Abstract
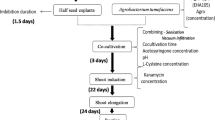
Modern genetic analysis and manipulation of soybean (Glycine max) depend heavily on an efficient and dependable transformation process, especially in public genotypes from which expressed sequence tag (EST), bacterial artificial chromosome and microarray data have been derived. Williams 82 is the subject of EST and functional genomics analyses. However, it has not previously been transformed successfully using either somatic embryogenesis-based or cotyledonary-node transformation methods, the two predominant soybean transformation systems. An advance has recently been made in using antioxidants to enhance Agrobacterium infection of soybean. Nonetheless, an undesirable effect of using these antioxidants is the compromised recovery of transgenic soybean when combined with the use of the herbicide glufosinate as a selective agent. Therefore, we optimized both Agrobacterium infection and glufosinate selection in the presence of l-cysteine for Williams 82. We have recovered transgenic lines of this genotype with an enhanced transformation efficiency using this herbicide selection system.

Similar content being viewed by others
Abbreviations
- DTT :
-
Dithiothreitol
- EST :
-
Expressed sequence tag
- GUS :
-
β-Glucuronidase
References
Bowley SR (1999) A hitchhiker’s guide to statistics in plant biology. Any Old Subject Books, Guelph, Ontario
Clemente TE, LaVallee BJ, Howe AR, Conner-Ward D, Rozman RJ, Hunter PE, Broyles DL, Kasten DS, Hinchee MA (2000) Progeny analysis of glyphosate selected transgenic soybean derived from Agrobacterium-mediated transformation. Crop Sci 40:797–803
Di R, Purcell V, Collins GB, Ghabrial SA (1996) Production of transgenic soybean lines expressing the bean pod mottle virus coat protein precursor gene. Plant Cell Rep 15:746–750
Enriquez-Obregon GA, Prieto-Samsonov DL, de la Riva GA, Perez M, Selman-Housein G, Vazquez-Padron RI (1999) Agrobacterium-mediated Japonica rice transformation: a procedure assisted by an antinecrotic treatment. Plant Cell Tissue Organ Cult 59:159–168
Enriquez-Obregon GA, Vazquez-Padron RI, Prieto-Samsonov DL, de la Riva GA, Selman-Housein G (1998) Herbicide-resistant sugarcane (Saccharum officinarum L.) plants by Agrobacterium-mediated transformation. Planta 206:20–27
Fagard M, Vaucheret H (2000) (Trans)gene silencing in plants: how many mechanisms? Annu Rev Plant Physiol Plant Mol Biol 51:167–194
Finer JJ, McMullen MD (1991) Transformation of soybean via particle bombardment of embryogenic suspension culture tissue. In Vitro Cell Dev Biol Plant 27:175–182
Finer JJ, Vain P, Jones MW, McMullen MD (1992) Development of the particle inflow gun for DNA delivery to plant cells. Plant Cell Rep 11:323–328
Frame B, Shou H, Chikwamba R, Zhang Z, Xiang C, Fonger T, Pegg SE, Li B, Nettleton D, Pei D, Wang K (2002) Agrobacterium-mediated transformation of maize embryos using a standard binary vector system. Plant Physiol 129:13–22
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Expt Cell Res 50:151–158
Hadi MZ, McMullen MD, Finer JJ (1996) Transformation of 12 different plasmids into soybean via particle bombardment. Plant Cell Rep 15:500–505
Hazel CB, Klein TM, Anis M, Wilde HD, Parrott WA (1998) Growth characteristics and transformability of soybean embryogenic cultures. Plant Cell Rep 17:765–772
Hinchee MA, Connor-Ward DV, Newell CA, McDonnell RE, Sato SJ, Gasser CS, Fischhoff DA, Re DB, Fraley RT, Horsch RB (1988) Production of transgenic soybean plants using Agrobacterium-mediated DNA transfer. BioTechnology 6:915–922
Hood EE, Helmer GL, Fraley RT, Chilton MD (1986) The hypervirulence of Agrobacterium tumefaciens A281 is encoded in a region of pTiBo542 outside of T-DNA. J Bacteriol 168:1291–1301
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Khan MRI, Table LM, Heath LC, Spencer D, Higgins TJV (1994) Agrobacterium-mediated transformation of subterranean clover (Trifolium subterraneum L.). Plant Physiol 105:81–88
Lohnes DG, Wagner RE, Bernard RL (1993) Soybean genes Rj2, Rmd, and Rps2 in linkage group 19. J Hered 84:109–111
Olhoft PM, Flagel LE, Donovan CM, Somers DA (2003) Efficient soybean transformation using hygromycin B selection in the cotyledonary-node method. Planta 216:723–735
Olhoft PM, Lin K, Galbraith J, Nielsen NC, Somers DA (2001) The role of thiol compounds increasing Agrobacterium-mediated transformation of soybean cotyledonary-node cells. Plant Cell Rep 20:731–737
Olhoft PM, Somers DA (2001) l-Cysteine increases Agrobacterium-mediated T-DNA delivery into soybean cotyledonary-node cells. Plant Cell Rep 20:706–711
Parrott WA, All JN, Adang MJ, Bailey MA, Boerma HR, Stewart CN Jr (1994) Recovery and evaluation of soybean plants transgenic for a Bacillus thuringiensis var. kurstaki insecticidal gene. In Vitro Cell Dev Biol-Plant 30:144–149
Perl A, Lotan O, Abu-Abied M, Holland D (1996) Establishment of an Agrobacterium-mediated transformation system for grape (Vitis vinifera L.): the role of antioxidants during grape-Agrobacterium interactions. Nat Biotechnol 14:624–628
Santarem ER, Finer JJ (1999) Transformation of soybean [Glycine max (L.) Merrill] using proliferative embryogenic tissue maintained on semisolid medium. In Vitro Cell Dev Biol Plant 35:451–455
Shoemaker R, Keim P, Vodkin L, Retzel E, Clifton SW, Waterston R, Smoller D, Coryell V, Khanna A, Erpelding J, Gai X, Brendel V, Raph-Schmidt C, Shoop EG, Vielweber CJ, Schmatz M, Pape D, Bowers Y, Theising B, Martin J, Dante M, Wylie T, Granger C (2002) A compilation of soybean ESTs: generation and analysis. Genome 45:329–338
Southern EM (1975) Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol 98:503–517
Stewart CN Jr, Adang MJ, All JN, Boerma HR, Cardineau G, Tucker D, Parrott WA (1996) Genetic transformation, recovery, and characterization of fertile soybean transgenic for a synthetic Bacillus thuringiensis cryIAc gene. Plant Physiol 112:121–129
Vancanneyt G, Schmidt R, O’Connor-Sanchez A, Willmitzer L, Rocha-Sosa M (1990) Construction of an intron-containing marker gene: splicing of the intron in transgenic plants and its use in monitoring early events in Agrobacterium-mediated plant transformation. Mol Gen Genet 220:245–250
Xing A, Zhang Z, Sato S, Staswick PE, Clemente TE (2000) The use of the two T-DNA binary system to derive marker-free transgenic soybeans. In Vitro Cell Dev Biol Plant 36:456–463
Zhang Z, Xing A, Staswick PE, Clemente TE (1999) The use of glufosinate as a selective agent in Agrobacterium-mediated transformation of soybean. Plant Cell Tissue Organ Cult 56:37–46
Acknowledgements
We would like to thank Jamie Schlereth and Lan Wang for their technical assistance. Special thanks to Dr. Hari Krishnan for critical review of this manuscript. This research is supported by grants #9872565 from the NSF Plant Genome Initiative and from the University of Missouri-Columbia Life Sciences Mission Enhancement Program. All experiments were conducted at the Plant Transformation Core Facility, University of Missouri—Columbia.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Ozias-Akins
Rights and permissions
About this article
Cite this article
Zeng, P., Vadnais, D.A., Zhang, Z. et al. Refined glufosinate selection in Agrobacterium-mediated transformation of soybean [Glycine max (L.) Merrill]. Plant Cell Rep 22, 478–482 (2004). https://doi.org/10.1007/s00299-003-0712-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-003-0712-8