Abstract
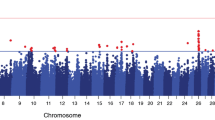
Mastitis is a detrimental disease in the dairy industry that decreases milk quality and costs upwards of $2 billion annually. Often, mastitis results from bacteria entering the gland through the teat opening. Streptococcus uberis is responsible for a high percentage of subclinical and clinical mastitis. Following an intramammary experimental challenge with S. uberis on Holstein cows (n = 40), milk samples were collected and somatic cell counts (SCC) were determined by the Dairy Herd Improvement Association Laboratory. Traditional genome-wide association studies (GWAS) have utilized test day SCC or SCC lactation averages to identify loci of interest. Our approach utilizes SCC collected following a S. uberis experimental challenge to generate three novel phenotypes: (1) area under the curve (AUC) of SCC for 0–7 days and (2) 0–28 days post-challenge; and (3) when SCC returned to below 200,000 cells/mL post-challenge (< 21 days, 21–28 days, or > 28 days). Polymorphisms were identified using Illumina’s BovineSNP50 v2 DNA BeadChip. Associations were tested using Plink software and identified 16 significant (p < 1.0 × 10−4) single-nucleotide polymorphisms (SNPs) across the phenotypes. Most significant SNPs were in genes linked to cell signaling, migration, and apoptosis. Several have been recognized in relation to infectious processes (ATF7, SGK1, and PACRG), but others less so (TRIO, GLRA1, CELSR2, TIAM2, CPE). Further investigation of these genes and their roles in inflammation (e.g., SCC) can provide potential targets that influence resolution of mammary gland infection. Likewise, further investigation of the identified SNP with mastitis and other disease phenotypes can provide greater insight to the potential of these SNP as genetic markers.





Similar content being viewed by others
References
Alter A, Fava VM, Huong NT, Singh M, Orlova M, Van Thuc N et al (2013) Linkage disequilibrium and age-at-diagnosis are critical for replicating genetic associations across ethnic groups in leprosy. Hum Genet 132:107–116
Baeuerle PA, Henkel T (1994) Function and activation of NF-kappaB in the immune system. Annu Rev Immunol 12:141–179
Bannerman DD, Paape MJ, Goff JP, Kimura K, Lippolis JD, Hope JC (2004) Innate immune respons to intramammary infection with Serratia marcescens and Streptococcus uberis. Vet Res 35:681–700
Beall SA, Boekelheide K, Johnson KJ (2005) Hybrid GPCR/cadherin (Celsr) proteins in rat testis are expressed with cell type specificity and exhibit differential sertoli cell—germ cell adhesion activity. J Androl 26:529–538
Boussaha M, Esquerre D, Barbieri J, Djari A, Pinton A, Letaief R et al (2015) Genome-wide study of structural variants in bovine Holstein, Montbeliarde and Normande dairy breeds. PLoS One 10(8):e0135931
Bradley AJ, Leach KA, Breen JE, Green LE, Green MJ (2007) Survey of the incidence and aetiology of mastitis on dairy farms in England and Wales. Vet Rec 160:253–258
Bragina EY, Trys ES, Rudko AA, Ivanisenko VA, Freidin MB (2016) Novel tuberculosis susceptibility candidate genes revealed by the reconstruction and analysis of associative networks. Infect Genet Evol 46:118–123
Chen JS, Su IJ, Leu YW, Young KC, Sun HS (2012) Expression of T-cell lymphoma invasion and metastasis 2 (TIAM2) promotes proliferation and invasion of liver cancer. Int J Cancer 130:1302–1313
Chiu CY, Leng S, Martin KA, Kim E, Gorman S, Duhl DM (1999) Cloning and characterization of T-cell lymphoma invasion and metastasis 2 (TIAM2), a novel guanine nucleotide exchange factor related to TIAM1. Genomics 61:66–73
Cole JB, VanRaden PM, O’Connell JR, Van Tassell CP, Sonstegard TS, Schnabel RD et al (2009) Distribution and location of genetic effects for dairy traits. J Dairy Sci 92:2931–2946
Cole JB, Wiggans GR, Ma L, Sonstegard TS, Lawlor TJ Jr, Crooker BA et al (2011) Genome-wide association analysis of thirty one production, health, reproduction and body conformation traits in contemporary U.S. Holstein cows. BMC Genomics 12:408. https://doi.org/10.1186/1471-2164-12-408
Collins T, Read MA, Neish AS, Whitley MZ, Thanos D, Maniatis T (1995) Transcriptional regulation of endothelial cell adhesion molecules: NF-kappa B and cytokine-inducible enhancers. FASEB J 9:899–909
Croft D, Mundo AF, Haw R, Milacic M, Weiser J, Wu G, Caudy M, Garapati P, Gillespie M, Kamdar MR, Jassal B, Jupe S, Matthews L, May B, Palatnik S, Rothfels K, Shamovsky V, Song H, Williams M, Birney E, Hermjakob H, Stein L, D'Eustachio P (2014) The Reactome pathway knowledgebase. Nucleic Acids Res 42:D472–D477
de Roos APW, Hayes BJ, Spelman RJ, Goddard ME (2008) Linkage disequilibrium and persistence of phase in Holstein–Friesian, Jersey and Angus cattle. Genetics 179:1503–1512
Dohoo IR, Smith J, Andersen S, Kelton DF, Godden S (2011) Diagnosing intramammary infections: evaluation of definitions based on a single milk sample. J Dairy Sci 94:250–261
Fabregat A, Jupe S, Matthews L, Sidiropoulos K, Gillespie M, Garapati P, Haw R, Jassal B, Korninger F, May B, Milacic M, Roca CD, Rothfels K, Sevilla C, Shamovsky V, Shorser S, Varusai T, Viteri G, Weiser J, Wu G, Stein L, Hermjakob H, D’Eustachio P (2018) The reactome pathway knowledgebase. Nucleic Acids Res 46:D649–D655
Fricker LD (1988) Carboxypeptidase E. Annu Rev Physiol 50:309–321
Gerard NP, Gerard C (1991) The chemotactic receptor for human C5a anaphylatoxin. Nature 349:614–617
Gunderson KL, Steemers FJ, Lee G, Mendoza LG, Chee MS (2005) A genome-wide scalable SNP genotyping assay using microarray technology. Nat Genet 37:549–554
Haile-Mariam M, Bowman PJ, Goddard ME (2003) Genetic and environmental relationship among calving interval, survival, persistency of milk yield and somatic cell count in dairy cattle. Livest Prod Sci 80:189–200
Han X, Tachado SD, Koziel H, Boisvert WA (2012) Leu128 (L128) and Val247(V247) of CXCR1 are critical amino acid residues for g protein coupling and receptor activation. PLoS One 7:e42765
Jayarao BM, Oliver SP (1994) Polymerase chain reaction-based DNA fingerprinting for identification of Streptococcus and Enterococcus species isolated from bovine milk. J Food Prot 57:240–245
Jayarao BM, Oliver SP, Matthews KR, King SH (1991) Comparative evaluation of Vitek Gram-positive identification system and API Rapid Strep system for identification of Streptococcus species of bovine origin. Vet Micro 26:301–308
Jayarao BM, Gillespie BE, Oliver SP (1996) Application of randomly amplified polymorphic DNA fingerprinting for species identification of bacteria isolated from bovine milk. J Food Prot 59:615–620
Jayarao BM, Gillespie BE, Lewis MJ, Dowlen HH, Oliver SP (1999) Epidemiology of Streptococcus uberis intramammary infections in a dairy herd. J Vet Med, Series B 46:433–442
Kennedy BW, Sethar MS, Moxley JE, Downey BR (1982) Heritability of somatic cell count and its relationship with milk yield and composition in Holsteins. J Dairy Sci 65:843–847
Lang F, Shumilina E (2013) Regulation of ion channels by the serum- and glucocorticoid-inducible kinase SGK1. FASEB J 27:3–12
Lang F, Artunc F, Vallon V (2009) The physiological impact of the serum- and glucocorticoid-inducible kinase SGK1. Curr Opin Nephrol Hypertens 18:439–448
Lang F, Strutz-Seebohm N, Seebohm G, Lang UE (2010) Significance of SGK1 in the regulation of neuronal function. J Physiol 588:3349–3354
Lawless N, Reinhardt TA, Bryan K, Baker M, Pesch B, Zimmerman D et al (2014) MicroRNA regulation of bovine monocyte inflammatory and metabolic networks in an in vivo infection model. G3 (Bethesda) 4:957–971
Lee TK, Murthy SR, Cawley NX, Dhanvantari S, Hewitt SM, Lou H et al (2011) An N-terminal truncated carboxypeptidase E splice isoform induces tumor growth and is a biomarker for predicting future metastasis in human cancers. J Clin Invest 121:880–892
Manly KF (2005) Reliability of statistical associations between genes and disease. Immunogenetics 57:549–558
Matsusaka T, Fujikawa K, Nishio Y, Mukaida N, Matsushima K, Kishimoto T, Akira S (1993) Transcription factors NF-IL6 and NF-kappa B synergistically activate transcription of the inflammatory cytokines. interleukin 6 and interleukin 8 PNAS 90:10193–10197
Meredith BK, Kearney FJ, Finlay EK, Bradley DG, Fahey AG, Berry DP, Lynn DJ (2012) Genome-wide associations for milk production and somatic cell score in Holstein-Friesian cattle in Ireland. BMC Genet 13:1–11
Østerås O, Sølverød L, Reksen O (2006) Milk culture results in a large Norwegian survey—effects of season, parity, days in milk, resistance, and clustering. J Dairy Sci 89:1010–1023
Paape MJ, Bannerman DD, Zhao X, Lee J-W (2003) The bovine neutrophil: structure and function in blood and milk. Vet Res 34:597–627
Pérez-Cabal MA, Charfeddine N (2013) Genetic relationship between clinical mastitis and several traits of interest in Spanish Holstein dairy cattle. Interbull Bulletin 47:77–81
Petrovski KR, Williamson NB, Lopez-Villalobos N, Parkinson TJ, Tucker IG (2011) Culture results from milk samples submitted to veterinary diagnostic laboratories from August 2003 to December 2006 in New Zealand. N Z Vet J 59:317–322
Piepers S, De Meulemeester L, de Kruif A, Opsomer G, Barkema HW, De Vliegher S (2007) Prevalence and distribution of mastitis pathogens in subclinically infected dairy cows in Flanders, Belgium. J Dairy Res 74:478–483
Pighetti GM et al. (2017) Vaccination with recombinant Streptococcus uberis Adhesion Molecule alters immune response to experimental challenge. Int J Vet Dairy Sci. Published online: 05/15/2017
Pryce JE, Bolormaa S, Chamberlain AJ, Bowman PJ, Savin K, Goddard ME, Hayes BJ (2010) A validated genome-wide association study in 2 dairy cattle breeds for milk production and fertility traits using variable length haplotypes. J Dairy Sci 93:3331–3345
Purcell S, Cherny SS, Sham PC (2003) Genetic Power Calculator: design of linkage and association genetic mapping studies of complex traits. Bioinformatics 19(1):149–150
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MAR, Bender D, Maller J, Sklar P, de Bakker PIW, Daly MJ, Sham PC (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81:559–575
Pyörälä S (2008) Mastitis in post-partum dairy cows. Reprod Domest Anim 43:252–259
Raman D, Sai J, Neel NF, Chew CS, Richmond A (2010) LIM and SH3 protein-1 modulates CXCR2-mediated cell migration. PLoS One 5:e10050
Rambeaud M, Pighetti G (2007) Differential calcium signaling in dairy cows with specific CXCR1 genotypes potentially related to interleukin-8 receptor functionality. Immunogenetics 59:53–58
Rambeaud M, Almeida RA, Pighetti GM, Oliver SP (2003) Dynamics of leukocytes and cytokines during experimentally induced Streptococcus uberis mastitis. Vet Immunol Immunopathol 96:193–205
Rambeaud M, Clift R, Pighetti GM (2006) Association of a bovine CXCR2 gene polymorphism with neutrophil survival and killing ability. Vet Immunol Immunopathol 111:231–238
Richardson RM, Marjoram RJ, Barak LS, Snyderman R (2003) Role of the cytoplasmic tails of CXCR1 and CXCR2 in mediating leukocyte migration, activation, and regulation. J Immunol 170:2904–2911
Robinson JT, Thorvaldsdottir H, Winckler W, Guttman M, Lander ES, Getz G, Mesirov JP (2011) Integrative genomics viewer. Nat Biotechnol 29:24–26
Rooney C, White G, Nazgiewicz A, Woodcock SA, Anderson KI, Ballestrem C, Malliri A (2010) The Rac activator STEF (Tiam2) regulates cell migration by microtubule-mediated focal adhesion disassembly. EMBO Rep 11:292–298
Rupp R, Boichard D (2003) Genetics of resistance to mastitis in dairy cattle. Vet Res 34:671–688
Ryman VE, Packiriswamy N, Sordillo LM (2016) Apoptosis of endothelial cells by 13-HPODE contributes to impairment of endothelial barrier integrity. Med Inflammation 2016:13. https://doi.org/10.1155/2016/9867138
Schepers AJ, Lam TJ, Schukken YH, Wilmink JB, Hanekamp WJ (1997) Estimation of variance components for somatic cell counts to determine thresholds for uninfected quarters. J Dairy Sci 80:1833–1840
Schukken YH, Günther J, Fitzpatrick J, Fontaine MC, Goetze L, Holst O, Leigh J, Petzl W, Schuberth HJ, Sipka A, Smith DGE, Quesnell R, Watts J, Yancey R, Zerbe H, Gurjar A, Zadoks RN, Seyfert HM (2011) Host-response patterns of intramammary infections in dairy cows. Vet Immunol Immunopathol 144:270–289
Seipel K, O’Brien SP, Iannotti E, Medley QG, Streuli M (2001) Tara, a novel F-actin binding protein, associates with the Trio guanine nucleotide exchange factor and regulates actin cytoskeletal organization. J Cell Sci 114:389–399
Shi Y (2017) The spliceosome: a protein-directed metalloribozyme. J Mol Biol 429:2640–2653
Shima Y, Kawaguchi SY, Kosaka K, Nakayama M, Hoshino M, Nabeshima Y, Hirano T, Uemura T (2007) Opposing roles in neurite growth control by two seven-pass transmembrane cadherins. Nat Neurosci 10:963–969
Sladek Z, Rysanek D, Ryznarova H, Faldyna M (2006) The role of neutrophil apoptosis during experimentally induced Streptococcus uberis mastitis. Vet Med(Praha) 51:437–447
Slama P, Sladek Z, Rysanek D, Langrova T (2009) Effect of Staphylococcus aureus and Streptococcus uberis on apoptosis of bovine mammary gland lymphocytes. Res Vet Sci 87:233–238
Spencer CCA, Su Z, Donnelly P, Marchini J (2009) Designing genome-wide association studies: sample size, power, imputation, and the choice of genotyping chip. PLoS Genet 5:e1000477
Spittler A et al (1999) Immunomodulatory effects of glycine on LPS-treated monocytes: reduced TNF-α production and accelerated IL-10 expression. FASEB J 13:563–571
Steemers FJ, Chang W, Lee G, Barker DL, Shen R, Gunderson KL (2006) Whole-genome genotyping with the single-base extension assay. Nat Methods 3:31–33
Sternberg EM (2006) Neural regulation of innate immunity: a coordinated nonspecific host response to pathogens. Nat Rev Immunol 6:318–328
Tassi R, McNeilly TN, Fitzpatrick JL, Fontaine MC, Reddick D, Ramage C et al (2013) Strain-specific pathogenicity of putative host-adapted and nonadapted strains of Streptococcus uberis in dairy cattle. J Dairy Sci 96(8):5129–5145
Van den Eynden J, Ali SS, Horwood N, Carmans S, Brone B, Hellings N et al (2009) Glycine and glycine receptor signalling in non-neuronal cells. Front Mol Neurosci 2:9
van Rijssel J, van Buul JD (2012) The many faces of the guanine-nucelotide exchange factor trio. Cell Adhes Migr 6:482–487
Verbeke J, Piepers S, Supré K, De Vliegher S (2014) Pathogen-specific incidence rate of clinical mastitis in Flemish dairy herds, severity, and association with herd hygiene. J Dairy Sci 97:6926–6934
Wedlock DN, Buddle BM, Williamson J, Lacy-Hulbert SJ, Turner SA, Subharat S, Heiser A (2014) Dairy cows produce cytokine and cytotoxic T cell responses following vaccination with an antigenic fraction from Streptococcus uberis. Vet Immunol Immunopathol 160(1–2):51–60
Weller JI, Saran A, Zeliger Y (1992) Genetic and environmental relationships among somatic cell count, bacterial infection, and clinical mastitis. J Dairy Sci 75:2532–2540
Wheeler M, Stachlewitz RF, Yamashina S, Ikejima K, Morrow AL, Thurman RG (2000) Glycine-gated chloride channels in neutrophils attenuate calcium influx and superoxide production. FASEB J 14:476–484
Wiggans GR, Shook GE (1987) A lactation measure of somatic cell count. J Dairy Sci 70:2666–2672
Wilson DJ, Gonzalez RN, Das HH (1997) Bovine mastitis pathogens in New York and Pennsylvania: prevalence and effects on somatic cell count and milk production. J Dairy Sci 80:2592–2598
Wu C, Yosef N, Thalhamer T, Zhu C, Xiao S, Kishi Y, Regev A, Kuchroo V (2013) Induction of pathogenic Th17 cells by inducible salt sensing kinase SGK1. Nature 496(7446):513–517
Yoshida K, Maekawa T, Zhu Y, Renard-Guillet C, Chatton B, Inoue K, Uchiyama T, Ishibashi KI, Yamada T, Ohno N, Shirahige K, Okada-Hatakeyama M, Ishii S (2015) The transcription factor ATF7 mediates lipopolysaccharide-induced epigenetic changes in macrophages involved in innate immunological memory. Nat Immunol 16:1034–1043
Zadoks RN (2007) Sources and epidemiology of Streptococcus uberis, with special emphasis on mastitis in dairy cattle. Perspectives in Agriculture, Veterinary Science, Nutrition and Natural Resources 2:15
Acknowledgements
The authors would like to thank Dr. Arnold Saxton of the department of Animal Science at the University of Tennessee for his assistance in converting the data format. A special thank you to the staff at the East Tennessee Research and Education Center Little River Unit for their aid in animal care and milking.
Funding
The challenge portion of this study was supported by USDA-NIFA-AFRI (2011-67015-30168). The remaining portions of the study were supported by UTAgResearch and the AgInnovation fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Siebert, L., Staton, M.E., Headrick, S. et al. Genome-wide association study identifies loci associated with milk leukocyte phenotypes following experimental challenge with Streptococcus uberis. Immunogenetics 70, 553–562 (2018). https://doi.org/10.1007/s00251-018-1065-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-018-1065-3