Abstract
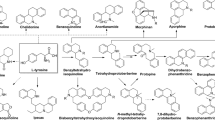
The exploration of new chemical entities from herbal medicines may provide candidates for the in silico screening of drug leads. However, this significant work is hindered by the presence of multiple classes of plant metabolites and many re-discovered structures. This study presents an integrated strategy that uses ultrahigh-performance liquid chromatography/linear ion-trap quadrupole/Orbitrap mass spectrometry (UHPLC/LTQ-Orbitrap-MS) coupled with in-house library data for the systematic characterization and discovery of new potentially bioactive molecules. Exploration of the indole alkaloids from Uncaria rhynchophylla (UR) is presented as a model study. Initially, the primary characterization of alkaloids was achieved using mass defect filtering and neutral loss filtering. Subsequently, phytochemical isolation obtained 14 alkaloid compounds as reference standards, including a new one identified as 16,17-dihydro-O-demethylhirsuteine by NMR analyses. The direct-infusion fragmentation behaviors of these isolated alkaloids were studied to provide diagnostic structural information facilitating the rapid differentiation and characterization of four different alkaloid subtypes. Ultimately, after combining the experimental results with a survey of an in-house library containing 129 alkaloids isolated from the Uncaria genus, a total of 92 alkaloids (60 free alkaloids and 32 alkaloid O-glycosides) were identified or tentatively characterized, 56 of which are potential new alkaloids for the Uncaria genus. Hydroxylation on ring A, broad variations in the C-15 side chain, new N-oxides, and numerous O-glycosides, represent the novel features of the newly discovered indole alkaloid structures. These results greatly expand our knowledge of UR chemistry and are useful for the computational screening of potentially bioactive molecules from indole alkaloids.

A four-step integrated strategy for the systematic characterization and efficient discovery of new indole alkaloids from Uncaria rhynchophylla







Similar content being viewed by others
Abbreviations
- DPIs:
-
Diagnostic product ions
- ESI-CID-MSn :
-
Electrospray ionization–collision-induced dissociation–tandem mass spectrometric fragmentation
- MDF:
-
Mass defect filtering
- NLF:
-
Neutral loss filtering
- pMIA:
-
Pentacyclic monoterpene indole alkaloids
- RDBeq:
-
Ring double-bond equivalent
- tMIA:
-
Tetracyclic monoterpene indole alkaloids
- tMIN:
-
Tetracyclic monoterpene indole N-oxides
- tMOA:
-
Tetracyclic monoterpene oxindole alkaloids
- UHPLC/LTQ-Orbitrap:
-
Ultra-high performance liquid chromatography/linear ion-trap quadrupole/Orbitrap
- UR:
-
Uncaria rhynchophylla
References
Bucar F, Wube A, Schmid M (2013) Nat Prod Res 30:525–545
Yang WZ, Hu Y, Wu WY, Ye M, Guo DA (2014) Phytochemistry 106:7–24
Yang WZ, Ye M, Qiao X, Liu CF, Miao WJ, Bo T, Tao HY, Guo DA (2012) Anal Chim Acta 739:56–66
Sticher O (2008) Nat Prod Res 25:517–554
Yang J, Liang Q, Wang M, Jeffries C, Smithson D, Tu Y, Boulos N, Jacob MR, Shelat AA, Wu YS, Ravu RR, Gilbertson R, Avery MA, Khan IA, Walker LA, Guy RK, Li XC (2014) J Nat Prod 77:902–909
Klitgaard A, Iversen A, Andersen MR, Larsen TO, Frisvad JC, Nielsen KF (2014) Anal Bioanal Chem 406:1933–1943
Pawlus AD, Cantos-Villar E, Richard T, Bisson J, Poupard P, Papastamoulis Y, Monti JP, Teissedre PL, Waffo-Téguo P, Mérillon JM (2013) J Chromatogr A 1289:19–26
Zhou JL, Qi LW, Li P (2009) J Chromatogr A 1216:7582–7594
Cao G, Chen XC, Wu X, Li QL, Zhang HY (2015) Biomed Chromatogr 29:698–708
Le PM, McCooeye M, Windust A (2013) Anal Bioanal Chem 405:4487–4498
Zhang JX, Guan SH, Sun JH, Liu T, Chen P, Feng RH, Chen X, Wu WY, Yang M, Guo DA (2015) Anal Bioanal Chem 407:581–595
Kalli A, Smith GT, Sweredoski MJ, Hess S (2013) J Proteome Res 12:3071–3086
Zhang JY, Wang ZJ, Zhang Q, Wang F, Ma Q, Lin ZZ, Lu JQ, Qiao YJ (2014) Talanta 124:111–122
Ji S, Wang Q, Qiao X, Guo HC, Yang YF, Bo T, Xiang C, Guo DA, Ye M (2014) J Pharm Biomed Anal 90:15–26
Qiao S, Shi XW, Shi R, Liu M, Liu T, Zhang KR, Wang Q, Yao MC, Zhang LT (2013) Anal Bioanal Chem 405:6721–6738
Sleno L (2012) J Mass Spectrom 47:226–236
Sleighter RL, Hatcher PG (2007) J Mass Spectrom 42:559–574
Zhang JY, Wang F, Zhang H, Lu JQ, Qiao YJ (2014) Phytochem Anal 25:405–414
Wang F, Zhang JY, Yin PH, Wang ZJ, Dong LY, Lu JQ (2014) Anal Methods 6:3515–3523
Mo ZX, Xu DD (2006) Lishizhen Med Mater Med Res 17:684–685
Ndagijimana A, Wang XM, Pan GX, Zhang F, Feng H, Olaleye O (2013) Fitoterapia 86:33–47
Zhang YB, Yang WZ, Yao CL, Feng RH, Yang M, Guo DA, Wu WY (2014) Fitoterapia 96:39–47
Wang JK, Liu JL (2011) Nat Prod Res Dev 23:776–788
Heitzman ME, Neto CC, Winiarz E, Vaisberg AJ, Hammond GB (2005) Phytochemistry 66:5–29
Lee J, Son D, Lee P, Kim SY, Kim H, Kim CJ, Lim E (2003) Neurosci Lett 348:51–55
Jung HY, Nam KN, Woo BC, Kim KP, Kim SO, Lee EH (2013) Mol Med Rep 7:154–158
Commission of National Pharmacopoeia (2010) China pharmacopoeia (part I). Chinese Medical Science and Technology Press, Beijing
Xie SL, Shi YY, Wang YX, Wu CY, Liu WY, Feng F, Xie N (2013) J Pharm Biomed Anal 81–82:56–64
Gai YN, Chen H, Wu CY, Feng F, Wang YX, Liu WY, Wang SL (2013) J Sep Sci 36:3723–3732
Yang WZ, Ye M, Qiao X, Wang Q, Bo T, Guo DA (2012) Eur J Mass Spectrom 18:493–503
Yang WZ, Qiao X, Bo T, Wang Q, Guo DA, Ye M (2014) Rapid Commun Mass Spectrom 28:385–395
Wenkert E, Bindra JS, Chang CJ, Cochran DW, Schell FM (1974) Acc Chem Res 7:45–51
Wang HB, Qi W, Zhang L, Yuan D (2014) Chem Pharm Bull 62:1100–1109
Kushida H, Fukutake M, Tabuchi M, Katsuhara T, Nishimura H, Ikarashi Y, Kanitani M, Kase Y (2013) Biomed Chem 27:1647–1656
Ma B, Wu CF, Yang JY, Wang R, Kano Y, Yuan D (2009) Helv Chim Acta 92:1575–1585
Acknowledgments
The authors acknowledge the financial support from the Twelfth Five-Year National Science & Technology Support Program (2012BAI29B06), the National Science and Technology Major Project for Major Drug Development (2013ZX09508104 and 2014ZX09304-307-001-007), and Special Scientific Research in the Chinese Medicine Industry of State Administration of Traditional Chinese Medicine (201307002).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Huiqin Pan and Wenzhi Yang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 2692 kb)
Rights and permissions
About this article
Cite this article
Pan, H., Yang, W., Zhang, Y. et al. An integrated strategy for the systematic characterization and discovery of new indole alkaloids from Uncaria rhynchophylla by UHPLC/DAD/LTQ-Orbitrap-MS. Anal Bioanal Chem 407, 6057–6070 (2015). https://doi.org/10.1007/s00216-015-8777-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-015-8777-0