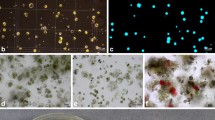
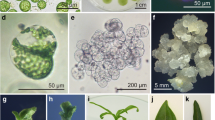
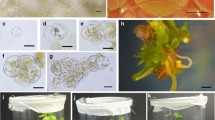
Abstract
Helianthus maximiliani is one of the wild Helianthus species with the genes for resistance to many pathogens including Sclerotinia sclerotiorum. Unfortunately, a transfer of disease resistance genes from this species into the cultivated sunflower is limited by its poor crossability with the cultivated sunflower and sterility of interspecific hybrids. To overcome this problem, mesophyll protoplasts of Sclerotinia sclerotiorum-resistant clone of H. maximiliani were electrically fused with etiolated hypocotyl protoplasts of the cultivated sunflower inbred line PH-BC1-91A. Fusion products were embedded in agarose droplets and subjected to different regeneration protocols. Developed microcalluses were released from the agarose and transferred into solid media. Shoot regeneration was achieved by culture of calluses on regeneration medium containing 2.2 mg l−1 BAP and 0.01 mg l−1 NAA after the treatment with a high concentration of 2,4 D for a limited period of time. A morphological and RAPD analysis confirmed a hybrid nature of the regenerated plants.






Similar content being viewed by others
Abbreviations
- 2,4 D:
-
2,4-Dichlorophenoxyacetic acid
- ACO:
-
Aconitase
- ACP:
-
Acid Phosphatase
- BAP:
-
6-Benzylaminopurine
- IAA:
-
3-Indolylacetic acid
- IBA:
-
4-(3-Indolyl)butanoic acid
- MDH:
-
Malate Dehydrogenase
- NAA:
-
1-Naphthylacetic acid
- PCR:
-
Polymerase Chain Reaction
- PEG:
-
Polyethylene Glycol
- PGD:
-
6-Phosphogluconate Dehydrogenase
- PGM:
-
Phosphogluco Mutase
- PHI:
-
Phosphohexose Isomerase
- RAPD:
-
Random Amplified Polymorphic DNA
References
Aslane-Chanabe C (1991) Regeneration de plantes a partir de protoplastes chez le genre Helianthus et hybridation somatique entre le tournesol cultive et les tournesols sauvages. Doctoral Thesis, INP, Toulouse, France
Atlagic J, Dozet B, Skoric D (1995) Meiosis and pollen grain viability in Helianthus mollis, H. salicifolius, H. maximiliani and their F1 hybrids with cultivated sunflower. Euphytica 81:259–263
Barth S, Voeste D, Wingender R, Schnabl H (1993) Somatic hybrids of sunflower (Helianthus annuus L.) identified at the callus by isozyme analysis. Bot Acta 106:100–102
Bazzalo ME, Dimarco P, Martinez F, Daleo GR (1991) Indictors of resistance of sunflower plant to basal rot (Sclerotinia sclerotiorum): symptological, biochemical, anatomical and morphological characters of the host. Euphytica 57:195–205
Binsfeld P, Schnabl H (2002) Molecular and cytogenetic constitution of plants obtained via two different somatic hybridization methods. Plant Cell Rep 21:58–62
Burrus M, Chanabe C, Alibert G, Bidney D (1991) Regeneration of fertile plants from protoplasts of sunflower (Helianthus annuus L.). Plant Cell Rep 10:161–166
Castano F, Hemery-Tardin MC, Tourvieille de Labrouhe D, Vear F (1992) The inheritance and biochemistry of resistance to Sclerotinia sclerotiorum leaf infections in sunflower (Helianthus anuus L.). Euphytica 58:209–219
Chanabe C, Burrus M, Alibert G (1989) Factors affecting the improvement of colony formation from sunflower protoplast. Plant Sci 64:125–132
Collonier C, Fock I, Daunay MC, Servaes A, Vedel F, Siljak-Yakovljev S, Souvannavong V, Sihachackr D (2003) Somatic hybrids between Solanum melongena and S. sismbrifoilium, as a useful source of resistance against bacterial and fungal wilts. Plant Sci 164(5):849–861
Davey MR, Anthony P, Power JB, Lowe KC (2005) Plant protoplasts: status and biotechnological perspectives. Biotechnol Adv 23:131–171
Fischer C, Klethi P, Hahne G (1992) Protoplasts from cotyledon and hypocotyl of sunflower (Helianthus annuus L.): shoot regeneration and seed production. Plant Cell Rep 11:623–636
Forsberg J, Lagercrantz U, Glimelius K (1998) Comparison of UV light, X-ray and restriction enzyme treatment as tools in production of asymmetric somatic hybrids between Brassica napus and Arabidopsis thaliana. Theor Appl Genet 96:1178–1185
Furuta H, Shinoyama H, Nomura Y, Maeda M, Makara K (2004) Production of intergeneric somatic hybrids of chrysanthemum (Dendranthema x grandiflorum (Ramat.)) Kitamura) and wormwood (Artemisia sieversiana J.F.Ehrh. ex. Willd) wild rust (Puccinia horiana Hening) resistance by electrofusion of protoplasts. Plant Sci 166:695–702
Gentzbittel L, Zhang G, Vear F, Griveau Y, Nicolas P (1994) RFLP studies of genetic relationships among inbred lines of cultivated sunflower (Helianthus annuus L.): evidence for distinct restorer and maintainer germplasm pools. Theor Appl Genet 89:19–425
Hemery-Tardin M, Tourvieille D, Vear F (1998) Effect of infection by Sclerotinia spp. on the phenolic metabolism of sunflower capitula and leaves. Helia 29:19–32
Henn HJ, Steiner R, Wingender R, Schnabl H (1997) Wild type sunflower clones: source of resistance against Sclerotinia sclerotiorum (Lib.) de Bary stem infection. Angew Bot 71:5–9
Henn HJ, Wingender R, Schnabl H (1998a) Regeneration of fertile interspecific hybrids from protoplast fusions between Helianthus annuus L. and wild Helianthus species. Plant Cell Rep 18:220–224
Henn HJ, Wingender R, Schnabl H (1998b) Regeneration of fertile plants from Helianthus nuttallii T&G and Helianthus giganteus L. mesophyll protoplast. Plant Cell Rep 18:288–291
Krasnyanski S, Polgar Z, Nemeth G, Menczel L (1992) Plant regeneration from callus and protoplast cultures of Helianthus giganteus L. Plant Cell Rep 11:7–10
Krasnyanski S, Menczel L (1993) Somatic embryogenesis and plant regeneration from hypocotyls protoplast of sunflower (Helianthus annuus L.). Plant Cell Rep 12:260–263
Krasnyanski S, Menczel L (1995) Production of fertile somatic hybrid plants of sunflower and Helianthus giganteus L. by protoplast fusion. Plant Cell Rep 14:232–235
Liu JH, Dixelius C, Eriksson I., Glimelius K (1995) Brassica napus (+) B. tourneforti, a somatic hybrid containing traits of agronomic importance for rapeseed breeding. Plant Sci 109(1):75–86
Masirevic S, Gulya TJ (1992) Sclerotinia and Phomopsis—two devastating sunflower pathogens. Field Crops Res 30:271–300
Murashige T, Skoog F (1962) A revised medium for growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Paterson KE, Everett NP (1985) Regeneration of Helianthus annuus inbred plants from callus. Plant Sci 42:125–132
Prats E, Bazzalo ME, Leon A, Jorrin JV (2003) Accumulation of soluble phenolic compounds in sunflower capitula correlates with resistance to Sclerotinia sclerotiorum. Euphytica 132:321–329
Robert N, Vear F, Tourvieille de Labrouche D (1987) L'heredite de la resistance au Sclerotinia sclerotiorum (Lib.) de Bary chez le tournesol. Etude des reaction a deux test myceliens. Agronomie 4:423–429
Roenicke S, Hahn V, Horn R, Groene I, Brahm L, Schnabl H, Friedt W (2004) Interspecific hybrids of sunflower as a source of Sclerotinia resistance. Plant Breed 123:152–157
Schmitz P, Schnabl H (1989) Regeneration and evaluation of protoplasts from mesophyll, hypocotyl and petioles from Helianthus annuus L. J Plant Physiol 135:223–227
Shillito RD, Paszkowski J, Potrykus I (1983) Agarose plating and a bead type culture technique enable and stimulate development of protoplast derivated colonies in a number of plant species. Plant Cell Rep 2:244–247
Skoric D, Rajcan I (1992) Breeding for Sclerotinia resistance in sunflower. In: Proceedings of the 13th International Sunflower Conference, Pisa, Italy, pp. 1257–1262
Sossey-Alaoui K, Serieys H, Tersac M, Lambert P, Schilling E, Griveau Y, Kaan F, Berville A (1998) Evidence for several genomes in Helianthus. Theor Appl Genet 97:422–430
Stuber CW, Wendel JF, Goodman MM, Smith JSC (1988) Techniques and scoring procedures for starch gel electrophoresis of enzymes from maize (Zea mays L.). Technical bulletin 286, North Carolina agricultural research service, pp 95
Taski K, Vasic D (2005) Different sterilization methods for overcoming internal bacterial infection in sunflower seeds. Proc Nat Sci, Matica Srpska 109:59–64
Trabace T, Fiore MC, D'Ambrosio C, Vanadia S, Sunseri F (1996) Sunflower cytoplasmic hybrids revealed by PCR assay using male sterility as selectable marker. J Genet Breed 50:29–34
Trabace T, Vischi M, Fiore MC, Sunseri F, Vanadia S, Marchetti S, Olivieri AM (1995) Plant regeneration from hypocotyl protoplast in sunflower (Helianthus annuus L). J Genet Breed 49:51–54
Valkonen JPT, Rokka V-M (1998) Combination and expression of two virus resistance mechanisms in interspecific somatic hybrids of potato. Plant Sci 131(1):85–94
Vasic D, Alibert G, Skoric D (2001a) Protocol for efficient repetitive and secondary somatic embryogenesis in the Helianthus maximiliani (Schrader). Plant Cell Rep 20:121–125
Vasic D, Skoric D, Gilbert A, Miklic V (2001b) Micropropagation of Helianthus maximiliani (Schrader) by shoot apex culture. Helia 24:63–68
Vasic D, Skoric D, Taski K, Stosic Lj (2002) Use of oxalic acid for screening intact sunflower plants for resistance to Sclerotinia in vitro. Helia 36:145–152
Vasic D (2003) Sunflower somatic hybridization. Andrejevic Endowment, Belgrade
Vlahova M, Hinnisdaels S, Frulleux F, Claeys M, Atanassov A, Jacobs M (1997). UV irradiation as a tool for obtaining asymmetric somatic hybrids between Nicotiana plumbaginifolia and Lycopersicon esculentum. Theor Appl Genet 94:184–191
Waara S, Glimelius K (1995) The potential of somatic hybridization in crop breeding. Euphytica 85:217–255
Wingender R, Henn HJ, Barth S, Voeste D, Machlab H, Schnabl H (1996) A regeneration protocol for sunflower (Helianthus annuus L.) protoplasts. Plant Cell Rep 15:742–745
Acknowledgements
This work was supported by the Serbian Ministry of Science, Technology and Development as a part of the project BTR.5.02.0401.B.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Atanassov
Rights and permissions
About this article
Cite this article
Taski-Ajdukovic, K., Vasic, D. & Nagl, N. Regeneration of interspecific somatic hybrids between Helianthus annuus L. and Helianthus maximiliani (Schrader) via protoplast electrofusion. Plant Cell Rep 25, 698–704 (2006). https://doi.org/10.1007/s00299-006-0134-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-006-0134-5