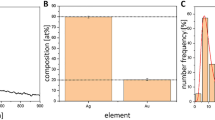
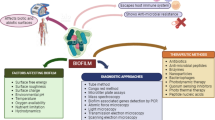
Abstract
The formation of biofilms by bacterial pathogens and the presence of persister cells in biofilms have become major concerns in the health sector, owing to their antibiotic resistance and tolerance. The transformation of bacterial pathogens into persister cells, either stochastically or due to stressful environmental factors, results in recalcitrant and recurring infections. Here, we sought to prepare gold nanoparticles from naturally occurring caffeine and explore their inhibitory action against biofilm formation and persister cells. Fourier transform infrared spectroscopy, UV-visible absorption spectroscopy, field emission transmission electron microscopy, energy-dispersive X-ray diffraction, and dynamic light scattering were used to characterize the gold nanoparticles obtained from caffeine (Caff-AuNPs). The Caff-AuNPs were found to exhibit a number of properties, including the ability to prevent biofilm formation, disperse mature biofilms, and kill different types of persister of gram-positive (Staphylococcus aureus and Listeria monocytogenes) and gram-negative (Pseudomonas aeruginosa and Escherichia coli) pathogenic bacteria. Microscopic analysis of the aforementioned bacterial cells, treated with Caff-AuNPs, revealed the bactericidal effect of Caff-AuNPs, although the underlying mechanism remains unknown. Collectively, the Caff-AuNPs synthesized in this study may be used as potential drugs to combat chronic infections caused by biofilm-forming pathogenic bacteria.
Key points
• Biofilm and persister cells are clinically relevant, as they either prolong or completely resist antibiotic treatments.
• Caffeine is used in the green synthesis of Caff-AuNPs, which have antibacterial and antibiofilm properties.
• Caff-AuNPs are effective against various pathogenic bacterial persister cells.







Similar content being viewed by others
References
Azam MW, Khan AU (2019) Updates on the pathogenicity status of Pseudomonas aeruginosa. Drug Discov Today 24(1):350–359. https://doi.org/10.1016/j.drudis.2018.07.003
Banerjee D, Shivapriya PM, Gautam PK, Misra K, Sahoo AK, Samanta SK (2020) A review on basic biology of bacterial biofilm infections and their treatments by nanotechnology-based approaches. Proc Natl Acad Sci India Sect B Biol Sci 90(2):243–259. https://doi.org/10.1007/s40011-018-01065-7
Bernier SP, Surette MG (2013) Concentration-dependent activity of antibiotics in natural environments. Front Microbiol 4:20. https://doi.org/10.3389/fmicb.2013.00020
Breijyeh Z, Jubeh B, Karaman R (2020) Resistance of gram-negative bacteria to current antibacterial agents and approaches to resolve it. Molecules 25(6). https://doi.org/10.3390/molecules25061340
Butt S, Hasan SMF, Hassan MM, Alkharfy KM, Neau SH (2019) Directly compressed rosuvastatin calcium tablets that offer hydrotropic and micellar solubilization for improved dissolution rate and extent of drug release. Saudi Pharm J 27(5):619–628. https://doi.org/10.1016/j.jsps.2019.03.002
Cohen NR, Lobritz MA, Collins JJ (2013) Microbial persistence and the road to drug resistance. Cell Host Microbe 13(6):632–642. https://doi.org/10.1016/j.chom.2013.05.009
Cui Y, Zhao Y, Tian Y, Zhang W, Lü X, Jiang X (2012) The molecular mechanism of action of bactericidal gold nanoparticles on Escherichia coli. Biomaterials 33(7):2327–2333. https://doi.org/10.1016/j.biomaterials.2011.11.057
de la Fuente-Núñez C, Korolik V, Bains M, Nguyen U, Breidenstein EB, Horsman S, Lewenza S, Burrows L, Hancock RE (2012) Inhibition of bacterial biofilm formation and swarming motility by a small synthetic cationic peptide. Antimicrob Agents Chemother 56(5):2696–2704. https://doi.org/10.1128/aac.00064-12
Elbehiry A, Al-Dubaib M, Marzouk E, Moussa I (2019) Antibacterial effects and resistance induction of silver and gold nanoparticles against Staphylococcus aureus-induced mastitis and the potential toxicity in rats. Microbiologyopen 8(4):e00698–e00698. https://doi.org/10.1002/mbo3.698
Exner M, Bhattacharya S, Christiansen B, Gebel J, Goroncy-Bermes P, Hartemann P, Heeg P, Ilschner C, Kramer A, Larson E, Merkens W, Mielke M, Oltmanns P, Ross B, Rotter M, Schmithausen RM, Sonntag HG, Trautmann M (2017) Antibiotic resistance: what is so special about multidrug-resistant Gram-negative bacteria? GMS Hyg Infect Control 12:Doc05. https://doi.org/10.3205/dgkh000290
Fisher RA, Gollan B, Helaine S (2017) Persistent bacterial infections and persister cells. Nat Rev Microbiol 15(8):453–464. https://doi.org/10.1038/nrmicro.2017.42
Girish VM, Liang H, Aguilan JT, Nosanchuk JD, Friedman JM, Nacharaju P (2019) Anti-biofilm activity of garlic extract loaded nanoparticles. Nanomedicine 20:102009. https://doi.org/10.1016/j.nano.2019.04.012
Grassi L, Di Luca M, Maisetta G, Rinaldi AC, Esin S, Trampuz A, Batoni G (2017) Generation of persister cells of Pseudomonas aeruginosa and Staphylococcus aureus by chemical treatment and evaluation of their susceptibility to membrane-targeting agents. Front Microbiol 8:1917. https://doi.org/10.3389/fmicb.2017.01917
Hall CW, Mah T-F (2017) Molecular mechanisms of biofilm-based antibiotic resistance and tolerance in pathogenic bacteria. FEMS Microbiol Rev 41(3):276–301. https://doi.org/10.1093/femsre/fux010
Han C, Romero N, Fischer S, Dookran J, Berger A, Doiron AL (2017) Recent developments in the use of nanoparticles for treatment of biofilms. Nanotechnol Rev 6(5):383–404. https://doi.org/10.1515/ntrev-2016-0054
Harms A, Maisonneuve E, Gerdes K (2016) Mechanisms of bacterial persistence during stress and antibiotic exposure. Science 354(6318):aaf4268. https://doi.org/10.1126/science.aaf4268
Huang X, El-Sayed MA (2010) Gold nanoparticles: optical properties and implementations in cancer diagnosis and photothermal therapy. J Adv Res 1(1):13–28. https://doi.org/10.1016/j.jare.2010.02.002
Kaldalu N, Hauryliuk V, Tenson T (2016) Persisters-as elusive as ever. Appl Microbiol Biotechnol 100(15):6545–6553. https://doi.org/10.1007/s00253-016-7648-8
Kamalakannan R, Mani G, Muthusamy P, Susaimanickam AA, Kim K (2017) Caffeine-loaded gold nanoparticles conjugated with PLA-PEG-PLA copolymer for in vitro cytotoxicity and anti-inflammatory activity. J Ind Eng Chem 51:113–121. https://doi.org/10.1016/j.jiec.2017.02.021
Kamaruzzaman NF, Tan LP, Hamdan RH, Choong SS, Wong WK, Gibson AJ, Chivu A, Pina MF (2019) Antimicrobial polymers: the potential replacement of existing antibiotics? Int J Mol Sci 20(11):2747. https://doi.org/10.3390/ijms20112747
Kaper JB, Nataro JP, Mobley HLT (2004) Pathogenic Escherichia coli. Nat Rev Microbiol 2(2):123–140. https://doi.org/10.1038/nrmicro818
Keijok WJ, Pereira RHA, Alvarez LAC, Prado AR, da Silva AR, Ribeiro J, de Oliveira JP, Guimarães MCC (2019) Controlled biosynthesis of gold nanoparticles with Coffea arabica using factorial design. Sci Rep 9(1):16019. https://doi.org/10.1038/s41598-019-52496-9
Khan F, Khan MM, Kim YM (2018) Recent progress and future perspectives of antibiofilm drugs immobilized on nanomaterials. Curr Pharm Biotechnol 19(8):631–643. https://doi.org/10.2174/1389201019666180828090052
Khan F, Lee JW, Manivasagan P, Pham DTN, Oh J, Kim YM (2019a) Synthesis and characterization of chitosan oligosaccharide-capped gold nanoparticles as an effective antibiofilm drug against the Pseudomonas aeruginosa PAO1. Microb Pathog 135:103623. https://doi.org/10.1016/j.micpath.2019.103623
Khan F, Manivasagan P, Lee JW, Pham DTN, Oh J, Kim YM (2019b) Fucoidan-stabilized gold nanoparticle-mediated biofilm inhibition, attenuation of virulence and motility properties in Pseudomonas aeruginosa PAO1. Mar Drugs 17(4). https://doi.org/10.3390/md17040208
Khan F, Lee JW, Pham DTN, Lee JH, Kim HW, Kim YK, Kim YM (2020a) Streptomycin mediated biofilm inhibition and suppression of virulence properties in Pseudomonas aeruginosa PAO1. Appl Microbiol Biotechnol 104(2):799–816. https://doi.org/10.1007/s00253-019-10190-w
Khan F, Pham DTN, Oloketuyi SF, Manivasagan P, Oh J, Kim YM (2020b) Chitosan and their derivatives: antibiofilm drugs against pathogenic bacteria. Colloids Surf B: Biointerfaces 185:110627. https://doi.org/10.1016/j.colsurfb.2019.110627
Khan F, Pham DTN, Tabassum N, Oloketuyi SF, Kim Y-M (2020c) Treatment strategies targeting persister cell formation in bacterial pathogens. Crit Rev Microbiol 46:1–24. https://doi.org/10.1080/1040841X.2020.1822278
Khan F, Yu H, Kim YM (2020d) Bactericidal activity of usnic acid-chitosan nanoparticles against persister cells of biofilm-forming pathogenic bacteria. Mar Drugs 18(5). https://doi.org/10.3390/md18050270
Kobayashi SD, Malachowa N, DeLeo FR (2015) Pathogenesis of Staphylococcus aureus abscesses. Am J Pathol 185(6):1518–1527. https://doi.org/10.1016/j.ajpath.2014.11.030
Kolb H, Kempf K, Martin S (2020) Health effects of coffee: mechanism unraveled? Nutrients 12(6):1842
Krishnamurthy S, Esterle A, Sharma NC, Sahi SV (2014) Yucca-derived synthesis of gold nanomaterial and their catalytic potential. Nanoscale Res Lett 9(1):627. https://doi.org/10.1186/1556-276X-9-627
Kwan BW, Valenta JA, Benedik MJ, Wood TK (2013) Arrested protein synthesis increases persister-like cell formation. Antimicrob Agents Chemother 57(3):1468–1473. https://doi.org/10.1128/aac.02135-12
Lee H, Lee DG (2018) Gold nanoparticles induce a reactive oxygen species-independent apoptotic pathway in Escherichia coli. Colloids Surf B: Biointerfaces 167:1–7. https://doi.org/10.1016/j.colsurfb.2018.03.049
Lee JH, Kim YG, Gwon G, Wood TK, Lee J (2016) Halogenated indoles eradicate bacterial persister cells and biofilms. AMB Express 6(1):123. https://doi.org/10.1186/s13568-016-0297-6
Lewis K (2001) Riddle of biofilm resistance. Antimicrob Agents Chemother 45(4):999–1007. https://doi.org/10.1128/aac.45.4.999-1007.2001
Lewis K (2007) Persister cells, dormancy and infectious disease. Nat Rev Microbiol 5(1):48–56. https://doi.org/10.1038/nrmicro1557
Lewis K (2008) Multidrug tolerance of biofilms and persister cells. Curr Top Microbiol Immunol 322:107–131. https://doi.org/10.1007/978-3-540-75418-3_6
Lewis K (2010) Persister cells. Annu Rev Microbiol 64(1):357–372. https://doi.org/10.1146/annurev.micro.112408.134306
Mah TF (2012) Biofilm-specific antibiotic resistance. Future Microbiol 7(9):1061–1072. https://doi.org/10.2217/fmb.12.76
Maisonneuve E, Gerdes K (2014) Molecular mechanisms underlying bacterial persisters. Cell 157(3):539–548. https://doi.org/10.1016/j.cell.2014.02.050
Manivasagan P, Khan F, Hoang G, Mondal S, Kim H, Hoang Minh Doan V, Kim YM, Oh J (2019) Thiol chitosan-wrapped gold nanoshells for near-infrared laser-induced photothermal destruction of antibiotic-resistant bacteria. Carbohydr Polym 225:115228. https://doi.org/10.1016/j.carbpol.2019.115228
Medina E, Pieper DH (2016) Tackling threats and future problems of multidrug-resistant bacteria. Curr Top Microbiol Immunol 398:3–33. https://doi.org/10.1007/82_2016_492
Mlynarcik P, Kolar M (2017) Starvation- and antibiotics-induced formation of persister cells in Pseudomonas aeruginosa. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 161(1):58–67. https://doi.org/10.5507/bp.2016.057
Mudshinge SR, Deore AB, Patil S, Bhalgat CM (2011) Nanoparticles: emerging carriers for drug delivery. Saudi Pharm J 19(3):129–141. https://doi.org/10.1016/j.jsps.2011.04.001
Munita JM, Arias CA (2016) Mechanisms of antibiotic resistance. Microbiol Spectr 4(2). https://doi.org/10.1128/microbiolspec.VMBF-0016-2015
Nidadavolu P, Amor W, Tran PL, Dertien J, Colmer-Hamood JA, Hamood AN (2012) Garlic ointment inhibits biofilm formation by bacterial pathogens from burn wounds. J Med Microbiol 61(Pt 5):662–671. https://doi.org/10.1099/jmm.0.038638-0
Paradkar MM, Irudayaraj J (2002) Rapid determination of caffeine content in soft drinks using FTIR–ATR spectroscopy. Food Chem 78(2):261–266. https://doi.org/10.1016/S0308-8146(02)00116-4
Patra JK, Das G, Fraceto LF, Campos EVR, Rodriguez-Torres MP, Acosta-Torres LS, Diaz-Torres LA, Grillo R, Swamy MK, Sharma S, Habtemariam S, Shin H-S (2018) Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnol 16(1):71. https://doi.org/10.1186/s12951-018-0392-8
Perveen K, Husain FM, Qais FA, Khan A, Razak S, Afsar T, Alam P, Almajwal AM, Abulmeaty MMA (2021) Microwave-assisted rapid green synthesis of gold nanoparticles using seed extract of Trachyspermum ammi: ROS mediated biofilm inhibition and anticancer activity. Biomolecules 11(2). https://doi.org/10.3390/biom11020197
Rajkumari J, Meena H, Gangatharan M, Busi S (2017) Green synthesis of anisotropic gold nanoparticles using hordenine and their antibiofilm efficacy against Pseudomonas aeruginosa. IET Nanobiotechnol 11(8):987–994. https://doi.org/10.1049/iet-nbt.2017.0069
Ramasamy M, Lee JH, Lee J (2017) Direct one-pot synthesis of cinnamaldehyde immobilized on gold nanoparticles and their antibiofilm properties. Colloids Surf B: Biointerfaces 160:639–648. https://doi.org/10.1016/j.colsurfb.2017.10.018
Shamaila S, Zafar N, Riaz S, Sharif R, Nazir J, Naseem S (2016) Gold Nanoparticles: an efficient antimicrobial agent against enteric bacterial human pathogen. Nanomaterials (Basel) 6(4):71. https://doi.org/10.3390/nano6040071
Shen Q, Zhou W, Hu L, Qi Y, Ning H, Chen J, Mo H (2017) Bactericidal activity of alpha-bromocinnamaldehyde against persisters in Escherichia coli. PLoS ONE 12(7):e0182122. https://doi.org/10.1371/journal.pone.0182122
Singh R, Lillard JW Jr (2009) Nanoparticle-based targeted drug delivery. Exp Mol Pathol 86(3):215–223. https://doi.org/10.1016/j.yexmp.2008.12.004
Singh M, Kalaivani R, Manikandan S, Sangeetha N, Kumaraguru AK (2013) Facile green synthesis of variable metallic gold nanoparticle using Padina gymnospora, a brown marine macroalga. Appl Nanosci 3(2):145–151. https://doi.org/10.1007/s13204-012-0115-7
Singh P, Pandit S, Beshay M, Mokkapati VRSS, Garnaes J, Olsson ME, Sultan A, Mackevica A, Mateiu RV, Lütken H, Daugaard AE, Baun A, Mijakovic I (2018) Anti-biofilm effects of gold and silver nanoparticles synthesized by the Rhodiola rosea rhizome extracts. Artif Cell, Nanomed B 46(sup3):S886–S899. https://doi.org/10.1080/21691401.2018.1518909
Slavin YN, Asnis J, Häfeli UO, Bach H (2017) Metal nanoparticles: understanding the mechanisms behind antibacterial activity. J Nanobiotechnol 15(1):65. https://doi.org/10.1186/s12951-017-0308-z
Swartz MN (2002) Human diseases caused by foodborne pathogens of animal origin. Clin Infect Dis 34(Supplement_3):S111–S122. https://doi.org/10.1086/340248
Vázquez-Boland JA, Kuhn M, Berche P, Chakraborty T, Domínguez-Bernal G, Goebel W, González-Zorn B, Wehland J, Kreft J (2001) Listeria pathogenesis and molecular virulence determinants. Clin Microbiol Rev 14(3):584–640. https://doi.org/10.1128/cmr.14.3.584-640.2001
Vestby LK, Grønseth T, Simm R, Nesse LL (2020) Bacterial biofilm and its role in the pathogenesis of disease. Antibiotics (Basel) 9(2). https://doi.org/10.3390/antibiotics9020059
Vouga M, Greub G (2016) Emerging bacterial pathogens: the past and beyond. Clin Microbiol Infect 22(1):12–21. https://doi.org/10.1016/j.cmi.2015.10.010
Wood TK, Knabel SJ, Kwan BW (2013) Bacterial persister cell formation and dormancy. Appl Environ Microbiol 79(23):7116–7121. https://doi.org/10.1128/aem.02636-13
Funding
This work was supported by Basic Science Research Program through the National Research Foundation of Korea funded by the Ministry of Education (NRF-2019R1A2C1087156).
Author information
Authors and Affiliations
Contributions
FK and YMK conceived the idea of the present study, designed the experiment, analyzed the data, and wrote the paper. The experiment was carried out by FK, SKP, NIB, and DKO.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competeing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 1.09 mb)
Rights and permissions
About this article
Cite this article
Khan, F., Park, SK., Bamunuarachchi, N.I. et al. Caffeine-loaded gold nanoparticles: antibiofilm and anti-persister activities against pathogenic bacteria. Appl Microbiol Biotechnol 105, 3717–3731 (2021). https://doi.org/10.1007/s00253-021-11300-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11300-3