Abstract
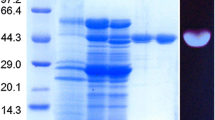
Most reported microbial β-1,3-1,4-glucanases belong to the glycoside hydrolase family 16. Here, we report a new acidic family 7 endo-β-1,3-1,4-glucanase (Bgl7A) from the acidophilic fungus Bispora sp. MEY-1. The cDNA of Bgl7A was isolated and over-expressed in Pichia pastoris, with a yield of about 1,000 U ml–1 in a 3.7-l fermentor. The purified recombinant Bgl7A had three activity peaks at pH 1.5, 3.5, and 5.0 (maximum), respectively, and a temperature optimum at 60°C. The enzyme was stable at pH 1.0–8.0 and highly resistant to both pepsin and trypsin. Belonging to the group of non-specific endoglucanase, Bgl7A can hydrolyze not only β-glucan and cellulose but also laminarin and oat spelt xylan. The specific activity of Bgl7A against barley β-glucan and lichenan (4,040 and 2,740 U mg–1) was higher than toward carboxymethyl cellulose sodium (395 U mg–1), which was different from other family 7 endo-β-glucanases.




Similar content being viewed by others
References
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modeling. Bioinformatics 22:195–201
Beckmann L, Simon O, Vahjen W (2006) Isolation and identification of mixed linked β-glucan degrading bacteria in the intestine of broiler chickens and partial characterization of respective 1, 3-1, 4-β-glucanase activities. J Basic Microbiol 46:175–185
Boyce A, Walsh G (2007) Production, purification and application-relevant characterization of an endo-1, 3(4)-β-glucanase from Rhizomucor miehei. Appl Microbiol Biotechnol 76:835–841
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Buliga GS, Brant DA, Fincher GB (1986) The sequence statistics and solution conformation of a barley (1, 3-1, 4)-β-D-glucan. Carbohydr Res 157:139–156
Celestino KRS, Cunha RB, Felix CR (2006) Characterization of a β-glucanase produced by Rhizopus microsporus var. microsporus, and its potential for application in the brewing industry. BMC Biochem 7:23
Chen H, Li XL, Ljungdahl LG (1997) Sequencing of a 1, 3-1, 4-β-D-glucanase (lichenase) from the anaerobic fungus Orpinomyces strain PC-2: properties of the enzyme expressed in Escherichia coli and evidence that the gene has a bacterial origin. J Bacteriol 179:6028–6034
Claeyssens M, van Tilbeurgh H, Kamerling JP, Berg J, Vrsanska M, Biely P (1990) Studies of the cellulolytic system of the filamentous fungus Trichoderma reesei QM 9414. Biochem J 270:251–256
Ekinci MS, McCrae SI, Flint HJ (1997) Isolation and overexpression of a gene encoding an extracellular β-(1, 3-1, 4)-glucanase from Streptococcus bovis JB1. Appl Environ Microbiol 63:3752–3756
Feng Y, Duan CJ, Pang H, Mo XC, Wu CF, Yu Y, Hu YL, Wei J, Tang JL, Feng JX (2007) Cloning and identification of novel cellulase genes from uncultured microorganisms in rabbit cecum and characterization of the expressed cellulases. Appl Microbiol Biotechnol 75:319–328
Görlach JM, van der Knaap E, Walton JD (1998) Cloning and targeted disruption of MLG1, a gene encoding two of three extracellular mixed-linked glucanases of Cochliobolus carbonum. Appl Environ Microbiol 64:385–391
Grishutin SG, Gusakov AV, Dzedzyulya EI, Sinitsyn AP (2006) A lichenase-like family 12 endo-(1, 4)-β-glucanase from Aspergillus japonicus: study of the substrate specificity and mode of action on β-glucans in comparison with other glycoside hydrolases. Carbohydr Res 341:218–229
Hasper AA, Dekkers E, van Mil M, van de Vondervoort PJI, de Graaff LH (2002) EglC, a new endoglucanase from Aspergillus niger with major activity towards xyloglucan. Appl Environ Microbiol 68:1556–1560
Hrmova M, Fincher GB (2001) Plant enzyme structure. Explaining substrate specificity and the evolution of function. Plant Physiol 125:54–57
Huang HQ, Yang PL, Luo HY, Tang HG, Shao N, Yuan TZ, Wang YR, Bai YG, Yao B (2008) High-level expression of a truncated 1, 3-1, 4-β-D-glucanase from Fibrobacter succinogenes in Pichia pastoris by optimization of codons and fermentation. Appl Microbiol Biotechnol 78:95–103
Jeanmougin F, Thompson JD, Gouy M, Higgins DG, Gibson TJ (1998) Multiple sequence alignment with Clustal X. Trends Biochem Sci 23:403–405
Kitamoto N, Go M, Shibayama T, Kimura T, Kito Y, Ohmiya K, Tsukagoshi N (1996) Molecular cloning, purification and characterization of two endo-1, 4-β-glucanases from Aspergillus oryzae KBN616. Appl Microbiol Biotechnol 46:538–544
Kleywegt GJ, Zou JY, Divne C, Davies GJ, Sinning I, Stahlberg J, Reinikainen T, Srisodsuk M, Teeri TT, Jones TA (1997) The crystal structure of the catalytic core domain of endoglucanase I from Trichoderma reesei at 3.6 A resolution, and a comparison with related enzymes. J Mol Biol 272:383–397
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Liu YG, Whittier RF (1995) Thermal asymmetric interlaced PCR: automatable amplification and sequencing of insert end fragments from P1 and YAC clones for chromosome walking. Genomics 25:674–681
Luo HY, Wang YR, Wang H, Yang J, Yang YH, Huang HQ, Yang PL, Bai YG, Shi PJ, Fan YL, Yao B (2009a) A novel highly acidic β-mannanase from the acidophilic fungus Bispora sp. MEY-1: gene cloning and overexpression in Pichia pastoris. Appl Microbiol Biotechnol 82:453–461
Luo HY, Wang YR, Li J, Wang H, Yang J, Yang YH, Huang HQ, Fan YL, Yao B (2009b) Cloning, expression and characterization of a novel acidic xylanase, XYL11B, from the acidophilic fungus Bispora sp. MEY-1. Enzyme Microb Technol 45:126–133
Mathlouthi N, Mallet S, Saulnier L, Quemener B, Larbier M (2002) Effects of xylanase and β-glucanase addition on performance, nutrient digestibility, and physico-chemical conditions in the small intestine contents and faecal microflora of broiler chickens fed a wheat and barley-based diet. Anim Res 51:395–406
McCarthy T, Hanniffy O, Savage A, Tuohy MG (2003) Catalytic properties and mode of action of three endo-β-glucanases from Talaromyces emersonii on soluble β-1, 4-and β-1, 3, 1, 4-linked glucans. Int J Biol Macromol 33:141–148
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Murray PG, Grassick A, Laffey CD, Cuffe MM, Higgins T, Savage AV, Planas A, Tuohy MG (2001) Isolation and characterization of a thermostable endo-β-glucanase active on 1, 3-1, 4-β-D-glucans from the aerobic fungus Talaromyces emersonii CBS 814.70. Enzyme Microb Technol 29:90–98
Nakazawa H, Okada K, Kobayashi R, Kubota T, Onodera T, Ochiai N, Omata N, Ogasawara W, Okada H, Morikawa Y (2008) Characterization of the catalytic domains of Trichoderma reesei endoglucanase I, II, and III, expressed in Escherichia coli. Appl Microbiol Biotechnol 81:681–689
Oh BC, Choi WC, Park S, Kim YO, Oh TK (2004) Biochemical properties and substrate specificities of alkaline and histidine acid phytases. Appl Microbiol Biotechnol 63:362–372
Planas A (2000) Bacterial 1, 3-1, 4-β-glucanases: structure, function and protein engineering. Biochem Biophys Acta 1543:361–382
Promdonkoy P, Tang K, Sornlake W, Harnpicharnchai P, Kobayashi RS, Ruanglek V, Upathanpreecha T, Vesaratchavest M, Eurwilaichitr L, Tanapongpipat S (2008) Expression and characterization of Aspergillus thermostable phytases in Pichia pastoris. FEMS Microbiol Lett 290:18–24
Schou C, Rasmussen G, Kaltoft MB, Henrissat B, Schülein M (1993) Stereochemistry, specificity and kinetics of the hydrolysis of reduced cellodextrins by nine cellulases. Eur J Biochem 217:947–953
Teng D, Wang J, Fan Y, Yang Y, Tian Z, Luo J, Yang G, Zhang F (2006) Cloning of β-1, 3-1, 4-glucanase gene from Bacillus licheniformis EGW039 (CGMCC 0635) and its expression in Escherichia coli BL21 (DE3). Appl Microbiol Biotechnol 72:705–712
Yang PL, Shi PJ, Wang YR, Bai YG, Meng K, Luo HY, Yuan TZ, Yao B (2007) Cloning and overexpression of a Paenibacillus β-glucanase in Pichia pastoris: purification and characterization of the recombinant enzyme. J Microbiol Biotechnol 17:58–66
Yang S, Yan Q, Jiang Z, Fan G, Wang L (2008) Biochemical characterization of a novel thermostable β-1, 3-1, 4-glucanase (lichenase) from Paecilomyces thermophila. J Agric Food Chem 56:5345–5351
Acknowledgments
This research was supported by the National High Technology Research and Development Program of China (863 program, grant no. 2007AA100601), Chinese Program on Research for Public Good (grant no. 2005DIB4J038), and 948 program of the Ministry of Agriculture (grant no. 2007-Z3).
Author information
Authors and Affiliations
Corresponding author
Additional information
Huiying Luo and Jun Yang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Luo, H., Yang, J., Yang, P. et al. Gene cloning and expression of a new acidic family 7 endo-β-1,3-1,4-glucanase from the acidophilic fungus Bispora sp. MEY-1. Appl Microbiol Biotechnol 85, 1015–1023 (2010). https://doi.org/10.1007/s00253-009-2119-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2119-0