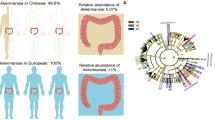
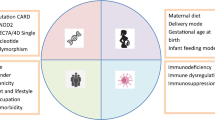
Abstract
Akkermansia muciniphila is a next-generation probiotic with significant application prospects. The role of A. muciniphila in metabolic diseases and tumor immunotherapy has been widely recognized. Recent clinical trials further confirmed its safety and therapeutic value in human metabolic diseases. A. muciniphila also shows potential in the treatment of intestinal inflammatory diseases, especially for inflammatory bowel disease (IBD). The improvement in the efficacy of washed microbiota transplantation (WMT) in treating IBD is closely related to the increase in the abundance of A. muciniphila in patients’ gut. However, there is still controversy regarding the pro-inflammatory or anti-inflammatory effect of A. muciniphila on IBD. Currently, several studies targeting the correlation between A. muciniphila and IBD have demonstrated opposite conclusions. Similarly, the interventional studies exploring causality between them also come to conflicting results. This article therefore aims to review the relationship between A. muciniphila and IBD, the effect of intervention of A. muciniphila on IBD, and the possible reasons for the contradictory role of A. muciniphila in the treatment of IBD.
Key points
-
The effect of A. muciniphila on inflammatory bowel disease is controversy.
-
A. muciniphila shows anti-inflammatory potential in IBD.
-
The colitogenicity of A. muciniphila is context dependent.
Similar content being viewed by others
References
Alvarado DM, Chen B, Iticovici M, Thaker AI, Dai N, VanDussen KL, Shaikh N, Lim CK, Guillemin GJ, Tarr PI, Ciorba MA (2019) Epithelial indoleamine 2,3-dioxygenase 1 modulates aryl hydrocarbon receptor and notch signaling to increase differentiation of secretory cells and alter mucus-associated microbiota. Gastroenterology 157(4):1093–1108.e11. https://doi.org/10.1053/j.gastro.2019.07.013
Ashrafian F, Behrouzi A, Shahriary A, Ahmadi Badi S, Davari M, Khatami S, Rahimi Jamnani F, Fateh A, Vaziri F, Siadat SD (2019) Comparative study of effect of Akkermansia muciniphila and its extracellular vesicles on toll-like receptors and tight junction. Gastroenterol Hepatol Bed Bench 12(2):163–168
Ayres JS, Trinidad NJ, Vance RE (2012) Lethal inflammasome activation by a multidrug-resistant pathobiont upon antibiotic disruption of the microbiota. Nat Med 18(5):799–806. https://doi.org/10.1038/nm.2729
Bajer L, Kverka M, Kostovcik M, Macinga P, Dvorak J, Stehlikova Z, Brezina J, Wohl P, Spicak J, Drastich P (2017) Distinct gut microbiota profiles in patients with primary sclerosing cholangitis and ulcerative colitis. World J Gastroenterol 23(25):4548–4558. https://doi.org/10.3748/wjg.v23.i25.4548
Becker N, Kunath J, Loh G, Blaut M (2011) Human intestinal microbiota: characterization of a simplified and stable gnotobiotic rat model. Gut Microbes 2(1):25–33. https://doi.org/10.4161/gmic.2.1.14651
Berclaz PY, Shibata Y, Whitsett JA, Trapnell BC (2002) GM-CSF, via PU.1, regulates alveolar macrophage Fcgamma R-mediated phagocytosis and the IL-18/IFN-gamma -mediated molecular connection between innate and adaptive immunity in the lung. Blood 100(12):4193–4200. https://doi.org/10.1182/blood-2002-04-1102
Bian X, Wu W, Yang L, Lv L, Wang Q, Li Y, Ye J, Fang D, Wu J, Jiang X, Shi D, Li L (2019) Administration of Akkermansia muciniphila ameliorates dextran sulfate sodium-induced ulcerative colitis in mice. Front Microbiol 10:2259. https://doi.org/10.3389/fmicb.2019.02259
Cani PD, de Vos WM (2017) Next-generation beneficial microbes: the case of Akkermansia muciniphila. Front Microbiol 8:1765. https://doi.org/10.3389/fmicb.2017.01765
Chen H, Xia Y, Zhu S, Yang J, Yao J, Di J, Liang Y, Gao R, Wu W, Yang Y, Shi C, Hu D, Qin H, Wang Z (2017) Lactobacillus plantarum LPOnlly alters the gut flora and attenuates colitis by inducing microbiome alteration in interleukin10 knockout mice. Mol Med Rep 16(5):5979–5985. https://doi.org/10.3892/mmr.2017.7351
Cui M, Xiao H, Li Y, Zhou L, Zhao S, Luo D, Zheng Q, Dong J, Zhao Y, Zhang X, Zhang J, Lu L, Wang H, Fan S (2017) Faecal microbiota transplantation protects against radiation-induced toxicity. EMBO Mol Med 9(4):448–461. https://doi.org/10.15252/emmm.201606932
Danilova NA, Abdulkhakov SR, Grigoryeva TV, Markelova MI, Vasilyev IY, Boulygina EA, Ardatskaya MD, Pavlenko AV, Tyakht AV, Odintsova AK, Abdulkhakov RA (2019) Markers of dysbiosis in patients with ulcerative colitis and Crohn’s disease. Ter Arkh 91(4):17–24. https://doi.org/10.26442/00403660.2019.04.000211
Depommier C, Everard A, Druart C, Plovier H, Van Hul M, Vieira-Silva S, Falony G, Raes J, Maiter D, Delzenne NM, de Barsy M, Loumaye A, Hermans MP, Thissen JP, de Vos WM, Cani PD (2019) Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat Med 25(7):1096–1103. https://doi.org/10.1038/s41591-019-0495-2
Derrien M, Vaughan EE, Plugge CM, de Vos WM (2004) Akkermansia muciniphila gen. nov., sp. nov., a human intestinal mucin-degrading bacterium. Int J Syst Evol Microbiol 54(Pt 5):1469–1476. https://doi.org/10.1099/ijs.0.02873-0
Dunn KA, Moore-Connors J, MacIntyre B, Stadnyk AW, Thomas NA, Noble A, Mahdi G, Rashid M, Otley AR, Bielawski JP, Van Limbergen J (2016) Early changes in microbial community structure are associated with sustained remission after nutritional treatment of pediatric Crohn’s disease. Inflamm Bowel Dis 22(12):2853–2862. https://doi.org/10.1097/MIB.0000000000000956
Earley H, Lennon G, Balfe A, Coffey JC, Winter DC, O’Connell PR (2019) The abundance of Akkermansia muciniphila and its relationship with sulphated colonic mucins in health and ulcerative colitis. Sci Rep 9(1):15683. https://doi.org/10.1038/s41598-019-51878-3
Fecal Microbiota Transplantation-standardization Study Group (2020) Nanjing consensus on methodology of washed microbiota transplantation. Chin Med J 133(19):2330–2332. https://doi.org/10.1097/cm9.0000000000000954
Ganesh BP, Klopfleisch R, Loh G, Blaut M (2013) Commensal Akkermansia muciniphila exacerbates gut inflammation in Salmonella Typhimurium-infected gnotobiotic mice. PLoS One 8(9):e74963. https://doi.org/10.1371/journal.pone.0074963
Grander C, Adolph TE, Wieser V, Lowe P, Wrzosek L, Gyongyosi B, Ward DV, Grabherr F, Gerner RR, Pfister A, Enrich B, Ciocan D, Macheiner S, Mayr L, Drach M, Moser P, Moschen AR, Perlemuter G, Szabo G, Cassard AM, Tilg H (2018) Recovery of ethanol-induced Akkermansia muciniphila depletion ameliorates alcoholic liver disease. Gut 67(5):891–901. https://doi.org/10.1136/gutjnl-2016-313432
Guo X, Li S, Zhang J, Wu F, Li X, Wu D, Zhang M, Ou Z, Jie Z, Yan Q, Li P, Yi J, Peng Y (2017) Genome sequencing of 39 Akkermansia muciniphila isolates reveals its population structure, genomic and functional diverisity, and global distribution in mammalian gut microbiotas. BMC Genomics 18(1):800. https://doi.org/10.1186/s12864-017-4195-3
Guo XK, Ou J, Liang S, Zhou X, Hu X (2018) Epithelial Hes1 maintains gut homeostasis by preventing microbial dysbiosis. Mucosal Immunol 11(3):716–726. https://doi.org/10.1038/mi.2017.111
Hakansson A, Tormo-Badia N, Baridi A, Xu J, Molin G, Hagslatt ML, Karlsson C, Jeppsson B, Cilio CM, Ahrne S (2015) Immunological alteration and changes of gut microbiota after dextran sulfate sodium (DSS) administration in mice. Clin Exp Med 15(1):107–120. https://doi.org/10.1007/s10238-013-0270-5
Hanninen A, Toivonen R, Poysti S, Belzer C, Plovier H, Ouwerkerk JP, Emani R, Cani PD, De Vos WM (2018) Akkermansia muciniphila induces gut microbiota remodelling and controls islet autoimmunity in NOD mice. Gut 67(8):1445–1453. https://doi.org/10.1136/gutjnl-2017-314508
Hoshi N, Schenten D, Nish SA, Walther Z, Gagliani N, Flavell RA, Reizis B, Shen Z, Fox JG, Iwasaki A, Medzhitov R (2012) MyD88 signalling in colonic mononuclear phagocytes drives colitis in IL-10-deficient mice. Nat Commun 3:1120. https://doi.org/10.1038/ncomms2113
Kang CS, Ban M, Choi EJ, Moon HG, Jeon JS, Kim DK, Park SK, Jeon SG, Roh TY, Myung SJ, Gho YS, Kim JG, Kim YK (2013) Extracellular vesicles derived from gut microbiota, especially Akkermansia muciniphila, protect the progression of dextran sulfate sodium-induced colitis. PLoS One 8(10):e76520. https://doi.org/10.1371/journal.pone.0076520
Kim SC, Tonkonogy SL, Albright CA, Tsang J, Balish EJ, Braun J, Huycke MM, Sartor RB (2005) Variable phenotypes of enterocolitis in interleukin 10-deficient mice monoassociated with two different commensal bacteria. Gastroenterology 128(4):891–906. https://doi.org/10.1053/j.gastro.2005.02.009
Kim MH, Kang SG, Park JH, Yanagisawa M, Kim CH (2013) Short-chain fatty acids activate GPR41 and GPR43 on intestinal epithelial cells to promote inflammatory responses in mice. Gastroenterology 145(2):396–406.e1-10. https://doi.org/10.1053/j.gastro.2013.04.056
Kim YS, Unno T, Kim BY, Park MS (2019) Sex differences in gut microbiota. World J Mens Health 38:48–60. https://doi.org/10.5534/wjmh.190009
Kump P, Wurm P, Gröchenig HP, Wenzl H, Petritsch W, Halwachs B, Wagner M, Stadlbauer V, Eherer A, Hoffmann KM, Deutschmann A, Reicht G, Reiter L, Slawitsch P, Gorkiewicz G, Högenauer C (2018) The taxonomic composition of the donor intestinal microbiota is a major factor influencing the efficacy of faecal microbiota transplantation in therapy refractory ulcerative colitis. Aliment Pharmacol Ther 47(1):67–77. https://doi.org/10.1111/apt.14387
Lee Y, Sugihara K, Gillilland MG 3rd, Jon S, Kamada N, Moon JJ (2019) Hyaluronic acid-bilirubin nanomedicine for targeted modulation of dysregulated intestinal barrier, microbiome and immune responses in colitis. Nat Mater 19:118–126. https://doi.org/10.1038/s41563-019-0462-9
Liso M, De Santis S, Verna G, Dicarlo M, Calasso M, Santino A, Gigante I, Eri R, Raveenthiraraj S, Sobolewski A, Palmitessa V, Lippolis A, Mastronardi M, Armentano R, Serino G, De Angelis M, Chieppa M (2020) A specific mutation in muc2 determines early dysbiosis in colitis-prone Winnie mice. Inflamm Bowel Dis 26(4):546–556. https://doi.org/10.1093/ibd/izz279
Lo Presti A, Del Chierico F, Altomare A, Zorzi F, Cella E, Putignani L, Guarino MPL, Monteleone G, Cicala M, Angeletti S, Ciccozzi M (2019) Exploring the genetic diversity of the 16S rRNA gene of Akkermansia muciniphila in IBD and IBS. Future Microbiol 14:1497–1509. https://doi.org/10.2217/fmb-2019-0175
Lo Sasso G, Khachatryan L, Kondylis A, Battey JND, Sierro N, Danilova NA, Grigoryeva TV, Markelova MI, Khusnutdinova DR, Laikov AV, Salafutdinov II, Romanova YD, Siniagina MN, Vasiliev IY, Boulygina EA, Solovyeva VV, Garanina EE, Kitaeva KV, Ivanov KY, Chulpanova DS, Kletenkov KS, Valeeva AR, Odintsova AK, Ardatskaya MD, Abdulkhakov RA, Ivanov NV, Peitsch MC, Hoeng J, Abdulkhakov SR (2020) Inflammatory bowel disease-associated changes in the gut: focus on Kazan patients. Inflamm Bowel Dis 27:418–433. https://doi.org/10.1093/ibd/izaa188
Lopez-Siles M, Enrich-Capo N, Aldeguer X, Sabat-Mir M, Duncan SH, Garcia-Gil LJ, Martinez-Medina M (2018) Alterations in the abundance and co-occurrence of Akkermansia muciniphila and Faecalibacterium prausnitzii in the colonic mucosa of inflammatory bowel disease subjects. Front Cell Infect Microbiol 8:281. https://doi.org/10.3389/fcimb.2018.00281
Marcella C, Cui B, Kelly CR, Ianiro G, Cammarota G, Zhang F (2021) Systematic review: the global incidence of faecal microbiota transplantation-related adverse events from 2000 to 2020. Aliment Pharmacol Ther 53(1):33–42. https://doi.org/10.1111/apt.16148
Myers GS, Rasko DA, Cheung JK, Ravel J, Seshadri R, DeBoy RT, Ren Q, Varga J, Awad MM, Brinkac LM, Daugherty SC, Haft DH, Dodson RJ, Madupu R, Nelson WC, Rosovitz MJ, Sullivan SA, Khouri H, Dimitrov GI, Watkins KL, Mulligan S, Benton J, Radune D, Fisher DJ, Atkins HS, Hiscox T, Jost BH, Billington SJ, Songer JG, McClane BA, Titball RW, Rood JI, Melville SB, Paulsen IT (2006) Skewed genomic variability in strains of the toxigenic bacterial pathogen, Clostridium perfringens. Genome Res 16(8):1031–1040. https://doi.org/10.1101/gr.5238106
Plovier H, Everard A, Druart C, Depommier C, Van Hul M, Geurts L, Chilloux J, Ottman N, Duparc T, Lichtenstein L, Myridakis A, Delzenne NM, Klievink J, Bhattacharjee A, van der Ark KC, Aalvink S, Martinez LO, Dumas ME, Maiter D, Loumaye A, Hermans MP, Thissen JP, Belzer C, de Vos WM, Cani PD (2017) A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat Med 23(1):107–113. https://doi.org/10.1038/nm.4236
Png CW, Lindén SK, Gilshenan KS, Zoetendal EG, McSweeney CS, Sly LI, McGuckin MA, Florin TH (2010) Mucolytic bacteria with increased prevalence in IBD mucosa augment in vitro utilization of mucin by other bacteria. Am J Gastroenterol 105(11):2420–2428. https://doi.org/10.1038/ajg.2010.281
Ring C, Klopfleisch R, Dahlke K, Basic M, Bleich A, Blaut M (2019) Akkermansia muciniphila strain ATCC BAA-835 does not promote short-term intestinal inflammation in gnotobiotic interleukin-10-deficient mice. Gut Microbes 10(2):188–203. https://doi.org/10.1080/19490976.2018.1511663
Seregin SS, Golovchenko N, Schaf B, Chen J, Pudlo NA, Mitchell J, Baxter NT, Zhao L, Schloss PD, Martens EC, Eaton KA, Chen GY (2017) NLRP6 Protects Il10(-/-) mice from colitis by limiting colonization of Akkermansia muciniphila. Cell Rep 19(4):733–745. https://doi.org/10.1016/j.celrep.2017.03.080
Smith PM, Howitt MR, Panikov N, Michaud M, Gallini CA, Bohlooly YM, Glickman JN, Garrett WS (2013) The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 341(6145):569–573. https://doi.org/10.1126/science.1241165
Van den Bossche L, Hindryckx P, Devisscher L, Devriese S, Van Welden S, Holvoet T, Vilchez-Vargas R, Vital M, Pieper DH, Vanden Bussche J, Vanhaecke L, Van de Wiele T, De Vos M, Laukens D (2017) Ursodeoxycholic acid and its taurine- or glycine-conjugated species reduce colitogenic dysbiosis and equally suppress experimental colitis in mice. Appl Environ Microbiol 83(7):e02766-16. https://doi.org/10.1128/AEM.02766-16
van der Velden AW, Lindgren SW, Worley MJ, Heffron F (2000) Salmonella pathogenicity island 1-independent induction of apoptosis in infected macrophages by Salmonella enterica serotype typhimurium. Infect Immun 68(10):5702–5709. https://doi.org/10.1128/iai.68.10.5702-5709.2000
Vigsnaes LK, Brynskov J, Steenholdt C, Wilcks A, Licht TR (2012) Gram-negative bacteria account for main differences between faecal microbiota from patients with ulcerative colitis and healthy controls. Benefic Microbes 3(4):287–297. https://doi.org/10.3920/BM2012.0018
Wagnerova A, Babickova J, Liptak R, Vlkova B, Celec P, Gardlik R (2017) Sex differences in the effect of resveratrol on DSS-induced colitis in mice. Gastroenterol Res Pract 2017:8051870–8051812. https://doi.org/10.1155/2017/8051870
Wang L, Tang L, Feng Y, Zhao S, Han M, Zhang C, Yuan G, Zhu J, Cao S, Wu Q, Li L, Zhang Z (2020a) A purified membrane protein from Akkermansia muciniphila or the pasteurised bacterium blunts colitis associated tumourigenesis by modulation of CD8(+) T cells in mice. Gut 69:1988–1997. https://doi.org/10.1136/gutjnl-2019-320105
Wang L, Tang L, Feng Y, Zhao S, Han M, Zhang C, Yuan G, Zhu J, Cao S, Wu Q, Li L, Zhang Z (2020b) A purified membrane protein from Akkermansia muciniphila or the pasteurised bacterium blunts colitis associated tumourigenesis by modulation of CD8(+) T cells in mice. Gut 69(11):1988–1997. https://doi.org/10.1136/gutjnl-2019-320105
Wu G, Zhang C, Wu H, Wang R, Shen J, Wang L, Zhao Y, Pang X, Zhang X, Zhao L, Zhang M (2017) Genomic microdiversity of Bifidobacterium pseudocatenulatum underlying differential strain-level responses to dietary carbohydrate intervention. mBio 8(1):e02348-16. https://doi.org/10.1128/mBio.02348-16
Yang L, Liu C, Zhao W, He C, Ding J, Dai R, Xu K, Xiao L, Luo L, Liu S, Li W, Meng H (2018) Impaired autophagy in intestinal epithelial cells alters gut microbiota and host immune responses. Appl Environ Microbiol 84(18):e00880-18. https://doi.org/10.1128/AEM.00880-18
Zhai R, Xue X, Zhang L, Yang X, Zhao L, Zhang C (2019) Strain-specific anti-inflammatory properties of two Akkermansia muciniphila strains on chronic colitis in mice. Front Cell Infect Microbiol 9:239. https://doi.org/10.3389/fcimb.2019.00239
Zhang T, Li Q, Cheng L, Buch H, Zhang F (2019) Akkermansia muciniphila is a promising probiotic. Microb Biotechnol 12(6):1109–1125. https://doi.org/10.1111/1751-7915.13410
Zhang T, Li P, Wu X, Lu G, Marcella C, Ji X, Ji G, Zhang F (2020a) Alterations of Akkermansia muciniphila in the inflammatory bowel disease patients with washed microbiota transplantation. Appl Microbiol Biotechnol 104:10203–10215. https://doi.org/10.1007/s00253-020-10948-7
Zhang T, Li P, Wu X, Lu G, Marcella C, Ji X, Ji G, Zhang F (2020b) Alterations of Akkermansia muciniphila in the inflammatory bowel disease patients with washed microbiota transplantation. Appl Microbiol Biotechnol 104(23):10203–10215. https://doi.org/10.1007/s00253-020-10948-7
Zhang T, Lu G, Zhao Z, Liu Y, Shen Q, Li P, Chen Y, Yin H, Wang H, Marcella C, Cui B, Cheng L, Ji G, Zhang F (2020c) Washed microbiota transplantation vs. manual fecal microbiota transplantation: clinical findings, animal studies and in vitro screening. Protein & cell 11(4):251–266. https://doi.org/10.1007/s13238-019-00684-8
Zhu W, Yan J, Zhi C, Zhou Q, Yuan X (2019) 1,25(OH)2D3 deficiency-induced gut microbial dysbiosis degrades the colonic mucus barrier in Cyp27b1 knockout mouse model. Gut Pathog 11:8. https://doi.org/10.1186/s13099-019-0291-z
Funding
This work was funded by Nanjing Medical University Fan Daiming Research Funds for HIM, the Graduate Research and Innovation Projects of Jiangsu Province (KYCX19_1171), and the Natural Science Fund for Colleges and Universities in Jiangsu Province, China (20KJD320002).
Author information
Authors and Affiliations
Contributions
TZ and XJ conceived the idea and wrote the manuscript. GL collected literature data and revised the manuscript. FZ conceived the idea and revised the manuscript. All authors approved the version to be submitted.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable.
Conflict of interest
Faming Zhang invented the concept of GenFMTer and transendoscopic enteral tubing and devices related to it. Other authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, T., Ji, X., Lu, G. et al. The potential of Akkermansia muciniphila in inflammatory bowel disease. Appl Microbiol Biotechnol 105, 5785–5794 (2021). https://doi.org/10.1007/s00253-021-11453-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11453-1