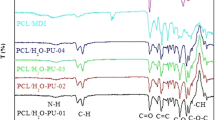
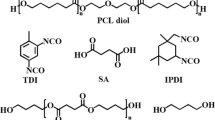
Summary
Polyurethanes based on a 50/50 copoly(L-lactide/ɛ-caprolactone) prepolymer and butanediisocyanate were made. Chain extending a diisocyanate terminated prepolymer with butanediamine was not possible due to the susceptibility of the lactide bond to aminolysis. Chain extension with butanediol resulted in a polymer with poor mechanical properties due to transesterification. When the copolymer prepolymer was chain-extended with an isocyanate terminated block, transesterification with the chain-extender was avoided and the mechanical properties were increased. When the length of the hard segments was increased the mechanical properties increased further. A L-lactide/ɛ-caprolactone based polyurethane with a tensile strength and modulus of respectively 45 MPa and 60 MPa was made. The polymer contained no poly(L-lactide) crystals and was easy to process. Compared to the high molecular weight 50/50 copoly(L-lactide/ɛ-caprolactone) the polyurethane showed better mechanical properties, is expected to have the same adhesive properties and is expected to have a slower degradation rate. These factors makes this polymer excessively useful for in-vivo tissue engineering in for instance meniscal reconstruction material, nerve guide and artificial skin.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 12 March 1998/Revised version: 26 June 1998/Accepted: 26 June 1998
Rights and permissions
About this article
Cite this article
de Groot, J., Spaans, C., Dekens, F. et al. On the role of aminolysis and transesterification in the synthesis of ɛ-caprolactone and L-lactide based polyurethanes. Polymer Bulletin 41, 299–306 (1998). https://doi.org/10.1007/s002890050366
Issue Date:
DOI: https://doi.org/10.1007/s002890050366