Abstract
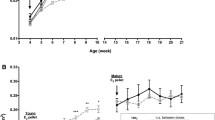
Our purpose was to characterize changes in bone remodeling associated with localized radiation that models therapeutic cancer treatment in ovary-intact (I) and ovariectomized (OVX) mice and to evaluate the influence of radiation on the pattern of bone mineral remodeling. Young adult, female BALB/c mice, I and OVX, were used (n = 71). All mice were intravenously injected with 15 μCi 45Ca. Thirty days post-45Ca administration, the hind limbs of 17 mice were exposed to a single dose of 16 Gy radiation (R). The time course of 45Ca excretion, serum CTx and osteocalcin markers, and cancellous bone volume fraction (BV/TV) and cortical thickness (Ct.Th) of the distal femur were assayed. Cellular activity and dynamic histomorphometry were performed. Irradiation resulted in rapid increases in fecal 45Ca excretion compared to control groups, indicating increased bone remodeling. CTx increased rapidly after irradiation, followed by an increase in osteocalcin concentration. BV/TV decreased in the I mice following irradiation. Ct.Th increased in the OVX groups following irradiation. I+R mice exhibited diminished osteoblast surface, osteoclast number, and mineral apposition. Our murine model showed the systemic effects (via 45Ca excretion) and local effects (via bone microarchitecture and surface activity) of clinically relevant, therapeutic radiation exposure. The I and OVX murine models have similar 45Ca excretion but different bone microarchitectural responses. The 45Ca assay effectively indicates the onset and rate of systemic bone mineral remodeling, providing real-time assessment of changes in bone histomorphometric parameters. Monitoring bone health via a bone mineral marker may help to identify the appropriate time for clinical intervention to preserve skeletal integrity.






Similar content being viewed by others
References
Guise T (2006) Bone loss and fracture risk associated with cancer therapy. Oncologist 11:1121–1131
Chen Z, Maricic M, Bassford T, Pettinger M, Ritenbaugh C, Lopez A, Barad D, Gass M, LeBoff M (2005) Fracture risk among breast cancer survivors: results from the Women’s Health Initiative Observational Study. Arch Intern Med 165:552
Baxter N, Habermann E, Tepper J, Durham S, Virnig B (2005) Risk of pelvic fractures in older women following pelvic irradiation. JAMA 294:2587–2593
Garnero P (2000) Markers of bone turnover for the prediction of fracture risk. Osteoporos Int 11(Suppl 6):S55–S65
Johnell O, Oden A, De Laet C, Garnero P, Delmas P, Kanis J (2002) Biochemical indices of bone turnover and the assessment of fracture probability. Osteoporos Int 13:523–526
Heaney RP (2003) Is the paradigm shifting? Bone 33:457–465
Seeman E, Delmas P (2006) Bone quality—the material and structural basis of bone strength and fragility. N Engl J Med 354:2250
Riis BJ, Hansen MA, Jensen AM, Overgaard K, Christiansen C (1996) Low bone mass and fast rate of bone loss at menopause: equal risk factors for future fracture: a 15-year follow-up study. Bone 19:9–12
Riggs BL, Melton LJ III (2002) Bone turnover matters: the raloxifene treatment paradox of dramatic decreases in vertebral fractures without commensurate increases in bone density. J Bone Miner Res 17:11–14
Hui SK, Fairchild GR, Kidder LS, Sharma M, Bhattacharya M, Jackson S, Le C, Yee D (2012) Skeletal remodeling following clinically relevant radiation-induced bone damage treated with zoledronic acid. Calcif Tissue Int 90:40–49
Small W Jr, Kachnic L (2005) Postradiotherapy pelvic fractures: cause for concern or opportunity for future research? JAMA 294:2635
Becker KL (2001) Principles and practice of endocrinology and metabolism. Lippincott Williams & Wilkins, Philadelphia
Cheong J, Gunaratna N, McCabe G, Jackson G, Weaver C (2009) Bone seeking labels as markers for bone turnover: effect of dosing schedule on labeling various bone sites in rats. Calcif Tissue Int 85:444–450
Sharma M, Bajzer Z, Hui SK (2011) Development of 41Ca-based pharmacokinetic model for the study of bone remodelling in humans. Clin Pharmacokinet 50:191–199
Välimäki M, Kinnunen K, Volin L, Tähtelä R, Löyttyniemi E, Laitinen K, Mäkelä P, Keto P, Ruutu T (1999) A prospective study of bone loss and turnover after allogeneic bone marrow transplantation: effect of calcium supplementation with or without calcitonin. Bone Marrow Transplant 23:355–361
Carlson K, Simonsson B, Ljunghall S (1994) Acute effects of high-dose chemotherapy followed by bone marrow transplantation on serum markers of bone metabolism. Calcif Tissue Int 55:408–411
Kang M, Lee W, Oh K, Han J, Song K, Cha B, Lee K, Son H, Kang S, Kim C (2000) The short-term changes of bone mineral metabolism following bone marrow transplantation. Bone 26:275–279
Denk E, Hillegonds D, Hurrell RF, Vogel J, Fattinger K, Hauselmann HJ, Kraenzlin M, Walczyk T (2007) Evaluation of 41calcium as a new approach to assess changes in bone metabolism: effect of a bisphosphonate intervention in postmenopausal women with low bone mass. J Bone Miner Res 22:1518–1525
Hui S, Prior J, Gelbart Z, Johnson R, Lentle B, Paul M (2007) A pilot study of the feasibility of long-term human bone balance during perimenopause using a 41Ca tracer. Nucl Instrum Methods Phys Res B 259:796–800
Elmore D, Bhattacaryya MH, Gibson NS (1990) 41Ca as a long-term biological tracer for bone resorption. Nucl Instrum Methods 52:531–535
Fowler J (2006) Development of radiobiology for oncology—a personal view. Phys Med Biol 51:263
Ma C (2002) AAPM TG-61 report on kilovoltage X-ray dosimetry: formalisms and applications. In: Proceedings of the 22nd Annual International Conference of the IEEE, Engineering in Medicine and Biology Society, Chicago, 23–28 July 2000, pp 2308–2312
Seuntjens J (2002) AAPM TG-61 report on kilovoltage X-ray dosimetry. II. Calibration procedures and correction factors. In: Proceedings of the 22nd Annual International Conference of the IEEE, Engineering in Medicine and Biology Society, Chicago, 23–28 July 2000, pp 2313–2316
Hui SK, Sharma M, Bhattacharyya M (2012) Liquid scintillation based quantitative measurement of dual radioisotopes (3H and 45Ca) in biological samples for bone remodeling studies. Appl Radiat Isot 70:63–68
Bouxsein M, Boyd S, Christiansen B, Guldberg R, Jepsen K, Müller R (2010) Guidelines for assessment of bone microstructure in rodents using microcomputed tomography. J Bone Miner Res 25:1468–1486
Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR (1987) Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 2:595–610
Recker RR, Kimmel DB, Parfitt AM, Davies KM, Keshawarz N, Hinders S (1988) Static and tetracycline-based bone histomorphometric data from 34 normal postmenopausal females. J Bone Miner Res 3:133–144
Weitzmann MN, Pacifici R (2006) Estrogen deficiency and bone loss: an inflammatory tale. J Clin Invest 116:1186
Klinck J, Campbell GM, Boyd SK (2007) Radiation effects on bone architecture in mice and rats resulting from in vivo micro-computed tomography scanning. Med Eng Phys 30:888–895
Willey J, Lloyd S, Robbins M, Bourland J, Smith-Sielicki H, Bowman L, Norrdin R, Bateman T (2008) Early increase in osteoclast number in mice after whole-body irradiation with 2 Gy X rays. Radiat Res 170:388–392
Hui SK, Khalil A, Zhang Y, Coghill K, Le C, Dusenbery K, Froelich J, Yee D, Levi D (2010) Longitudinal assessment of bone loss from diagnostic CT scans in gynecologic cancer patients treated with chemotherapy and radiation. Am J Obstet Gynecol 203:e351–e353
Cao X, Wu X, Frassica D, Yu B, Pang L, Xian L, Wan M, Lei W, Armour M, Tryggestad E (2011) Irradiation induces bone injury by damaging bone marrow microenvironment for stem cells. Proc Natl Acad Sci USA 108:1609
Chen H, Lee B, Guo H, Su W, Chiu N (2002) Changes in bone mineral density of lumbar spine after pelvic radiotherapy. Radiother Oncol 62:239–242
Overgaard M (1988) Spontaneous radiation-induced rib fractures in breast cancer patients treated with postmastectomy irradiation—a clinical radiobiological analysis of the influence of fraction size and dose-response relationships on late bone damage. Acta Oncol 27:117–122
Wientroub S, Weiss J, Catravas G, Reddi A (1990) Influence of whole body irradiation and local shielding on matrix-induced endochondral bone differentiation. Calcif Tissue Int 46:38–45
Acknowledgments
This work was supported by the National Institutes of Health (grants 1R03AR055333-01A1 and 1K12-HD055887-01) and by the PHS Cancer Center Support (grant P30 CA77398) and the Joseph E. Wargo cancer research fund from the University of Minnesota. S. K. H. is a scholar of the Building Interdisciplinary Careers in Women’s Health program. The authors acknowledge and thank the University of Minnesota – University Imaging Center, for aid in UV microscopy and Luke Arentsen, Kathleen Coghill, and Eftu Boru for laboratory assistance as well as Rajaram Gopalakrishnan, Kim Mansky, and Seymor Levitt for fruitful discussion pertaining to this experiment and report.
Disclosures
The authors also state that they have full control of all primary data and they agree to allow Calcified Tissue to review their data if requested.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have stated that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hui, S.K., Fairchild, G.R., Kidder, L.S. et al. The Influence of Therapeutic Radiation on the Patterns of Bone Remodeling in Ovary-Intact and Ovariectomized Mice. Calcif Tissue Int 92, 372–384 (2013). https://doi.org/10.1007/s00223-012-9688-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-012-9688-0