Abstract
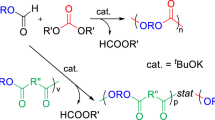
The random copolymers of ε-caprolactone (CL) and 2,2-ethylenedioxy propane-1,3-diol carbonate (EOPDC) were synthesized in bulk at 120 °C using Sn(Oct)2 as a catalyst. The poly(EOPDC-co–CL)s obtained were characterized by FT IR, 1H NMR, 13C NMR, GPC and DSC. The copolymers were obtained with yield of 84.2–97.8 %. The number-average molecular weight of the copolymer is 2.75–7.76 × 104 with a polydispersity of 1.52–1.68. The properties of the copolymer including the enzymatic degradation by Pseudomonas Cepacia lipase and drug-controlled release property were also investigated. The results showed that the copolymers are degradable at physiological conditions, and their degradation rate and release of Tegafur in the copolymers increase with increasing CL content in the copolymers.






Similar content being viewed by others
References
Martina M, Hutmacher DW (2007) Biodegradable polymers applied in tissue engineering research: a review. Polym Int 56:145–157
Labet M, Thielemans W (2009) Synthesis of polycaprolactone: a review. Chem Soc Rev 38:3484–3504
Wei ZY, Liu L, Yu FY, Wang P, Qu C, Qi M (2008) Synthesis of poly(epsilon-caprolactone)-poly(l-lactide) block copolymers by melt or solution sequential copolymerization using nontoxic dibutylmagnesium as initiator. Polym Bull 61:407–413
Sivalingam G, Madras G (2004) Thermal degradation of binary physical mixtures and copolymers of poly(ε-caprolactone), poly(d, l-lactide), poly(glycolide). Polym Degrad Stab 84:393–398
Cho DK, Park JW, Kim SH, Kim YH, Im SS (2003) Effect of molecular orientation on biodegradability of poly(glycolide-co-epsilon-caprolactone). Polym Degrad Stab 80:223–232
Storey RF, Mullen BD, Melchert KM (2001) Synthesis of novel hydrophilic poly(ester-carbonates) containing pendent carboxylic acid groups. J Macromol Sci, Pure Appl Chem 38:897–917
Guan HL, Xie ZG, Tang ZH, Xu XY, Chen XS, Jing XB (2005) Preparation of block copolymer of epsilon-caprolactone and 2-methyl-2-carboxyl-propylene carbonate. Polymer 46:2817–2824
Pego AP, Luyn MJAV, Brouwer LA, Wachem PBV, Poot AA, GrijiPma DW (2003) In vivo behavior of poly(1,3-trimethylene carbonate) and copolymers of 1,3-trimethylene carbonate with d, l-lactide or epsilon-caprolactone: degradation and tissue response. J Biomed Mater Res 67(A):1044–1054
Ling J, Zhu WP, Shen ZQ (2004) Controlling ring-opening copolymerization of epsilon- caprolactone with trimethylene carbonate by scandium tris(2,6-di-tert -butyl-4-methylphenolate). Macromolecules 37:758–763
Schappacher M, Fabre T, Mingotaud AF, Soum A (2001) Study of a (trimethylenecarbonate-co-epsilon-caprolactone) polymer—part 1: preparation of a new nerve guide through controlled random copolymerization using rare earth catalysts. Biomaterials 22:2849–2855
Fabre T, Schappacher M, Bareille R, Dupuy B, Soum A, Bertrand-Barat J, Baquey C (2001) Study of a (trimethylenecarbonate-co- epsilon-caprolactone) polymer—part 2: in vitro cytocompatibility analysis and in vivo ED1 cell response of a new nerve guide. Biomaterials 22:2951–2958
Zhu GX, Ling J, Shen ZQ (2003) Isothermal crystallization of random copolymers of epsilon-caprolactone with 2,2-dimethyltrimethylene carbonate. Polymer 44:5827–5832
Shen ZQ, Zhu GX, Ling J (2002) Homo- and copolymerization of epsilon-caprolactone and 2,2-dimethyltrimethylene carbonate by rare earth initiators. Chin J Chem 20:1369–1374
Yasuda H, Aludin MS, Kitamura N, Tanabe M, Sirahama H (1999) Syntheses and physical properties of novel optically active poly(ester-carbonate)s by copolymerization of substituted trimethylene carbonate with epsilon-caprolactone and their biodegradation behavior. Macromolecules 32:6047–6057
Raquez JM, Degee P, Narayan R, Dubois P (2004) Diblock copolymers based on 1,4-dioxan-2-one and epsilon-caprolactone: characterization and thermal properties. Macromol Chem Phys 205:1764–1773
Ge H, Hu Y, Jiang X, Cheng D, Yuan Y, Bi H, Yang C (2002) Preparation, characterization, and drug release behaviors of drug nimodipine-loaded poly(epsilon-caprolactone)-poly(ethylene oxide)-poly(epsilon-caprolactone) amphiphilic triblock copolymer micelles. J Pharm Sci 91:1463–1473
He F, Li S, Vert M, Zhuo R (2003) Enzyme-catalyzed polymerization and degradation of copolymers prepared from ε-caprolactone and poly(ethylene glycol). Polymer 44:5145–5151
You JH, Choi SW, Kim JH (2008) Synthesis and microphase separation of biodegradable poly(ε-caprolactone)-poly(ethylene glycol)-poly(ε-caprolactone) multiblock copolymer films. Macromol Res 16:609–613
Rokicki G (2000) Aliphatic cyclic carbonates and spiroorthocarbonates as monomers. Prog Polym Sci 25:259–342
Feng J, Zhuo RX, Zhang XZ (2012) Construction of functional aliphatic polycarbonates for biomedical applications. Prog Polym Sci 37:211–236
Mullen BD, Tang CN, Storey RF (2003) New aliphatic poly(ester-carbonates) based on 5-methyl-5-allyloxycarbonyl-1,3-dioxan-2-one. J Polym Sci, Part A: Polym Chem 41:1978–1991
He F, Wang YP, Liu G, Jia HL, Feng J, Zhuo RX (2008) Synthesis, characterization and ring-opening polymerization of a novel six-membered cyclic carbonate bearing pendent allyl ether group. Polymer 49:1185–1190
Wang XL, Zhuo RX, Liu LJ, He F, Liu G (2002) Synthesis and characterization of novel aliphatic polycarbonates. J Polym Sci, Part A: Polym Chem 40:70–75
Li ZM, Yan GP, Ai CW, Zhang Q, Li L, Liu F, Yu XH, Zhao BA (2012) Synthesis and properties of polycarbonate copolymers of trimethylene carbonate and 2-phenyl-5,5- bis(hydroxymethyl) trimethylene carbonate. J Appl Polym Sci 124:3704–3713
Sanda F, Kamatani J, Endo T (2001) Synthesis and anionic ring-opening polymerization behavior of amino acid-derived cyclic carbonates. Macromolecules 34:1564–1569
Al-Azemi TF, Bisht KS (2002) One-step synthesis of polycarbonates bearing pendant carboxyl groups by lipase-catalyzed ring-opening polymerization. J Polym Sci, Part A: Polym Chem 40:1267–1274
Zhou Y, Zhuo RX, Liu ZL (2005) Synthesis and characterization of novel aliphatic poly(carbonate-ester)s with functional pendent groups. Macromol Rapid Commun 26:1309–1404
Zelikin AN, Zawaneh PN, Putnam D (2006) A functionalizable biomaterial based on dihydroxyacetone. an intermediate of glucose metabolism. Biomacromolecules 7:3239–3244
Wang LS, Cheng SX, Zhuo RX (2004) Novel biodegradable aliphatic polycarbonate based on ketal protected dihydroxyacetone. Macromol Rapid Commun 25:959–963
Wang LS, Jiang XS, Wang H, Cheng SX, Zhuo RX (2005) Preparation and cytotoxicity of novel aliphatic polycarbonate synthesized from dihydroxyacetone. Chin Chem Lett 16:572–574
Acknowledgments
The authors are grateful for the financial support of national key basic research and development program (G1999064703) and natural science foundation of China (Grant No. 20174029).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, Ls., Cheng, Sx. & Zhuo, Rx. Syntheses and properties of novel copolymers of polycaprolactone and aliphatic polycarbonate based on ketal-protected dihydroxyacetone. Polym. Bull. 71, 47–56 (2014). https://doi.org/10.1007/s00289-013-1044-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-013-1044-7