK, Ahvazi B, Casanova JE, Randazzo PA (2006) ARAP2 effects on the actin cytoskeleton are dependent on Arf6-specific GTPase-activating-protein activity and binding to RhoA-GTP. J Cell Sci 119:4650–4666
Yu CC, Huang SP, Lee YC et al (2013a) Molecular markers in sex hormone pathway genes associated with the efficacy of androgen-deprivation therapy for prostate cancer. PLoS ONE 8:e54627
Yu CC, Lin VC, Huang CY et al (2013b) Prognostic significance of cyclin D1 polymorphisms on prostate-specific antigen recurrence after radical prostatectomy. Ann Surg Oncol 20(Suppl 3):492–499
Zeitlinger J, Simon I, Harbison CT, Hannett NM, Volkert TL, Fink GR, Young RA (2003) Program-specific distribution of a transcription factor dependent on partner transcription factor and MAPK signaling. Cell 113:395–404
Acknowledgments
This work was supported by grants from the National Science Council (NSC), Taiwan (NSC-98-2320-B-039-019-MY3, NSC-100-2314-B-039-009-MY3, and NSC-102-2628-B-039-005-MY3) and Kaohsiung Medical University Hospital (KMUH100-0R42). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. We also thank Chao-Shih Chen for data analysis and the National Center for Genome Medicine, NSC, Taiwan, for technical support.
Conflict of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Victor C. Lin and Chao-Yuan Huang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lin, V.C., Huang, CY., Lee, YC. et al. Genetic variations in TP53 binding sites are predictors of clinical outcomes in prostate cancer patients. Arch Toxicol 88, 901–911 (2014). https://doi.org/10.1007/s00204-014-1196-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-014-1196-8