Abstract
Purpose
Modulation of estrogen receptor (ER) plays a central role in selective estrogen receptor modulators (SERMs) molecular mechanism of action, although studies have indicated that additional, non-ER-mediated mechanisms exist. It has been suggested that the induction of oxidative stress by SERM could be one of the non-ER-mediated mechanisms held responsible for their pro-apoptotic role in ER-negative cells. Tumor cells are known for their high requirement of glutamine (Gln) that serves multiple functions within the cells, including nutritional and energy source, as well as one of the precursors for the synthesis of natural antioxidant glutathione (GSH). We hypothesized that one of the mechanisms responsible for ER-independent anti-neoplastic properties of SERMs and also for their adverse side effects could be dependent on the inhibition of Gln uptake.
Methods
Human ER-negative MDA-MB231 breast cancer cells were treated with different doses of Tam and Ral. Gln uptake was monitored by using [3H]Gln assay. The effect of Tam and Ral on Gln transporter ASCT2 expression, glutathione (GSH) levels and cellular proliferation was determined.
Results
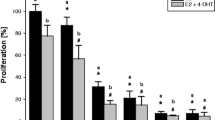
Tam and Ral inhibited Gln uptake in a dose-dependent manner through inhibition of ASCT2 Gln transporter. This effect of the anti-estrogens was associated with inhibition of GSH production and apoptosis. Treatment of cells with N-acetyl L-cysteine and 17 beta-estradiol 2 reversed the effects of Ral and Tam.
Conclusions
Our results indicate that one of the mechanisms of action (and possibly some of the side effects) of TAM and RAL is associated with inhibition of cellular Gln uptake, oxidative stress and induction of apoptosis.




Similar content being viewed by others
References
Love RR (1989) Tamoxifen therapy in primary breast cancer: biology, efficacy, and side effects. J Clin Oncol 7:803–815
Jaiyesimi IA, Buzdar AU, Decker DA, Hortobagyi GN (1995) Use of tamoxifen for breast cancer: twenty-eight years later. J Clin Oncol 13:513–529
Perry RR, Kang Y, Greaves B (1995) Effects of tamoxifen on growth and apoptosis of oestrogen-dependent and -independent human breast cancer cells. Ann Surg Oncol 2:238–245
Ferlini C, Scambia G, Marone M, Distefano M, Gaggini C, Ferrandina G, Fattorossi A, Isola G, Benedetti Panici P, Mancuso S (1999) Tamoxifen induces oxidative stress and apoptosis in oestrogen receptor-negative human cancer cell lines. Br J Cancer 79:257–263
Corti PS (1993) Apuzzo MLJ: Clinical and radiographic response in a minority of patients with recurrent malignant gliomas treated with high-dose tamoxifen. Neurosurgery 32:485–490
Couldwell WT, Hinton DR, He S, Chen TC, Sebat I, Weiss MH, Law RE (1994) Protein kinase C inhibitors induce apoptosis in human malignant glioma cell lines. FEBS Lett 345:43–46
McClay EF, Albright KD, Jones JA, Christen RD, Howell SB (1993) Tamoxifen modulation of cisplatin sensitivity in human malignant melanoma cells. Cancer Res 53:1571–1576
Taylor OM, Benson EA, McMahon MJ (1993) Clinical trial of tamoxifen in patients with irresectable pancreatic adenocarcinoma. The Yorkshire Gastrointestinal Tumor Group. Br J Surg 80:384–386
Gelmann EP (1997) Tamoxifen for the treatment of malignancies other than breast and endometrial carcinoma. Semin Oncol 24:S1-65–S1-70
Ward RL, Morgan G, Falley D, Kelly PJ (1993) Tamoxifen reduces bone turnover and prevents lumbar spine proximal femoral bone loss in early postmenopausal women. Bone Miner 22:87–94
Recker R (1993) Clinical review 41: current therapy for osteoporosis. J Clin Endocrinol Metab 76:14–16
Grainger DJ, Witchell CM (1995) Metcalfe JC: Tamoxifen elevates transforming growth factor-ß suppresses diet-induced formation of lipid lesions in mouse aorta. Nat Med 1:1067–1073
Reckless J, Metcalfe JC, Grainger DJ (1997) Tamoxifen decreases cholesterol seven-fold and abolishes lipid lesion development in apolipoprotein E knock out mice. Circulation 95:1542–1548
Sthoeger Z, Dayan M, Zinger H, Kalush F, Mor G, Zlatman YA, Kohen F, Mozes E (1997) Treatments with tamoxifen and an antiestradiol antibody have beneficial effects on experimental SLE via cytokine modulation. Ann N Y Acad Sci 815:367–368
Yildiz A, Guleryuz S, Ankerst DP, Ongür D, Renshaw PF (2008) Protein kinase C inhibition in the treatment of mania: a double-blind, placebo-controlled trial of tamoxifen. Arch Gen Psychiatry 65:255–263
Mandlekar S, Kong A-NT (2001) Mechanisms of tamoxifen-induced apoptosis. Apoptosis 6:469–477
Eagle H (1955) Nutrition needs of mammalian cells in tissue culture. Science 122:501–514
Labow BI, Souba WW (2000) Glutamine. W J Surgery 24:1503–1513
Meister A, Anderson MA (1983) Glutathione. Annu Rev Biochem 52:711–721
Abcouwer SF, Bode BP, Souba WW (1996) Glutamine as a metabolic intermediate. In: Fisher JE (ed) Nutrition and metabolism in the surgical patient. Little Brown, Boston, pp 353–384
Coles NW, Johnstone RM (1962) Glutamine metabolism in Ehrlich ascites-carcinoma cells. Biochem J 83:284–291
Souba WW (1993) Glutamine and cancer. Ann Surgery 218:715–728
Mates JM, Perez-Gomez C, Nunez DC, Asenjo IM, Marquez J (2002) Glutamine and its relationship with intracellular redox status, oxidative stress and cell proliferation/death. Int J Biochem Cell Biol 34:439–458
DeBerardinis RJ, DeBerardinis RJ, Mancuso A, Daikhin E, Nissim I, Yudkoff M, Wehrli S, Thompson CB (2007) Beyond aerobic glycolysis: transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc Natl Acad Sci USA 104:19345–19350
Klimberg VS (1996) Glutamine, cancer, and its therapy. Am J Surgery 172:418–424
Chen MK, Espat NJ, Bland KI, Copeland EM III, Souba WW (1993) Influence of progressive tumor growth on glutamine metabolism in skeletal muscle and kidney. Ann Surg 217:655–666
Bode BP (2001) Recent molecular advances in mammalian glutamine transport. J Nutr 131(Suppl):2475S–2487S
Fuchs BC, Bode BP (2005) Amino acid transporters ASCT2 and LAT1 in cancer: partners in crime? Sem Cancer Biol 15:254–266
Bungard CI, McGivan JD (2004) Glutamine availability up-regulates expression of the amino acid transporter protein ASCT2 in HepG2 cells and stimulates the ASCT2 promoter. Biochem J 382:27–32
Jaiyesimi IA, Buzdar AU, Decker DA, Hortobagyi GN (1995) Use of tamoxifen for breast cancer: twenty-eight years later. J Clin Oncol 13:513–529
O’Brian CA, Housey GM, Weinstein IB (1988) Specific and direct binding of protein kinase C to an immobilized tamoxifen analogue. Cancer Res 48:3626–3629
Kang Y, Cortina R, Perry RR (1996) Role of c-myc in tamoxifen induced apoptosis estrogen-independent breast cancer cells. J Natl Cancer Inst 88:279–284
Maurer BJ, Metelitsa LS, Seeger RC, Cabot MC, Reynolds CP (1999) Increase of ceramide and induction of mixed apoptosis/necrosis by N-(4-hydroxyphenyl)- retinamide in neuroblastoma cell lines. J Natl Cancer Inst 91:1138–1146
Mandlekar S, Yu R, Tan TH, Kong AN (2000) Activation of caspase-3 and c-Jun NH2-terminal kinase-1 signaling pathways in tamoxifen-induced apoptosis of human breast cancer cells. Cancer Res 60:5995–6000
Koli KM, Ramsey TT, Ko Y, Dugger TC, Brattain MG, Arteaga CL (1997) Blockade of transforming growth factor-beta signaling does not abrogate antiestrogen-induced growth inhibition of human breast carcinoma cells. J Biol Chem 272:8296–8302
Perry RR, Kang Y, Greaves BR (1995) Relationship between tamoxifen-induced transforming growth factor beta 1 expression, cytostasis and apoptosis in human breast cancer cells. Br J Cancer 72:1441–1446
Brünner N, Yee D, Kern FG, Spang-Thomsen M, Lippman ME, Cullen KJ (1993) Effect of endocrine therapy on growth of T61 human breast cancer xenografts is directly correlated to a specific down-regulation of insulin-like growth factor II (IGF-II). Eur J Cancer 29A(4):562–569
Ferlini C, Scambia G, Marone M, Distefano M, Gaggini C, Ferrandina G, Fattorossi A, Isola G, Benedetti Panici P, Mancuso S (1999) Tamoxifen induces oxidative stress and apoptosis in oestrogen receptor-negative human cancer cell lines. Br J Cancer 79:257–263
Hayon T, Dvilansky A, Oriev L, Nathan I (1999) Non-steroidal antiestrogens induce apoptosis in HL60 and MOLT3 leukemic cells; involvement of reactive oxygen radicals and protein kinase C. Anticancer Res 19:2089–2093
Mandlekar S, Kong AN (2001) Mechanisms of tamoxifen-induced apoptosis. Apoptosis 6:469–477
Dietze EC, Caldwell LE, Grupin SL, Mancini M, Seewaldt VL (2001) Tamoxifen but not 4-hydroxytamoxifen initiates apoptosis in p53(–) normal human mammary epithelial cells by inducing mitochondrial depolarization. J Biol Chem 276:5384–5394
Nazarewicz RR, Zenebe WJ, Parihar A, Larson SK, Alidema E, Choi J, Ghafourifar P (2007) Tamoxifen induces oxidative stress and mitochondrial apoptosis via stimulating mitochondrial nitric oxide synthase. Cancer Res 67:1282–1290
Kosower NS, Kosower EM (1978) The glutathione status of cells. Intl Rev Cytology 54:109–156
Kehrer JP, Lund LG (1994) Cellular reducing equivalents and oxidative stress. Free Rad Biol Med 17:65–75
Griffith OW (1999) Biologic and pharmacologic regulation of mammalian glutathione synthesis. Free Radic Biol Med 27(9–10):922–935
Amores-Sanchez MI, Medina MA (1999) Glutamine, as a precursor of glutathione, and oxidative stress. Mol Genet Metabol 67:100–105
Zafarullah M, Li WQ, Sylvester J, Ahmad M (2003) Molecular mechanisms of N-acetylcysteine actions. Cell Mol Life Sci 60:6–20
Baggetto LG (1992) Deviant energetic metabolism of glycolytic cancer cells. Biochimie 74:959–974
Mazurek S, Eigenbrodt E (2003) The tumor metabolome. Anticancer Res 23:1149–1154
Medina MA, Sanchez-Jimenes F, Marquez J, Rodruguez Quesada A, Nunez de Castro I (1992) Relevance of glutamine metabolism to tumor cell growth. Mol Cell Biochem 113:1–15
Palacin M, Estevez R, Bertran J, Zorzano A (1998) Molecular biology of mammalian plasma membrane amino acid transporters. Physiol Rev 78:969–1054
Collins CL, Wasa M, Souba WW, Abcouwer SF (1998) Determinants of glutamine dependence and utilization by normal and tumor-derived breast cell lines. J Cell Physiol 176:166–178
Fuchs BC, Perez JC, Suetterlin JE, Chaudhry SB, Bode BP (2004) Inducible antisense RNA targeting amino acid transporter ATB0/ASCT2 elicits apoptosis in human hepatoma cells. Am J Physiol Gastrointest Liver Physiol 286:G467–G478
Wang X, Dykens JA, Perez E, Liu R, Yang S, Covey DF, Simpkins JW (2006) Neuroprotective effects of 17beta-estradiol and nonfeminizing estrogens against H2O2 toxicity in human neuroblastoma SK-N-SH cells. Mol Pharmacol 70:395–404
Acknowledgments
This work was supported by VA Merit Review Award to VS Klimberg.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Todorova, V.K., Kaufmann, Y., Luo, S. et al. Tamoxifen and raloxifene suppress the proliferation of estrogen receptor-negative cells through inhibition of glutamine uptake. Cancer Chemother Pharmacol 67, 285–291 (2011). https://doi.org/10.1007/s00280-010-1316-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-010-1316-y