Abstract
Purpose
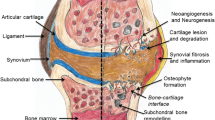
The aim of this study is to evaluate the outcomes of autologous microfragmented adipose tissue (MFAT) injection in elderly patients with knee osteoarthritis (OA). We hypothesized that MFAT knee infiltration for the treatment of knee OA would yield good clinical results out to two years follow-up.
Methods
Multi-centric, international, open-label study conducted by orthopedic surgery, and/or regenerative medicine facilities utilizing patient registries. Subjects recruited for eligibility. The primary outcome measure was Knee Injury and Osteoarthritis Outcome Score (KOOS). Outcomes and patient factors were compared to baseline, at six, 12, and 24 months. Statistical models were used to assess KOOS subscores and probability of exceeding the Minimally Clinically Important Difference (MCID) or Patient Acceptable Symptom State (PASS), and to assess the effect of the treatment variables on KOOS - Pain.
Results
Seventy-five patients, 120 primary treatments, mean age 69.6 years, (95%CI 68.3–70.9), BMI 28.4 (95%CI 27.3–29.6), with KL grade 2 to 4 knee OA treated with a single MFAT injection. KL grades 2 (15.1%), 3 (56.3%), and 4 (28.6%), with 20.8% of knees having previously undergone surgery. Patients with KL grade 2 disease had the best results in KOOS - Pain (P = 0.001), at six, 12, and 24 months. Including advanced KL grade 3 and 4 osteoarthritis patients, significant functional and quality of life success was seen in 106/120 treatments (88.3%, 66 patients) at all follow-up time points. Fourteen treatments (11.7%, 9 patients) failed prior to the study endpoint.
Conclusion
This study shows that a single-dose MFAT injection leads to clinical, functional, and quality of life improvement at two years in elderly patients, in KL grades 2 to 4 of knee osteoarthritis. These findings provide evidence that this treatment modality could be a safe and effective option to other commonly available treatments in carefully selected patients.




Similar content being viewed by others
Availability of data and material statement
Underlying data from this manuscript may be requested by qualified researchers upon request. Investigators may request access to deidentified patient data and redacted study documents which may include raw datasets, analysis-ready data sets, blank data forms. Prior to the use of data, proposals need to be approved by an independent review panel at www.clinicalstudyrequest.com and a signed data-sharing agreement will need to be executed. Some documents are available in Italian, and others in English.
References
Deshpande BR, Katz JR, Solomon DH et al (2016) Number of persons with symptomatic knee osteoarthritis in the US: impact of race and ethnicity, age, sex, and obesity. Arthritis Care Res 68:1743–1750
Guermazi A, Niu J, Hayashi D et al (2012) Prevalence of abnormalities in knees detected by MRI in adults without knee osteoarthritis: population based observational study (Framingham osteoarthritis study). BMJ 345:e5339
Litwic A, Edwards MH, Dennison EM et al (2013) Epidemiology and burden of osteoarthritis. Br Med Bull 105:185–199
Jevsevar DS (2013) Treatment of osteoarthritis of the knee: evidence-based guideline, 2nd edition. J Am Acad Orthop Surg 21:571–576
Cerza F, Carnì S, Carcangiu A et al (2012) Comparison between hyaluronic acid and platelet-rich plasma, intra-articular infiltration in the treatment of gonarthrosis. Am J Sports Med 40:2822–2827
Gobbi A, Karnatzikos G, Mahajan V et al (2014) Platelet-rich plasma treatment in symptomatic patients with knee osteoarthritis: preliminary results in a group of active patients. Sports Health 4:162–172
Ude CC, Sulaiman SB, Min-Hwei N et al (2014) Cartilage regeneration by chondrogenic induced adult stem cells in osteoarthritic sheep model. PLoS One 9:e98770
Guercio A, Di Marco P, Casella S et al (2012) Production of canine mesenchymal stem cells from adipose tissue and their application in dogs with chronic osteoarthritis of the humeroradial joints. Cell Biol Int 36:189–194
Kellgren JH, Lawrence JS (1957) Radiological assessment of osteoarthrosis. Ann Rheum Dis 16:494–502
Tremolada C, Ricordi C, Caplan AI et al (2016) Mesenchymal stem cells in Lipogems, a reverse story: from clinical practice to basic science. Methods Mol Biol 1416:109–122
Ulasli AM, Ozcakar L, Murrell WD (2019) Ultrasound imaging and guidance in the management of knee osteoarthritis in regenerative medicine field. J Clin Orthop Trauma 10:24–31
Roos EM, Lohmander LS (2003) The Knee Injury and Osteoarthritis Outcome Score (KOOS): from joint injury to osteoarthritis. Health Qual Life Outcomes 1:64
Lyman S, Lee YY, McLawhorn AS et al (2018) What are the minimal and substantial improvements in the HOOS and KOOS and JR versions after total joint replacement? Clin Orthop Relat Res 476:2432–2441
Çelik D, Coban Ö, Kiliçoğlu O (2019) Minimally clinically important difference of commonly used hip-, knee-, foot-, and ankle-specific questionnaires: a systematic review. J Clin Epidemiol 113:44–57
Statista from 2018 survey for TKR. Assessed 11.7.2020; https://www.statista.com/statistics/189975/cost-of-a-knee-replacement-in-various-countries/
Medical Tourism - 5 Spots for affordable high quality knee replacement.. Assessed 11.7.2020; https://internationalliving.com/medical-tourism-5-spots-for-an-affordable-high-quality-knee-replacement/ cost in Italy $22,729. 2018 newsletter
Connelly JW, Galea VP, Rojanasopondist P et al (2019) Patient acceptable symptom state at 1 and 3 years after total knee arthroplasty: thresholds for the Knee Injury and Osteoarthritis Outcome Score (KOOS). J Bone Joint Surg Am 101:995–1003
York Health Economics Consortium (2016) Incremental cost-effectiveness ratio (ICER). Assessed 22.7.2020; https://yhec.co.uk/glossary/incremental-cost-effectiveness-ratio-icer/
Skou ST, Roos E, Laursen M et al (2020) Cost-effectiveness of total knee replacement in addition to non-surgical treatment: a 2-year outcome from a randomised trial in secondary care in Denmark. BMJ Open 10:e033495
Nassar I, Fahey J, Mitchell D (2020) Rapid recovery following hip and knee arthroplasty using local infiltration analgesia: length of stay, rehabilitation protocol and cost savings. ANZ J Surg 90:355–359
Russo A, Screpis D, Di Donato SL et al (2017) Autologous and micro-fragmented adipose tissue for the treatment of diffuse degenerative knee osteoarthritis. J Exp Orthop 4:33
Murphy MB, Moncivais M, Caplan AI (2013) Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine. Exp Mol Med 45:e54
Lopa S, Colombini A, Moretti M et al (2019) Injective mesenchymal stem cell-based treatments for knee osteoarthritis: from mechanisms of action to current clinical evidences. Knee Surg Sports Traumatol Arthrosc 27:2003–2020
Koh YG, Choi YJ (2012) Infrapatellar fat pad-derived mesenchymal stem cell therapy for knee osteoarthritis. Knee 19:902–907
Koh YG, Jo SB, Kwon OR et al (2013) Mesenchymal stem cell injections improve symptoms of knee osteoarthritis. Arthroscopy 29:748–755
Adriani E, Moio M, Di Paola B et al (2017) Percutaneous fat transfer to treat knee osteoarthritis symptoms: preliminary results. Joints 5:89–92
Russo A, Screpis D, Di Donato SL et al (2018) Autologous micro-fragmented adipose tissue for the treatment of diffuse degenerative knee osteoarthritis: an update at 3 year follow-up. J Exp Orthop 5:52
Garza JR, Campbell RE, Tjoumakaris FP et al (2020) Clinical efficacy of intra-articular mesenchymal stromal cells for the treatment of knee osteoarthritis: a double-blinded prospective randomized controlled clinical trial. Am J Sports Med 48:588–598
Pancha J, Malanga G, Sheinkop M (2018) Safety and efficacy of percutaneous injection of lipogems micro-fractured adipose tissue for osteoarthritic knees. Am J Orthop (Belle Mead NJ) 47. https://doi.org/10.12788/ajo.2018.0098
Caplan AI, Correa D (2010) The MSC: an injury drugstore. Cell Stem Cell 9:11–15
Caplan AI (2017) Mesenchymal stem cells: time to change the name! Stem Cells Transl Med 6:1445–1451
Murray IR, Chahla J, Safran MR et al (2019) International expert consensus on a cell therapy communication tool: DOSES. J Bone Joint Surg Am 101:904–911
Murray IR, Geeslin AG, Goudie EB et al (2017) Minimum information for studies evaluating biologics in Orthopaedics (MIBO): platelet-rich plasma and mesenchymal stem cells. J Bone Joint Surg Am 99:809–819
Chu CR, Rodeo S, Bhutani N (2019) Optimizing clinical use of biologics in orthopaedic surgery: consensus recommendations from the 2018 AAOS/NIH U-13 conference. J Am Acad Orthop Surg 27:e50–e63
Marenah M, Li J, Kumar A et al (2019) Quality assurance and adverse event management in regenerative medicine for knee osteoarthritis: current concepts. J Clin Orthop Trauma 10:53–58
Acknowledgments
The authors would like to acknowledge Corey Scholes, EBM Analytics, and Nico Nagelkerke for their contribution to the statistical analysis of this work. The authors would also like to acknowledge Pam Jackson, PhD, for editorial contributions.
Funding
Self-funded.
Author information
Authors and Affiliations
Contributions
All authors analyzed and/or interpreted data. All authors collaborated in the drafting and critical revision of the manuscript. All authors approved the final version of the manuscript and vouch for the accuracy of the analysis and the fidelity of the study to the protocol.
Corresponding author
Ethics declarations
Consent to participate
All patients were approved for treatment by written informed consent.
Consent for publication
Written informed consent was obtained from all patients.
Competing interests
C.R.: Consultant, Lipogems, Inc.; Medical Director and shareholder, Personalized Stem Cells, Inc.; Co-Founder and shareholder, DataBiologics, Inc.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Level of Evidence: Level 4
Rights and permissions
About this article
Cite this article
Gobbi, A., Dallo, I., Rogers, C. et al. Two-year clinical outcomes of autologous microfragmented adipose tissue in elderly patients with knee osteoarthritis: a multi-centric, international study. International Orthopaedics (SICOT) 45, 1179–1188 (2021). https://doi.org/10.1007/s00264-021-04947-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-021-04947-0