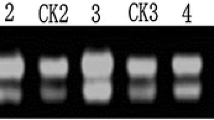
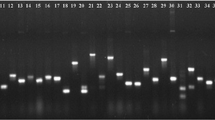
Abstract
Mycosphaerella fijiensis, a hemibiotrophic fungus, is the causal agent of black leaf streak disease, the most serious foliar disease of bananas and plantains. To analyze the compatible interaction of M. fijiensis with Musa spp., a suppression subtractive hybridization (SSH) cDNA library was constructed to identify transcripts induced at late stages of infection in the host and the pathogen. In addition, a full-length cDNA library was created from the same mRNA starting material as the SSH library. The SSH procedure was effective in identifying specific genes predicted to be involved in plant–fungal interactions and new information was obtained mainly about genes and pathways activated in the plant. Several plant genes predicted to be involved in the synthesis of phenylpropanoids and detoxification compounds were identified, as well as pathogenesis-related proteins that could be involved in the plant response against M. fijiensis infection. At late stages of infection, jasmonic acid and ethylene signaling transduction pathways appear to be active, which corresponds with the necrotrophic life style of M. fijiensis. Quantitative PCR experiments revealed that antifungal genes encoding PR proteins and GDSL-like lipase are only transiently induced 30 days post inoculation (dpi), indicating that the fungus is probably actively repressing plant defense. The only fungal gene found was induced 37 dpi and encodes UDP-glucose pyrophosphorylase, an enzyme involved in the biosynthesis of trehalose. Trehalose biosynthesis was probably induced in response to prior activation of plant antifungal genes and may act as an osmoprotectant against membrane damage.




Similar content being viewed by others
Abbreviations
- BLSD:
-
Black leaf streak disease
- CTAB:
-
Cetyl trimethyl ammonium bromide
- Dpi:
-
Days post inoculation
- EST:
-
Expressed sequence tag
- ET:
-
Ethylene
- FA:
-
Fatty acid
- HCN:
-
Hydrogen cyanide
- Hpi:
-
Hours post inoculation
- INIBAP:
-
International network for the improvement of banana and plantain
- ITS:
-
Internal transcribed spacer
- JA:
-
Jasmonate
- JA-Ile:
-
Jasmonyl-isoleucine
- NCBI:
-
National Center for Biotechnology Information
- NR:
-
Non-redundant
- PCR:
-
Polymerase chain reaction
- PDB:
-
Potato dextrose broth
- PR:
-
Pathogenesis related
- qPCR:
-
Quantitative polymerase chain reaction
- RT:
-
Reverse transcriptase
- SA:
-
Salicylic acid
- SSH:
-
Suppression subtractive hybridization
References
Abad LR, D’Urzo MP, Liu D, Narasimhan ML, Reuveni M, Zhu JK, Niu X, Singh NK, Hasegawa PM, Bressan RA (1996) Antifungal activity of tobacco osmotin has specificity and involves plasma membrane permeabilization. Plant Sci 118:11–23
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Alvarado Y, Leiva M, Rodríguez MA, Acosta M, Cruz M, Portal N, Kosky R, García L, Bermúdez I, Padrón J (2003) Early evaluation of black leaf streak resistance by using mycelia suspensions of Mycosphaerella fijiensis. In: Jacome L, Lepoivre P, Marin D, Ortiz R, Romero R, Escalant JV (eds) Mycosphaerella leaf spot diseases of bananas: present status and outlook. Proceedings of the second international workshop on Mycosphaerella leaf spot diseases, San José, pp 169–175
Antonini E, Brunori M, Greenwood C, Malmstrom BG, Rotilio GC (1971) The interaction of cyanide with cytochrome oxidase. Eur J Biochem 23:396–400
Azhar M, Heslop-Harrison JS (2008) Genomes, diversity and resistance gene analogues in Musa species. Cytogenet Genome Res 121:59–66
Bennett M, Mehta M, Grant M (2005) Biophoton imaging: a non-destructive method for assaying R gene responses. Mol Plant–Microbe Interact 18:95–102
Bormann C, Baier D, Horr I, Raps C, Berger J, Jung G, Schwarz H (1999) Characterization of a novel, antifungal, chitin-binding protein from Streptomyces tendae Tu901 that interferes with growth polarity. J Bacteriol 181:7421–7429
Bostock RM (2005) Signal crosstalk and induced resistance: straddling the line between cost and benefit. Annu Rev Phytopathol 43:545–580
Busogoro JP, Etamé JJ, Hareliman G, Lognay G, Messiaen J, Lepoivre P, van Cutsem P (2004) Experimental evidence for the action of M. fijiensis toxins on banana photosynthetic apparatus. In: Jain SM, Swennen R (eds) Banana improvement: cellular, molecular biology, and induced mutations. Science Publishers Inc, Enfield, pp 171–181
Carlier J, Lebrun MH, Zapater MF, Dubois C, Mourichon X (1996) Genetic structure of the global population of banana black leaf streak fungus, Mycosphaerella fijiensis. Mol Ecol 5:499–510
Chen TH, Murata N (2002) Enhancement of tolerance of abiotic stress by metabolic engineering of betaines and other compatible solutes. Curr Opin Plant Biol 5:250–257
Cho Y, Hou S, Zhong S (2008) Analysis of expressed sequence tags from the fungal banana pathogen Mycosphaerella fijiensis. Open Mycol J 2:61–73
Cruz-Cruz CA, Ramírez-Tec G, García-Sosa K, Escalante-Erosa F, Hill L, Osbourn AE, Peña-Rodríguez LM (2010) Phytoanticipins from banana (Musa acuminata cv. Grande Naine) plants, with antifungal activity against Mycosphaerella fijiensis, the causal agent of black sigatoka. Eur J Plant Pathol 126:459–463
Davidson RM, Reeves PA, Manosalva PM, Leach JE (2009) Germins: a diverse protein family important for crop improvement. Plant Sci 177:499–510
Diatchenko L, Lau YFC, Campbell AP (1996) Suppression subtractive hybridization: a method for generating differentially regulated or tissue-specific cDNA probes and libraries. Proc Natl Acad Sci USA 93:6025–6030
Dixon RA, Achnine L, Kota P, Chang-Jun L, Reddy MSS, Wang L (2002) The phenylpropanoid pathway and plant defense a genomics perspective. Mol Plant Pathol 3:371–390
Doehlemann G, Berndt P, Hahn M (2006) Different signalling pathways involving a G (alpha) protein, cAMP and a MAP kinase control germination of Botrytis cinerea conidia. Mol Microbiol 59:821–835
Dunwell JM, Gibbings JG, Mahmood T, Saqlan-Naqvi SM (2008) Germin and germin-like proteins: evolution, structure, and function. Crit Rev Plant Sci 27:342–375
Edreva A (2005) Pathogenesis-related proteins: research progress in the last 15 years. Gen Appl Plant Physiol 31:105–124
Elbein AD, Pan YT, Pastuszak I, Carroll D (2003) New insights on trehalose: a multifunctional molecule. Glycobiology 13:17R–27R
Endah R, Beyene G, Kiggundu A, van den Berg N, Schlüter U, Kunert K, Chikwamba R (2008) Elicitor and fusarium-induced expression of NPR1-like genes in banana. Plant Physiol Biochem 46:1007–1014
Fahrendorf T, Ni W, Shorrosh BS, Dixon RA (1995) Stress responses in alfalfa (Medicago sativa L.) XIX. Transcriptional activation of oxidative pentose phosphate pathway genes at the onset of the isoflavonoid phytoalexin response. Plant Mol Biol 28:885–900
Ferreira R, Monteiro S, Freitas R, Santos C, Borges A, Chen Z, Batista L, Duarte J, Teixeira A (2007) The role of plant defence proteins in fungal pathogenesis. Mol Plant Pathol 8:677–700
Foster AJ, Jenkinson JM, Talbot NJ (2003) Trehalose synthesis and metabolism are required at different stages of plant infection by Magnaporthe grisea. EMBO J 22:225–235
Frendo P, Harrison J, Norman C, Hernández-Jiménez MJ, Van de Sype G, Gilabert A, Puppo A (2005) Glutathione and homoglutathione play a critical role in the nodulation process of Medicago truncatula. Mol Plant–Microbe Interact 18:168–174
Gao XP, Wang XF, Lu YF, Zhang LY, Shen YY, Liang Z, Zhang DP (2004) Jasmonic acid is involved in the water-stress-induced betaine accumulation in pear leaves. Plant Cell Environ 27:497–507
Glazebrook J (1999) Genes controlling expression of defence responses in Arabidopsis. Curr Opin Plant Biol 2:280–286
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Ann Rev Phytopathol 43:205–227
Grant M, Lamb C (2006) Systemic immunity. Curr Opin Plant Biol 9:414–420
Gundlach H, Müller MJ, Kutchan TM, Zenk MH (1992) Jasmonic acid is a signal transducer in elicitor-induced plant cell cultures. Proc Natl Acad Sci USA 89:2389–2393
Gurskaya NG, Diachenko L, Chenchik A, Siebert PD, Khaspekov GL, Lukyanov K, Vagner LL, Ermolaeva OD, Lukyanov S, Sverdlov ED (1996) The equalizing cDNA subtraction based on selective suppression of polymerase chain reaction: cloning of the Jurkat cells transcripts induced by phytohemaglutinin and phorbol 12-myristate 13-acetate. Anal Biochem 240:90–97
Hammond-Kosack KE, Kanyuka K (2007) Resistance genes (R genes) in plants. Encyclopedia of life sciences. John Wiley and Sons, ltd, London
Harelimana G, Lepoivre P, Jijakli H, Mourichon X (1997) Use of Mycosphaerella fijiensis toxins for selection of banana cultivars resistant to black leaf streak. Euphytica 96:125–128
Hayden HL, Carlier J, Aitken EAB (2003) The genetic structure of Mycosphaerella fijiensis from Australia, Papua New Guinea and the Pacific Islands. Plant Pathol 52:703–712
Hidalgo W, Duque L, Saez J, Arango R, Gil J, Rojano B, Schneider B, Otálvaro F (2009) Structure-activity relationship in the interaction of substituted perinaphthenones with Mycosphaerella fijiensis. J Agric Food Chem 57:7417–7421
Horry JP, Jay M (1990) An evolutionary background of bananas as deduced from flavonoid diversification. In: Jarret RL (ed) Identification of genetic diversity in the genus Musa. INIBAP, Montpellier, pp 41–45
Hoss R, Helbig J, Bochow H (2000) Function of host and fungal metabolites in resistance response of banana and plantain in the black sigatoka disease pathosystem (Musa spp.–Mycosphaerella fijiensis). J Phytopathol 148:387–394
Hrazdina G, Jensen RA (1992) Spatial organization of enzymes in plant metabolic pathways. Annu Rev Plant Phys 43:241–267
Huang X, Madan A (1999) CAP3: a DIVA sequence assembly program. Genome Res 9:868–877
Islas-Flores I, Peraza-Echevarria L, Canto-Canché B, Rodríguez-García CM (2006) Extraction of high-quality, melanin-free RNA from Mycosphaerella fijiensis for cDNA preparation. Mol Biotechnol 34:45–50
Johanson A, Jeger MJ (1993) Use of PCR for detection of Mycosphaerella fijiensis and M. musicola, the causal agents of sigatoka leaf spots on banana and plantain. Mycol Res 97:670–674
Kachroo A, Venugopal SC, Lapchyk L, Falcone D, Hildebrand D, Kachroo P (2004) Oleic acid levels regulated by glycerolipid metabolism modulate defense gene expression in Arabidopsis. Proc Natl Acad Sci USA 101:5152–5157
Kader JC (1996) Lipid-transfer proteins in plants. Annu Rev Plant Phys 47:627–654
Khayat E (2004) Discovery of functional genes in the Musa genome. In: Jain SM, Swennen R (eds) Banana improvement: cellular, molecular biology, and induced mutations. Science Publishers Inc, Enfield, pp 321–329
Knaggs AR (2003) The biosynthesis of shikimate metabolites. Nat Prod Rep 20:119–136
Kombrink E, Somssich IE (1997) Pathogenesis-related proteins and plant defense. In: Carrol GC, Tudzynski P (eds) The mycota V part A, plant relationships. Springer-Verlag, Berlin, pp 107–128
Kwon SJ, Jin HC, Lee S, Nam MH, Chung JH, Kwon SI, Ryu C, Park OK (2009) GDSL lipase-like 1 regulates systemic resistance associated with ethylene signaling in Arabidopsis. Plant J 58:235–245
Lepoivre P, Busogoro JP, Etame JJ, El Hadrami A, Carlier J, Harelimana G, Mourichon X, Panis B, Riveros AS, Salle G, Strosse H, Swennen R (2003) Banana–Mycosphaerella fijiensis interactions. In: Jacome L, Lepoivre P, Marin D, Ortiz R, Romero R, Escalant JV (eds) Mycosphaerella leaf spot diseases of bananas: present status and outlook. Proceedings of the second international workshop on Mycosphaerella leaf spot diseases, San José, pp 151–159
Li C, Liu G, Xu C, Lee GI, Bauer P, Ling HQ, Ganal MW, Howe GA (2003) The tomato suppressor of prosystemin-mediated response2 gene encodes a fatty acid desaturase required for the biosynthesis of jasmonic acid and the production of a systemic wound signal for defense gene expression. Plant Cell 15:1646–1661
Liao Z, Chen M, Gong Y, Tang F, Sun X, Tang K (2004) Rapid isolation of high-quality total RNA from Taxus and Ginkgo. Prep Biochem Biotechnol 34:209–214
Liebman JF, Greenberg A (1988) Mechanistic principles of enzyme activity. VCH Publishers, New York
Liu X, Huang B, Lin J, Fei J, Chen Z, Pang P, Sun X, Tang T (2006) A novel pathogenesis-related protein (SsPR10) from Solanum surattense with ribonucleolytic and antimicrobial activity is stress and pathogen-inducible. J Plant Physiol 163:546–556
Livingstone JR, Maruo T, Yoshida I, Tarui Y, Hirooka K, Yamamoto Y, Tsutui N, Hirasawa E (2003) Purification and properties of betaine aldehyde dehydrogenase from Avena sativa. J Plant Res 116:133–140
Lorenzo O, Solano R (2005) Molecular players regulating the jasmonate signalling network. Curr Opin Plant Biol 8:532–540
Lorito M, Woo SL, D’Ambrosio M, Harman GE, Hayes CK, Kubicek CP, Scala F (1996) Synergistic interaction between cell wall degrading enzymes and membrane affecting compounds. Mol Plant–Microbe Interact 9:206–313
Lowe RG, Lord M, Rybak K, Trengove RD, Oliver RP, Solomon PS (2009) Trehalose biosynthesis is involved in sporulation of Stagonospora nodorum. Fungal Genet Biol 46:381–389
Marín DH, Romero RA, Guzmán M, Sutton TB (2003) Black sigatoka: an increasing threat to banana cultivation. Plant Dis 87:208–222
Marrs KA (1996) The functions and regulation of glutathione S-transferases in plants. Annu Rev Plant Phys 47:127–158
May MJ, Vernoux T, Leaver C, Van Montague M, Inze D (1998) Glutathione homeostasis in plants: implications for environmental sensing and plant development. J Exp Bot 49:649–667
McCue KF, Conn EE (1989) Induction of 3-deoxy-D-arabino-heptulosonate 7-phosphate synthase by fungal elicitor in cultures of Petroselinum crispum. Proc Natl Acad Sci USA 86:7374–7377
McDowell JM, Dangl JL (2000) Signal transduction in the plant immune response. Trends Biochem Sci 25:79–82
Meredith DS, Lawrence JS (1969) Black leaf streak disease of bananas (Mycosphaerella fijiensis): symptoms of disease in Hawaii, and notes on conidial state of causal fungus. Trans Br Mycol Soc 52:459–476
Miller RNG, Bertioli DJ, Baurens FC, Santos CMR, Alves PC, Martins N, Togawa RC, Souza M, Pappas G Jr (2008) Analysis of non-TIR NBS-LRR resistance gene analogs in Musa acuminata Colla: isolation, RFLP marker development, and physical mapping. BMC Plant Biol 8:15
Mobambo KN, Gauhl F, Swennen R, Pasberg-Gauhl C (1996) Assessment of the cropping cycle effects of black leaf streak severity and yield decline of plantain and plantain hybrids. Int J Pest Manag 42:1–7
Molina A, Segura A, Garcia-Olmedo F (1993) Lipid transfer proteins (nsLTPs) from barley and maize leaves are potent inhibitors of bacterial and fungal plant pathogens. FEBS Lett 316:119–122
Mourichon X, Peter D, Zapater MF (1987) Inoculation experimentale de M. fijiensis Morelet sur jeunes plantules de bananier issues de culture in vitro. Fruits 42:195–198
Neuefeind T, Reinemer P, Bieseler B (1997) Plant glutathione S-transferases and herbicide detoxification. Biol Chem 378:199–205
Niki T, Mitsuhara I, Seo S, Ohtsubo N, Ohashi Y (1998) Antagonistic effect of salicylic acid and jasmonic acid on the expression of pathogenesis-related (PR) protein genes in wounded mature tobacco leaves. Plant Cell Physiol 39:500–507
Noctor G, Gomez L, Vanacker H, Foyer CH (2002) Interactions between biosynthesis, compartmentation and transport in the control of glutathione homeostasis and signalling. J Exp Bot 53:1283–1304
Oh IS, Park AR, Bae MS, Kwon SJ, Kim YS, Lee JE, Kang NY, Lee S, Cheong H, Park OK (2005) Secretome analysis reveals an Arabidopsis lipase involved in defense against Alternaria brassicicola. Plant Cell 17:2832–2847
Otálvaro F, Echeverri F, Quiñones W, Torres F, Schneider B (2002) Correlation between phenylphenalenone phytoalexins and phytopathological properties in Musa and the role of a dihydrophenylphenalene triol. Molecules 7:331–340
Otálvaro F, Nanclares J, Vásquez LE, Quiñones W, Echeverri F, Arango R, Schneider B (2007) Phenalenone-type compounds from Musa acuminata var. ‘Yangambi km5′ (AAA) and their activity against Mycosphaerella fijiensis. J Nat Prod 70:887–890
Pasberg-Gauhl C, Gauhl F, Jones D (2000) Fungal disease of foliage. Sigatoka leaf spots. Black leaf streak. Distribution and economic importance. In: Jones DR (ed) Diseases of banana, abacá and enset. CABI Publishing, Wallingford, pp 37–44
Pei X, Li S, Jiang Y, Zhang Y, Wang Z, Jia S (2007) Isolation, characterization and phylogenetic analysis of the resistance gene analogues (RGAs) in banana (Musa spp.). Plant Sci 172:1166–1174
Peiser G, Wang TT, Hoffman NE, Yang SF, Liu HW, Walsh CT (1984) Formation of cyanide from carbon 1 of 1-aminocyclopropane-1-carboxylic acid during its conversion to ethylene. Proc Natl Acad Sci USA 81:3059–3063
Peraza-Echeverria S, James-Kay A, Canto-Canché B, Castillo-Castro E (2007) Structural and phylogenetic analysis of Pto-type disease resistance gene candidates in banana. Mol Genet Genomics 278:443–453
Piotrowski M, Volmer JJ (2006) Cyanide metabolism in higher plants: cyanoalanine hydratase is a NIT4 homolog. Plant Mol Biol 61:111–122
Pozo MJ, van Loon LC, Pieterse CMJ (2005) Jasmonates signals in plant–microbe interactions. J Plant Growth Regul 23:211–222
Pyee J, Yu H, Kolattukudy PE (1994) Identification of a lipid transfer protein as a major protein in the surface wax of broccoli (Brassica oleracea) leaves. Arch Biochem Biophys 311:460–468
Quiñones W, Escobar G, Echeverri F, Torres F, Rosero Y, Arango V, Cardona G, Gallego A (2000) Synthesis and antifungal activity of Musa phytoalexins and structural analogs. Molecules 5:974–980
Rasmussen S, Dixon RA (1999) Transgene-mediated and elicitor-induced perturbation of metabolic channeling at the entry point into the phenylpropanoid pathway. Plant Cell 11:1537–1552
Ratnakumar S, Tunnacliffe A (2006) Intracellular trehalose is neither necessary nor sufficient for desiccation tolerance in yeast. FEMS Yeast Res 6:902–913
Reymond P, Farmer EE (1998) Jasmonate and salicylate as global signals for defense gene expression. Curr Opin Plant Biol 1:404–411
Roberts WK, Selitrennikoff CP (1990) Zeamatin, an antifungal protein from maize with membrane permeabilizing activity. J Gen Microbiol 136:1771–1778
Rozen S, Skaletsky HJ (2000) Primer3 on the WWW for general users and for biologist programmers. In: Krawetz S, Misener S (eds) Bioinformatics methods and protocols: methods in molecular biology. Humana Press, Totowa, pp 365–386
Sánchez-Rodríguez A, Portal O, Rojas LE, Ocaña B, Mendoza-Rodríguez M, Acosta-Suárez M, Jiménez E, Höfte M (2008) An efficient method for the extraction of high-quality fungal total RNA to study the Mycosphaerella fijiensis–Musa spp. interaction. Mol Biotechnol 40:299–305
Schenk PM, Kazan K, Wilson I, Anderson JP, Richmond T, Somerville SC, Manners JM (2000) Coordinated plant defense responses in Arabidopsis revealed by microarray analysis. Proc Natl Acad Sci USA 97:11655–11660
Segura A, Moreno F, García-Olmedo F (1993) Purification and antipathogenic activity of lipid transfer proteins (LTPs) from the leaves of Arabidopsis and spinach. FEBS Lett 332:243–246
Sels J, Mathys J, De Coninck B, Cammue BPA, De Bolle M (2008) Plant pathogenesis-related (PR) proteins: a focus on PR peptides. Plant Physiol Biochem 46:941–950
Seo HS, Song JT, Cheong JJ, Lee YH, Lee YW, Hwang I, Lee JS, Choi YD (2001) Jasmonic Itranferase: a key enzyme for jasmonate-regulated plant responses. Proc Natl Acad Sci USA 98:4788–4793
Somssich IK, Halbrock K (1998) Pathogen defense in plants a paradigm of biological complexity. Trends Plant Sci 3:86–90
Staswick PE (2008) JAZing up jasmonate signaling. Trends Plant Sci 13:66–71
Staswick PE, Tiryaki I (2004) The oxylipin signal jasmonic acid is activated by an enzyme that conjugates it to isoleucine in Arabidopsis. Plant Cell 16:2117–2127
Stergiopoulos I, van den Burg HA, Okmen B, Beenen HG, van Liere S, Kema GH, de Wit PJ (2010) Tomato Cf resistance proteins mediate recognition of cognate homologous effectors from fungi pathogenic on dicots and monocots. Proc Natl Acad Sci USA 107:7610–7615
Sterk P, Booij H, Schellekens GA, Van Kammen A, De Vries SC (1991) Cell-specific expression of the carrot EP2 lipid transfer protein. Plant Cell 3:907–921
Strosse H, Van den Houwe I, Panis B (2004) Banana cell and tissue culture-review. In: Jain SM, Swennen R (eds) Banana improvement: cellular, molecular biology, and induced mutations. Science Publishers Inc, Enfield, pp 1–12
Takayama K, Armstrong EL (1976) Isolation, characterization, and function of 6 mycolyl-6-acetyltrehalose in the H37Ra strain of Myocobacterium tuberculosis. Biochemistry 15:441–447
Theis T, Stahl U (2004) Antifungal proteins: targets, mechanisms and prospective applications. Cell Mol Life Sci 61:437–455
Thevelein JM (1984) Regulation of trehalose mobilization in fungi. Microb Rev 48:42–59
Thomma BPHJ, Penninckx IAMA, Broekaert WF, Cammue BPA (2001) The complexity of disease signaling in Arabidopsis. Curr Opin Immunol 13:63–68
Umemura K, Ogawa N, Shimura M, Koga J, Usami H, Kono T (2003) Possible role of phytocassane, rice phytoalexin, in disease resistance of rice against the blast fungus Magnaporthe grisea. Biosci Biotechnol Biochem 67:899–902
van Etten HD, Mansfield JW, Bailey JA, Farmer EE (1994) Two classes of plant antibiotics: phytoalexins versus phytoanticipins. Plant Cell 6:1191–1192
van Loon LC (2000) Systemic induced resistance. In: Slusarenko AJ, Fraser RSS, van Loon LC (eds) Mechanisms of resistance to plant diseases. Kluwer Academic Publishers, Dordrecht, pp 521–574
van Loon LC, Rep M, Pieterse CMJ (2006) Significance of inducible defense-related proteins in infected plants. Ann Rev Phytopathol 44:135–162
Vanacker H, Carver TLW, Foyer CH (2000) Early accumulation in mesophyll cells leads to induction of glutathione during the hyper-sensitive response in the barley-powdery mildew interaction. Plant Physiol 123:1289–1300
Ververidis F, Trantas E, Douglas C, Vollmer G, Kretzschmar G, Panopoulos N (2007) Biotechnology of flavonoids and other phenylpropanoid-derived natural products. Part II: Reconstruction of multienzyme pathways in plants and microbes. Biotechnol J 2:1235–1249
von Dahl CC, Winz R, Halitschke R, Kühnemann F, Gase K, Baldwin IT (2007) Tuning the herbivore-induced ethylene burst: the role of transcripts accumulation and ethylene perception in Nicotiana attenuata. Plant J 51:293–307
Wang Z, Thornton CR, Kersshaw MJ, Debao Li, Talbot NJ (2003) The glyoxylate cycle is required for temporal regulation of virulence by the plant pathogenic fungus Magnaporthe grisea. Mol Microbiol 47:1601–1612
Wang YD, Yuan YJ, Wu JC (2004) Induction studies of methyl jasmonate and salicylic acid on taxane production in suspension cultures of Taxus chinensis var. mairei. Biochem Eng J 19:259–265
Weber H (2002) Fatty acid-derived signals in plants. Trends Plant Sci 7:217–224
Wingate VPM, Lawton MA, Lamb CJ (1988) Glutathione causes a massive and selective induction of plant defense genes. Plant Physiol 87:206–210
Wuyts N, Lognay G, Verscheure M, Marlier M, De Waele D, Swennen R (2007) Potential physical and chemical barriers to infection by the burrowing nematode Radopholus similis in roots of susceptible and resistant banana (Musa spp.). Plant Pathol 56:878–890
Yaeno T, Matsuda O, Iba K (2004) Role of chloroplast trienoic fatty acids in plant disease defense response. Plant J 40:931–941
Yang Q, Gong ZZ (2002) Purification and characterization of an ethylene-induced antifungal protein from leaves of guilder rose (Hydrangea macrophylla). Protein Expr Purif 24:76–82
Yang L, Guan T, Gerace L (1997) Integral membrane proteins of the nuclear envelope are dispersed throughout the endoplasmic reticulum during mitosis. J Cell Biol 137:1199–1210
Yip WK, Yang SF (1998) Ethylene biosynthesis in relation to cyanide metabolism. Bot Bull Acad Sin 39:1–7
Yun DJ, Zhao Y, Pardo JM, Narasimhan ML, Damsz B, Lee H, Abad LR, D’Urzo MP, Hasegawa PM, Bressan RA (1997) Stress proteins on the yeast cell surface determine resistance to osmotin, a plant antifungal protein. Proc Natl Acad Sci USA 94:7082–7087
Zhao JT, Huang X, Chen YP, Chen YF, Huang XL (2009) Molecular cloning and characterization of an ortholog of NPR1 gene from Dongguan Dajiao (Musa spp. ABB). Plant Mol Biol Rep 27:243–249
Acknowledgments
The authors thank Yelenys Alvarado-Capó (Instituto de Biotecnología de las Plantas, Cuba) for fruitful discussions concerning the design of the cDNA libraries construction. We are grateful to Luis Perez Vicente (INISAV, Cuba) and Joke Pannecoucque (Ghent University, Belgium) for critical comments on the manuscript. This research was performed in the frame of the Institutional University Cooperation Programme with Universidad Central “Marta Abreu” de Las Villas, supported by the Flemish Interuniversity Council (IUC UCLV/VLIR). This work also was supported in part by INIBAP (currently Bioversity International) through the project ‘Molecular analysis of Mycosphaerella fijiensis–banana interactions’ (LOA 2003-04). David De Vleesschauwer is indebted to the FWO-Vlaanderen for a postdoctoral fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Guiderdoni.
O. Portal and Y. Izquierdo contributed equally to the article.
A contribution to the Special Issue: Plant Biotechnology in Support of the Millennium Development Goals.
Rights and permissions
About this article
Cite this article
Portal, O., Izquierdo, Y., De Vleesschauwer, D. et al. Analysis of expressed sequence tags derived from a compatible Mycosphaerella fijiensis–banana interaction. Plant Cell Rep 30, 913–928 (2011). https://doi.org/10.1007/s00299-011-1008-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-011-1008-z