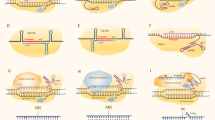
Abstract
Increasing crop production to meet the demands of a growing population depends largely on crop improvement through new plant-breeding techniques (NPBT) such as genome editing. CRISPR/Cas systems are NPBTs that enable efficient target-specific gene editing in crops, which is supposed to accelerate crop breeding in a way that is different from genetically modified (GM) technology. Herein, we review the applications of CRISPR/Cas systems in crop breeding focusing on crop domestication, heterosis, haploid induction, and synthetic biology, and summarize the screening methods of CRISPR/Cas-induced mutations in crops. We highlight the importance of molecular characterization of CRISPR/Cas-edited crops, and pay special attentions to emerging highly specific genome-editing tools such as base editors and prime editors. We also discuss future improvements of CRISPR/Cas systems for crop improvement.



Similar content being viewed by others
References
Ainley WM, Dent SL, Welter ME, Murray MG, Zeitler B, Amora R, Corbin DR, Miles RR, Arnold NL, Strange TL, Simpson MA (2013) Trait stacking via targeted genome editing. Plant Biotechnol J 11:1126–1134. https://doi.org/10.1111/pbi.12107
Ali Z, Shami A, Sedeek K, Kamel R, Alhabsi A, Tehseen M, Hassan N, Butt H, Kababji A, Hamdan SM, Mahfouz MM (2020) Fusion of the Cas9 endonuclease and the VirD2relaxase facilitates homology-directed repair forprecise genome engineering in rice. Commun Biol 3(1):44. https://doi.org/10.1038/s42003-020-0768
Aman R, Ali Z, Butt H, Mahas A, Aljedaani F, Khan MZ, Ding S, Mahfouz M (2018) RNA virus interference via CRISPR/Cas13a system in plants. Genome Biol 19:1. https://doi.org/10.1186/s13059-017-1381-1
Anzalone AZ, Randolph PB, Davis JR, Sousa AA, Koblan LW, Levy JM, Chen PJ, Wilson C, Newby GA, Raguram A, Liu DR (2019) Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 576:149–157. https://doi.org/10.1038/s41586-019-1711-4
Anzalone AZ, Koblan LW, Liu DR (2020) Genome editing with CRISPR-Cas nucleases, base editors, transposases and prime editors. Nat Biotechnol 38:824–844. https://doi.org/10.1038/s41587-020-0561-9
Araki M, Ishii T (2015) Towards social acceptance of plant breeding by genome editing. Trends Plant Sci 20:145–149. https://doi.org/10.1016/j.tplants.2015.01.010
Arndell T, Sharma N, Langridge P, Baumann U, Watson-Haigh NS, Whitford R (2019) gRNA validation for wheat genome editing with the CRISPR-Cas9 system. BMC Biotechnol 19:71. https://doi.org/10.1186/s12896-019-0565-z
Baltes NJ, Voytas DF (2015) Enabling plant synthetic biology through genome engineering. Trends Biotechnol 33:120–131. https://doi.org/10.1016/j.tibtech.2014.11.008
Birchler JA, Yao H, Chudalayandi S, Vaiman D, Veitia RA (2010) Heterosis. Plant Cell 22:2105–2122. https://doi.org/10.1105/tpc.110.076133
Biswas S, Li R, Yuan Z, Zhang D, Zhao X, Shi J (2019) Development of methods for effective identification of CRISPR/Cas9- induced indels in rice. Plant Cell Rep 38(4):503–510. https://doi.org/10.1007/s00299-019-02392-3
Biswas S, Li R, Hong J, Zhao X, Yuan Z, Zhang D, Shi J (2020a) Effective identification of CRISPR/Cas9-induced and naturally occurred mutations in rice using a multiplex ligation-dependent probe amplification-based method. Theor Appl Genet 133:2323–2334. https://doi.org/10.1007/s00122-020-03600-5
Biswas S, Tian J, Li R, Chen X, Luo Z, Chen M, Zhao X, Zhang D, Persson S, Yuan Z, Shi J (2020b) Investigation of CRISPR/Cas9-induced SD1 rice mutants highlights the importance of molecular characterization in plant molecular breeding. J Genet Genom. https://doi.org/10.1016/j.jgg.2020.04.004
Brauer EK, Balcerzak M, Rocheleau H, Leung W, Schernthaner J, Subramaniam R, Ouellet T (2020) Genome editing of a deoxynivalenol-induced transcription factor confers resistance to Fusarium graminearum in wheat. Mol Plant Microbe Interact 33(3):553–560. https://doi.org/10.1094/MPMI-11-19-0332-R
Butt H, Eid A, Momin AA, Bazin J, Crespi M, Arold ST, Mahfouz MM (2019) CRISPR directed evolution of the spliceosome for resistance to splicing inhibitors. Genome Biol 20:73. https://doi.org/10.1186/s13059-019-1680-9
Butt H, Rao GS, Sedeek K, Aman R, Mahfouz M (2020) Engineering herbicide resistance via prime editing in rice. Plant Biotechnol J 18:2370–2372. https://doi.org/10.1111/pbi.13399
Čermák T, Baltes NJ, Čegan R, Zhang Y, Voytas DF (2015) High-frequency, precise modification of the tomato genome. Genome Biol 16:232. https://doi.org/10.1186/s13059-015-0796-9
Chen K, Gao C (2013) TALENs: Customizable molecular DNA scissors for genome engineering of plants. J Genet Genomics 40:271–279. https://doi.org/10.1016/j.jgg.2013.03.009
Chen K, Wang Y, Zhang R, Zhang H, Gao C (2019) CRISPR/Cas genome editing and precision plant breeding in agriculture. Annu Rev Plant Biol 70:667–697. https://doi.org/10.1146/annurev-arplant-050718-100049
Curtin SJ, Zhang F, Sander JD, Haun WJ, Starker C, Baltes NJ, Reyon D, Dahlborg EJ, Goodwin MJ, Coffman AP, Dobbs D, Joung JK, Voytas DF, Stupar RM (2011) Targeted mutagenesis of duplicated genes in soybean with zinc-finger nucleases. Plant Physiol 156:466–473. https://doi.org/10.1104/pp.111.172981
Curtin SJ, Xiong Y, Michno JM, Campbell BW, Stec AO, Cermak T, Starker C, Voytas DF, Eamens AL, Stupar RM (2018) CRISPR/Cas9 and TALENs generate heritable mutations for genes involved in small RNA processing of Glycine max and Medicago truncatula. Plant Biotechnol J 16:1125–1137. https://doi.org/10.1111/pbi.12857
Do PT, Nguyen CX, Bui HT, Tran LTN, Stacey G, Gillman JD, Zhang ZJ, Stacey MG (2019) Demonstration of highly efficient dual gRNA CRISPR/Cas9 editing of the homeologous GmFAD2–1A and GmFAD2–1B genes to yield a high oleic, low linoleic and α-linolenic acid phenotype in soybean. BMC Plant Biol 19:311. https://doi.org/10.1186/s12870-019-1906-8
Doebley JF, Gaut BS, Smith BD (2006) The molecular genetics of crop domestication. Cell 127:1309–1321. https://doi.org/10.1016/j.cell.2006.12.006
Doench JG, Fusi N, Sullender M, Hegde M, Vaimberg EW, Donovan KF, Smith I, Tothova Z, Wilen C, Orcherd R, Virgin HW, Listgarten J, Root DE (2016) Optimized sgRNA design to maximize activity and minimize off-target effects of CRISPR-Cas9. Nat Biotechnol 34:184–191. https://doi.org/10.1038/nbt.3437
Dong OX, Yu S, Jain R, Zhang N, Duong PQ, Butler C, Li Y, Lipzen A, Martin JA, Barry KW, Schmutz J, Tian L, Ronald PC (2020) Marker-free carotenoid-enriched rice generated through targeted gene insertion using CRISPR-Cas9. Nat Commun 11:1178. https://doi.org/10.1038/s41467-020-14981-y
Doudna JA, Charpentier E (2014) The new frontier of genome engineering with CRISPR-Cas9. Science 346:1258096. https://doi.org/10.1126/science.1258096
Eckerstorfer MF, Engelhard M, Heissenberger A, Simon S, Teichmann H (2019) Plants developed by new genetic modification techniques-Comparison of existing regulatory frameworks in the EU and Non-EU countries. Front Bioeng Biotechnol 7:26. https://doi.org/10.3389/fbioe.2019.00026
El-Mounadi K, Morales-Floriano ML, Garcia-Ruiz H (2020) Principles, applications, and biosafety of plant genome editing using CRISPR-Cas9. Front Plant Sci 11:56. https://doi.org/10.3389/fpls.2020.00056
Eriksson D, Kershen D, Nepomuceno A, Pogson BJ, Prieto H, Purnhagen K, Smyth S, Wesseler J, Whelan A (2019) A comparison of the EU regulatory approach to directed mutagenesis with that of other jurisdictions, consequences for international trade and potential steps forward. New Phytol 222:1673–1684. https://doi.org/10.1111/nph.15627
Eş I, Gavahian M, Marti-Quijal FJ, Lorenzo JM, Khaneghah AM, Tsatsanis C, Kampranis SC, Barba FJ (2019) The application of the CRISPR-Cas9 genome editing machinery in food and agricultural science: Current status, future perspectives, and associated challenges. Biotechnol Adv 37:410–421. https://doi.org/10.1016/j.biotechadv.2019.02.006
Fauser F, Schiml S, Puchta H (2014) Both CRISPR/Cas-based nucleases and nickases can be used efficiently for genome engineering in Arabidopsis thaliana. Plant J 79:348–359. https://doi.org/10.1111/tpj.12554
Feng Z, Mao Y, Xu N, Zhang B, Wei P, Yang DL, Wang Z, Zhang Z, Zheng R, Yang L, Zeng L, Liu X, Zhu ZK (2014) Multigeneration analysis reveals the inheritance, specificity, and patterns of CRISPR/Cas-induced gene modifications in Arabidopsis. Proc Natl Acad Sci U S A 111:4632–4637. https://doi.org/10.1073/pnas.1400822111
Fernie AR, Yan J (2019) De novo domestication: an alternative route toward new crops for the future. Mol Plant 5:615–631. https://doi.org/10.1016/j.molp.2019.03.016
Friedrichs S, Takasu Y, Kearns P, Dagalliar B, Oshima R, Schofield J, Moreddu C (2019) Meeting report of the OECD conference on “genome editing: applications in agriculture-implications for health, environment and regulation.” Transgenic Res 28:419–463. https://doi.org/10.1007/s11248-019-00154-1
Fritsche S, Poovaiah C, MacRae E, Thorlby G (2018) A New Zealand perspective on the application and regulation of gene editing. Front Plant Sci 9:1323. https://doi.org/10.3389/fpls.2018.01323
Gao C (2021) Genome engineering for crop improvement and future agriculture. Cell 184:1621–1635. https://doi.org/10.1016/j.cell.2021.01.005
Gao W, Xu WT, Huang KL, Guo MZ, Luo YB (2018) Risk analysis for genome editing-derived food safety in China. Food Control 84:128–137. https://doi.org/10.1016/j.foodcont.2017.07.032
Gao H, Gadlage MJ, Lafitte HR, Lenderts B, Yang M, Schroder M, Farrell J, Snopek K, Peterson D, Feigenbutz L, Jones S, Clair GST, Rahe M, Sanyour-Doyel N, Peng C, Wang L, Young JK, Beatty M, Dahlke B, Hazebroek J, Greene TW, Cigan AM, Chilcoat ND, Meeley RB (2020) Superior field performance of waxy corn engineered using CRISPR–Cas9. Nat Biotechnol 38:579–581. https://doi.org/10.1038/s41587-020-0444-0
Garcia-Doval C, Jinek M (2017) Molecular architectures and mechanisms of Class 2 CRISPR-associated nucleases. Curr Opin Struc Biol 47:157–166. https://doi.org/10.1016/j.sbi.2017.10.015
Gaudelli NM, Komor AC, Rees HA, Packer MS, Badran AH, Bryson DI, Liu DR (2017) Programmable base editing of A•T to G•C in genomic DNA without DNA cleavage. Nature 551:464–471. https://doi.org/10.1038/nature24644
Gaudelli NM, Lam DK, Rees HA, Solá-Esteves NM, Barrera LA, Born DA, Edwards A, Gehrke JM, Lee SJ, Liquori AJ, Murray R, Packer MS, Rinaldi C, Slaymaker IM, Yen J, Young LE, Ciaramella G (2020) Directed evolution of adenine base editors with increased activity and therapeutic application. Nature Biotechnol 38:892–900. https://doi.org/10.1038/s41587-020-0491-6
Gomez MA, Lin ZD, Moll T, Chauhan RD, Hayden L, Renninger K, Beyene G, Taylor NJ, Carrington JC, Staskawicz BJ, Bart BS (2019) Simultaneous CRISPR/Cas9-mediated editing of cassava eIF4E isoforms nCBP-1 and nCBP-2 reduces cassava brown streak disease symptom severity and incidence. Plant Biotechnol J 17:421–434. https://doi.org/10.1111/pbi.12987
Guo J, Gaj T, Barbas CF (2010) Directed evolution of an enhanced and highly efficient FokI cleavage domain for zinc finger nucleases. J Mol Biol 400:96–107. https://doi.org/10.1016/s13007-018-0305-8
Guo J, Li K, Jin L, Xu R, Miao K, Yang F, Qi C, Zhang L, Botella JR, Wang R, Miao Y (2018) A simple and cost-effective method for screening of CRISPR/Cas9-induced homozygous/biallelic mutants. Plant Methods 14:40. https://doi.org/10.1186/s13007-018-0305-8
Harrington LB, Burstein D, Chen JS, Paez-Espino D, Ma E, White IP, Cofsky JC, Kyrpides NC, Banfield JF, Doudna JA (2018) Programmed DNA destruction by miniature CRISPR-Cas14 enzymes. Science 362:839–842. https://doi.org/10.1126/science.aav4294
He Y, Zhu M, Wang L, Wu J, Wang Q, Wang R, Zhao Y (2018) Programmed self-elimination of the CRISPR/Cas9 construct greatly accelerates the isolation of edited and transgene-free rice plants. Mol Plant 11:1210–1213. https://doi.org/10.1016/j.molp.2018.05.005
Holubova K, Hensel G, Vojta P, Tarkowski P, Bergougnoux V, Galuszka P (2018) Modification of barley plant productivity through regulation of cytokinin content by reverse-genetics approaches. Front Plant Sci 9:1676. https://doi.org/10.3389/fpls.2018.01676
Hong J, Shi Q, Biswas S, Jiang SC, Shi J (2021) Moving genome edited crops forward from the laboratory bench to the kitchen table. Food Cont 122:107790. https://doi.org/10.1016/j.foodcont.2020.107790
Hooghvorst I, Nogués S (2020) Chromosome doubling methods in doubled haploid and haploid inducer-mediated genome-editing systems in major crops. Plant Cell Rep. https://doi.org/10.1007/s00299-020-02605-0
Hua Y, Wang C, Huang J, Wang K (2017) A simple and efficient method for CRISPR/Cas9-induced mutant screening. J Genet Genomics 44:207–213. https://doi.org/10.1016/j.jgg.2017.03.005
Jaganathan D, Ramasamy K, Sellamuthu G, Jayabalan S, Venkataraman G (2018) CRISPR for crop improvement: an update review. Front Plant Sci 9:985. https://doi.org/10.3389/fpls.2018.00985
Jin S, Zong Y, Gao Q, Zhu Z, Wang Y, Qin P, Liang C, Wang D, Qiu JL, Zhang F, Gao C (2019) Cytosine, but not adenine, base editors induce genome-wide off-target mutations in rice. Science 364:292–295. https://doi.org/10.1126/science.aaw7166
Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E (2012) A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337:816–821. https://doi.org/10.1126/science.1225829
Jouanin A, Boyd LA, Visser RGF, Smulders MJ (2018) Development of wheat with hypoimmunogenic gluten obstructed by the gene editing policy in Europe. Front Plant Sci 9:1523. https://doi.org/10.3389/fpls.2018.01523
Karunarathna NL, Wang H, Harloff HJ, Jiang L, Jung C (2020) Elevating seed oil content in a polyploid crop by induced mutations in SEED FATTY ACID REDUCER genes. Plant Biotechnol J 18:2251–2266. https://doi.org/10.1111/pbi.13381
Kelliher T, Starr D, Su X, Tang G, Chen Z, Carter J, Wittich PE, Song S, Green J, Burch E, McCuiston J, Gu W, Sun Y, Strebe T, Roberts J, Bate NJ, Que Q (2019) One-step genome editing of elite crop germplasm during haploid induction. Nat Biotechnol 37:287–292. https://doi.org/10.1038/s41587-019-0038-x
Khan MSS, Basnet R, Islam SA, Shu Q (2019a) Mutational analysis of OsPLDα1 reveals its involvement in phytic acid biosynthesis in rice grains. J Agric Food Chem 41:11436–11443. https://doi.org/10.1021/acs.jafc.9b05052
Khan MZ, Haider S, Mansoor S, Amin I (2019b) Targeting plant ssDNA viruses with engineered miniature CRISPR-Cas14a. Trends Biotech 37:800–804. https://doi.org/10.1016/j.tibtech.2019.03.015
Khanday I, Skinner D, Yang B, Mercier R, Sundaresan V (2019) A male-expressed rice embryogenic trigger redirected for asexual propagation through seeds. Nature 565:91–95. https://doi.org/10.1038/s41586-018-0785-8
Kim H, Kim ST, Ryu J, Kang BC, Kim JS, Kim SG (2017) CRISPR/Cpf1-mediated DNA-free plant genome editing. Nat Commun 8:14406. https://doi.org/10.1038/ncomms14406
Kim D, Alptekin B, Budak H (2018) CRISPR/Cas9 genome editing in wheat. Funct Integr Genomics 18:31–41. https://doi.org/10.1007/s10142-017-0572-x
Kis A, Hamar E, Tholt G, Bán R, Havelda Z (2019) Creating highly efficient resistance against wheat dwarf virus in barley by employing CRISPR/Cas9 system. Plant Biotechnol J 17(6):1004–1006. https://doi.org/10.1111/pbi.13077
Komor AC, Kim YB, Packer MS, Zuris JA, Liu DR (2016) Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 533:420–424. https://doi.org/10.1038/nature17946
Kuang Y, Li S, Ren B, Yan F, Spetz C, Li X, Zhou X, Zhou H (2020) Base-editing-mediated artificial evolution of OsALS1 in planta to develop novel herbicide-tolerant rice germplasms. Mol Plant 13:565–572. https://doi.org/10.1016/j.molp.2020-01-010
Kumar N, Galli M, Ordon J, Stuttmann J, Kogel K-H, Imani J (2018) Further analysis of barley MORC1 using a highly efficient RNA-guided Cas9 gene-editing system. Plant Biotechnol j 16(11):1892–1903. https://doi.org/10.1111/pbi.12924
Kumar P, Alok A, Kumar J (2020a) Expanding the potential of CRISPR-Cas9 technology for crops improvement. In: Singh V (ed) Advances in Synthetic Biology. Springer, Singapore. https://doi.org/10.1007/978-981-15-0081-7_15
Kumar VVS, Verma RK, Yadav RK, Yadav P, Watts A, Rao MV, Chinnusamy V (2020b) CRISPR-Cas9 mediated genome editing of drought and salt tolerance (OsDST) gene in indica mega rice cultivar MTU1010. Physiol Mol Biol Plants 26:1099–1110. https://doi.org/10.1007/s12298-020-00819-w
Kwon CT, Heo J, Lemmon ZH, Capua Y, Hutton SF, Eck JV, Park SJ, Lippman ZB (2020) Rapid customization of Solanaceae fruit crops for urban agriculture. Nat Biotechnol 38:182–188. https://doi.org/10.1038/s41587-019-0361-2
Lacchini E, Kiegle E, Castellani M, Adam H, Jouannic S, Gregis V, Kater MM (2020) CRISPR-mediated accelerated domestication of African rice landraces. PLoS ONE 15(3):e0229782. https://doi.org/10.1371/journal.pone.0229782
Lema MA (2019) Regulatory aspects of gene editing in Argentina. Transgenic Res 28:147–150. https://doi.org/10.1007/s11248-019-00145-2
Li T, Liu B, Spalding MH, Weeks DP, Yang B (2012) High-efficiency TALEN-based gene editing produces disease-resistant rice. Nat Biotechnol 30:390–392. https://doi.org/10.1038/nbt.2199
Li M, Li X, Zhou Z, Wu P, Fang M, Pan X, Lin Q, Luo W, Wu G, Li H (2016) Reassessment of the four yield-related genes Gn1a, DEP1, GS3, and IPA1 in rice using a CRISPR/Cas9 system. Front Plant Sci 7:377. https://doi.org/10.3389/fpls.2016.00377
Li T, Yang X, Yu Y, Si X, Zhai X, Zhang H, Dong W, Gao C, Xu C (2018a) Domestication of wild tomato is accelerated by genome editing. Nature Biotechnol 36:1160–1163. https://doi.org/10.1038/nbt.4273
Li X, Wang Y, Chen S, Tian H, Fu D, Zhu B, Luo Y, Zhu H (2018b) Lycopene is enriched in tomato fruit by CRISPR/Cas9-mediated multiplex genome editing. Front Plant Sci 9:559. https://doi.org/10.3389/fpls.2018.00559
Li J, Li Y, Ma L (2019a) CRISPR/Cas9-based genome editing and its applications for functional genomic analyses in plants. Small Methods 3:1800473. https://doi.org/10.1002/smtd.201800473
Li R, Liu C, Zhao R, Wang L, Chen L, Yu W, Zhang S, Sheng J, Shen L (2019b) CRISPR/Cas9-Mediated SlNPR1 mutagenesis reduces tomato plant drought tolerance. BMC Plant Biol 19:38. https://doi.org/10.1186/s12870-018-1627-4
Li C, Zhang R, Meng X, Chen S, Zong Y, Lu C, Qiu J-L, Chen Y-H, Li J, Gao C (2020a) Targeted, random mutagenesis of plant genes with dual cytosine and adenine base editors. Nat Biotechnol 38:875–882. https://doi.org/10.1038/s41587-019-0393-7
Li H, Li J, Chen J, Yan L, Xia L (2020b) Precise modifications of both exogenous and endogenous genes in rice by prime editing. Mol Plant 13:671–674. https://doi.org/10.1016/j.molp.2020.03.011
Li H, Li X, Xu Y, Liu H, He M, Tian X, Wang Z, Wu X, Bu Q, Yang J (2020c) High-efficiency reduction of rice amylose content via CRISPR/Cas9-mediated base editing. Rice Sci 27(6):445–448. https://doi.org/10.1016/j.rsci.2020.09.001
Li Q, Wu G, Zhao Y, Wang B, Zhao B, Kong D, Wei H, Chen C, Wang H (2020d) CRISPR/Cas9-mediated knockout and overexpression studies reveal a role of maize phytochrome C in regulating flowering time and plant height. Plant Biotechnol J. https://doi.org/10.1111/pbi.13429
Li R, Ba Y, Song Y, Cui J, Zhang X, Zhang D, Yuan Z, Yang L (2020e) Rapid and sensitive screening and identification of CRISPR/Cas9 edited rice plants using quantitative real-time PCR coupled with high resolution melting analysis. Food Cont 112:107088. https://doi.org/10.1016/j.foodcont.2020.107088
Liang Z, Zhang K, Chen K, Gao C (2014) Targeted mutagenesis in Zea mays using TALENs and the CRISPR/Cas system. J Genet Genom 41:63–68. https://doi.org/10.1016/j.jgg.2013.12.001
Liao S, Qin X, Luo L, Han Y, Wang X, Usman B, Nawaz G, Zhao N, Liu Y, Li R (2019) CRISPR/Cas9-induced mutagenesis of semi-rolled leaf1,2 confers curled leaf phenotype and drought tolerance by influencing protein expression patterns and ROS scavenging in rice (Oryza sativa L.). Agrnomoy 9:728. https://doi.org/10.3390/agronomy9110728
Lin Q, Zong Y, Xue C, Wang S, Jin S, Zhu Z, Wang Y, Anzalone AV, Raguram A, Domam JL, Liu DR, Gao C (2020) Prime genome editing in rice and wheat. Nat Biotechnol 38:582–585. https://doi.org/10.1038/s41587-020-0455-x
Liu H, Wang K, Jia Z, Gong Q, Lin Z, Du L, Pei X, Ye X (2019a) Efficient induction of haploid plants in wheat by editing of TaMTL using an optimized Agrobacterium-mediated CRISPR system. J Exp Bot 71:1337–1349. https://doi.org/10.1093/jxb/erz529
Liu Y, Li G, Zhang Y, Chen L (2019b) Current advances on CRISPR/Cas genome editing technologies in plants. J South China Agric Univ 40:38–49. https://doi.org/10.7671/j.issn.1001-411X.201905058
Liu L, Kuang Y, Yan F, Li S, Ren B, Gosavi G, Spetz C, Li X, Wang X, Zhou X, Zhou H (2020a) Developing a novel artificial rice germplasm for dinitroaniline herbicide resistance by base editing of OsTubA2. Plant Biotechnol J. https://doi.org/10.1111/pbi.13430
Liu X, Qin R, Li J, Liao S, Shan T, Xu R, Wu D, Wei P (2020b) A CRISPR-Cas9-mediated domain-specific base-editing screen enables functional assessment of ACCase variants in rice. Plant Biotechnol J 18:1845–1847. https://doi.org/10.1111/pbi.13348
Lusser M, Parisi C, Plan D, Rodríguez-Cerezo E, Rodríguez-Cerezo E (2012) Deployment of new biotechnologies in plant breeding. Nat Biotechnol 30:231–239. https://doi.org/10.1038/nbt.2142
Ma X, Zhang Q, Zhu Q, Liu W, Chen Y, Qiu R, Wang B, Yang Z, Li H, Lin Y, Xie Y, Shen R, Chen S, Wang Z, Chen Y, Guo J, Chen L, Zhao X, Dong Z, Liu YG (2015) A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol Plant 8:1274–1284. https://doi.org/10.1016/j.molp.2015.04.007
Ma X, Zhu Q, Chen Y, Liu YG (2016) CRISPR/Cas9 platforms for genome editing in plants: developments and applications. Mol Plant 9:961–974. https://doi.org/10.1016/j.molp.2016.04.009
Mikami M, Toki S, Engdo M (2015) Parameters affecting frequency of CRISPR/Cas9 mediated targeted mutagenesis in rice. Plant Cell Rep 34:1807–1815. https://doi.org/10.1007/s00299-015-1826-5
Miladinovic D, Antunes D, Yildirim K, Bakhsh A, Cvejić S, Kondić-Špika A, Jeromela AM, Opsahl-Sorteberg HG, Zambounis A, Hilioti Z (2021) Targeted plant improvement through genome editing: from laboratory to field. Plant Cell Rep. https://doi.org/10.1007/s00299-020-02655-4
Ming M, Ren Q, Pan C, He Y, Zhang Y, Liu S, Zhong Z, Wang J, Malzahn AA, Wu J, Zheng X, Zhang Y, Qi Y (2020) Crispr–Cas12b enables efficient plant genome engineering. Nat Plants 6:202–208. https://doi.org/10.1038/s41477-020-0614-6
Moon SB, Lee JM, Kang JG, Lee NE, Ha DI, Kim DY, Kim SH, Yoo K, Kim D, Ko JH, Kim YS (2018) Highly efficient genome editing by CRISPR-Cpf1 using CRISPR RNA with a uridinylate-rich 3’-overhang. Nature Commun 9:3651. https://doi.org/10.1038/s41467-018-06129-w
Moon SB, Kim DY, Ko JH, Kim YS (2019) Recent advances in the CRISPR genome editing tool set. Exp Mol Med 51:1–11. https://doi.org/10.1038/s12276-019-0339-7
Murugan K, Seetharam AS, Severin AJ, Sashital DG (2020) CRISPR-Cas12a has widespread off-target and dsDNA-nicking effects. J Biol Chem 295:5538–5553. https://doi.org/10.1074/jbc.RA120.012933
Nekrasov V, Staskawicz B, Weigel D, Jones JDG, Kamoun S (2013) Targeted mutagenesis in the model plant Nicotiana benthamiana using Cas9 RNA-guided endonuclease. Nat Biotechnol 31:691–693. https://doi.org/10.1038/nbt.2655
Ortigosa A, Gimenez-lbanez S, Leonhardt N, Solano R (2019) Design of a bacterial speck resistant tomato by CRISPR/Cas9-mediated editing of SlJAZ2. Plant Biotechnol J 17:665–673. https://doi.org/10.1111/pbi.13006
Osakabe Y, Osakabe K (2015) Genome editing with engineered nucleases in plants. Plant Cell Physiol 56:389–400. https://doi.org/10.1093/pcp/pcu170
Pacher M, Puchta H (2017) From classical mutagenesis to nuclease-based breeding-directing natural DNA repair for a natural end-product. Plant J 90:819–833. https://doi.org/10.1111/tpj.13469
Peng C, Wang H, Xu X, Wang X, Chen X, Wei W, Lai Y, Liu G, Godwin ID, Li J, Zhang L, Xu J (2018) High-throughput detection and screening of plants modified by gene editing using quantitative real-time polymerase chain reaction. Plant J 95:557–567. https://doi.org/10.1111/tpj.13961
Pröbsting M, Schenke D, Hossain R, Häder C, Thurao T et al (2020) Loss of function of CRT1a (calreticulin) reduces plant susceptibility to Verticillium longisporum in both Arabidopsis thaliana and oilseed rape (Brassica napus). Plant Biotechnol j 18:2328–2344. https://doi.org/10.1111/pbi.13394
Razzaq A, Saleem F, Kanwal M, Mustafa G, Yousaf S, Arshad HMI, Hameed MK, Khan MS, Joyia FA (2019) Modern trends in plant genome editing: an inclusive review of the CRISPR/Cas9 toolbox. Int J Mol Sci 20:4045. https://doi.org/10.3390/ijms20164045
Ren Q, Sretenovic S, Liu S, Tang X, Huang L, He Y, Liu L, Guo Y, Zhong Z, Liu G, Cheng Y, Zheng X, Pan C, Yin D, Zhang Y, Li W, Qi L, Li C, Qi Y, Zhang Y (2021) PAM-less plant genome editing using a CRISPR–SpRY toolbox. Nat Plants. https://doi.org/10.1038/s41477-020-00827-4
Safari F, Zare K, Negahdaripour M, Barekati-Mowahed M, Ghasemi Y (2019) CRISPR- Cpf1 proteins: structure, function and implications for genome editing. Cell Biosci 9:36. https://doi.org/10.1186/s13578-019-0298-7
Salesse-Smith CE et al (2018) Overexpression of Rubisco subunits with RAF1 increases Rubisco content in maize. Nat Plants 4:802–810. https://doi.org/10.1038/s41477-018-0252-4
Sánchez-León S, Gil-Humanes J, Ozuna CV, Giménez MJ, Sousa C, Voytas DF, Barro F (2018) Low-gluten, nontransgenic wheat engineered with CRISPR/Cas9. Plant Biotechnol J 16:902–910. https://doi.org/10.1111/pbi.12837
Sashidhar N, Harloff HJ, Potgieter L, Jung C (2020) Gene editing of three BnITPK genes in tetraploid oilseed rape leads to significant reduction of phytic acid in seeds. Plant Biotechnol J 18:2241–2250. https://doi.org/10.1111/pbi.13380
Schaart JG, van de Wiel CCM, Lotz LAP, Smulders MJM (2016) Opportunities for products of new plant breeding techniques. Trends Plant Sci 21:438–449. https://doi.org/10.1016/j.tplants.2015.11.006
Schaeffer SM, Nakata PA (2015) CRISPR/Cas9-mediated genome editing and gene replacement in plants: Transitioning from lab to field. Plant Sci 240:130–142. https://doi.org/10.1016/j.plantsci.2015.09.011
Schindele P, Puchta H (2020) Engineering CRISPR/LbCas12a for highly efficient, temperature-tolerant plant gene editing. Plant Biotechnol J 8:1118–1120. https://doi.org/10.1111/pbi.13275
Schindele A, Dorn A, Puchta H (2019) CRISPR/Cas brings plant biology and breeding into the fast lane. Curr Opin Biotechnol 61:7–14. https://doi.org/10.1016/j.copbio.2019.08.006
Schnable PS, Springer NM (2013) Progress toward understanding heterosis in crop plants. Annu Rev Plant Biol 64:71–88. https://doi.org/10.1141/annurev-arplant-042110-103827
Schwander T, Schada von Borzyskowski L, Burgener S, Cortina NS, Erb TJ (2016) A synthetic pathway for the fixation of carbon diox-ide in vitro. Science 354:900–904. https://doi.org/10.1126/science.aah5237
Schwartz C, Lenderts B, Feigenbutz L, Barone P, Llaca V, Fengler K, Svitashev S (2020) CRISPR–Cas9-mediated 75.5-Mb inversion in maize. Nat Plants 6:1427–1431. https://doi.org/10.1038/s41477-020-00817-6
Shan Q, Wang Y, Li J, Gao C (2014) Genome editing in rice and wheat using the CRISPR/Cas system. Nat Protoc 9(10):2395–2410. https://doi.org/10.1038/nprot.2014.157
Shimatani Z, Kashojiya S, Takayama M, Terada R, Arazoe T, Ishii H, Teramura H, Yamamoto T, Komatsu H, Miura K, Ezura H, Nishida K, Ariizumi T, Kondo A (2017) Targeted base editing in rice and tomato using a CRISPR-Cas9 cytidine deaminase fusion. Nat Biotechnol 35:441–443. https://doi.org/10.1038/nbt.3833
Smyth SJ (2017) Canadian regulatory perspectives on genome engineered crops. GM Crop Food 8:35–43. https://doi.org/10.1080/21645698.2016.1257468
South PF, Cavanagh AP, Liu HW, Ort DR (2019) Synthetic glycolate metabolism pathways stimulate crop growth and productivity in the field. Science 363:9077. https://doi.org/10.1126/science.aat9077
Tang X, Liu G, Zhou J, Ren Q, You Q, Tian L, Xin X, Zhong Z, Liu B, Zheng X, Zhang D, Malzahn A, Gong Z, Qi Y, Zhang T, Zhang Y (2018) A large-scale whole-genome sequencing analysis reveals highly specific genome editing by both Cas9 and Cpf1 (Cas12a) nucleases in rice. Genome Biol 19:84. https://doi.org/10.1186/s13059-018-1458-5
Tang X, Sretenovic S, Ren Q, Jia X, Li M et al (2020) Plant prime editors enable precise gene editing in rice cells. Mol Plant 13:667–670. https://doi.org/10.1016/j.molp.2020.03.010
Teng F, Cui T, Feng G, Guo L, Xu K, Gao Q, Li T, Li J, Zhou Q, Li W (2018) Repurposing CRISPR-Cas12b for mammalian genome engineering. Cell Discov 4:63. https://doi.org/10.1038/s41421-018-0069-3
Tian S, Jiang L, Cui X, Zhang J, Guo S, Li M, Zhang H, Ren Y, Gong G, Zong M, Liu F, Chen Q, Xu Y (2018) Engineering herbicide-resistant watermelon variety through CRISPR/Cas9-mediated base-editing. Plant Cell Rep 37:1353–1356. https://doi.org/10.1007/s00299-018-2299-0
Tian X, Wang JW, Li J, Han B (2020) Designing future crops: challenges and strategies forsustainable agriculture. Plant J 105:1165–1178. https://doi.org/10.1111/tpj.15107
Tripathi L, Ntui VO, Tripathi JN (2020) CRISPR/Cas9-based genome editing of banana for disease resistance. Curr Opin Plant Biol 56:118–126. https://doi.org/10.1016/j.pbi.2020.05.003
Tuncel A, Corbin KR, Ahn-Jarvis J, Harris S, Hawkins E et al (2019) Cas9-mediated mutagenesis of potato starch-branching enzymes generates a range of tuber starch phenotypes. Plant Biotechnol j 17(12):2259–2271. https://doi.org/10.1111/pbi.13137
Van Vu T, Sung YW, Kim J, Doan DTH, Tran MT, Kim JY (2019) Challenges and perspectives in homology-directed gene targeting in monocot plants. Rice 12:95. https://doi.org/10.1186/s12284-019-0355-1
Veillet F, Chauvin L, Kermarrec MP, Sevestre F, Merrer M et al (2019a) The Solanum tuberosum GBSSI gene: a target for assessing gene and base editing in tetraploid potato. Plant Cell Rep 38:1065–1080. https://doi.org/10.1007/s00299-019-02426-w
Veillet F, Perrot L, Chauvin L, Kermarrec MP, Guyon-Debast A, Chauvin JE, Nogue F, Mazier M (2019b) Transgene-free genome editing in tomato and potato plants using agrobacterium-mediated delivery of a CRISPR/Cas9 cytidine base editor. Int j Mol Sci 20:402. https://doi.org/10.3390/ijms20020402
Vu TV, Sivankalyani V, Kim EJ, Doan DTH, Tran MT, Kim J, Sung YW, Park M, Kang YJ, Kim JY (2020) Highly efficient homology-directed repair using CRISPR/Cpf1-geminiviral replicon in tomato. Plant Biotechnol J 18:2133–2143. https://doi.org/10.1111/pbi.13373
Wang Y, Cheng X, Shan Q, Zhang Y, Liu J, Gao C, Qiu JL (2014) Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nat Biotechnol 32:947–951. https://doi.org/10.1038/nbt.2969
Wang K, Mei DY, Liu QN, Xiao XH, Ruan WM, Huang T, Cao GS (2015) Research of methods to detect genomic mutations induced by CRISPR/Cas systems. J Biotechnol 214:128–132. https://doi.org/10.1016/j.jbiotec.2015.09.029
Wang C, Liu Q, Shen Y, Hua Y, Wang J, Lin J, Wu M, Sun T, Cheng Z, Mercier R, Wang K (2019) Clonal seeds from hybrid rice by simultaneous genome engineering of meiosis and fertilization genes. Nat Biotechnol 37:283–286. https://doi.org/10.1038/s41587-018-0003-0
Wang C, Wang G, Gao Y, Lu G, Habben JE, Mao G, Chen G, Wang J, Yang F, Zhao X, Zhang J, Mo H, Qu P, Liu J, Greene TW (2020a) A cytokinin-activation enzyme-like gene improves grain yield under various field conditions in rice. Plant Mol Biol 102:373–388. https://doi.org/10.1007/s11103-019-00952-5
Wang S, Yang Y, Guo M, Zhong C, Yan C, Sun S (2020b) Targeted mutagenesis of amino acid transporter genes for rice quality improvement using the CRISPR/Cas9 system. Crop J 8:457–464. https://doi.org/10.1016/j.cj.2020.02.005
Wolter F, Puchta H (2018) The CRISPR/Cas revolution reaches the RNA world: Cas13, a new Swiss Army Knife for plant biologists. Plant J 94:767–775. https://doi.org/10.1111/tpj.13899
Wu F, Qiao X, Zhao Y, Zhang Z, Gao Y, Shi L, Du H, Wang L, Zhang YJ, Zhang Y, Liu L, Wang Q, Kong D (2020) Targeted mutagenesis in Arabidopsis thaliana using CRISPR-Cas12b/C2c1. J Integr Plant Biol 11:1653–1658. https://doi.org/10.1111/jipb.12944
Wurtzel ET, Vickers CE, Hanson AD, Millar AH, Cooper M, Voss-Fels KP, Nikel PI, Erb TJ (2019) Revolutionizing agriculture with synthetic biology. Nat Plants 5:1207–1210. https://doi.org/10.1038/s41477-019-0539-0
Xu RF, Li H, Qin RY, Li J, Qiu CH, Yang YC, Ma H, Li L, Wei PC, Yang JB (2015) Generation of inheritable and “transgene clean” targeted genome-modified rice in later generations using the CRISPR/Cas9 system. Sci Rep 5:11491. https://doi.org/10.1038/srep11491
Xu R, Qin R, Li H, Li D, Li L, Wei P, Yang J (2017) Generation of targeted mutant rice using CRISPR-Cpf1 system. Plant Biotechnol J 15:713–717. https://doi.org/10.1111/pbi.12669
Xu Z, Xu X, Gong Q, Li Z, Li Y, Wang S, Yang Y, Ma W, Liu L, Zhu B, Zou L, Chen G (2019) Engineering broad-spectrum bacterial blight resistance by simultaneously disrupting variable TALE-binding elements of multiple susceptibility genes in rice. Mol Plant 12:1434–1446. https://doi.org/10.1016/j.molp.2019.08.006
Xu Y, Lin Q, Li X, Wang F, Chen Z, Wang J, Li W, Fan F, Tao Y, Jiang Y, Wei X, Zhang R, Zhu Q-H, Bu Q, Yang J, Gao C (2020) Fine-tuning the amylose content of rice by precise base-editing of the Wx gene. Plant Biotechnol J 19:11–13. https://doi.org/10.1111/pbi.13433
Xu Z, Kuang Y, Ren B, Yan D, Yan F, Spetz C, Sun W, Wang G, Zhou X, Zhou H (2021) SpRY greatly expands the genome editing scope in rice with highly flexible PAM recognition. Genome Biol 22:6. https://doi.org/10.1186/s13059-020-02231-9
Yan D, Ren B, Liu L, Yan F, Zhou H (2021) High-efficiency and multiplex adenine base editing in plants using new tada variants. Mol Plant. https://doi.org/10.1016/j.molp.2021.02.007
Yang Q, Zhong X, Li Q, Lan J, Tang H et al (2020) Mutation of the d-hordein gene by RNA-guided Cas9 targeted editing reducing the grain size and changing grain compositions in barley. Food Chem 311:125892. https://doi.org/10.1016/j.foodchem.2019.125892
Yoon YZ, Venkatesh J, Lee JH, Kim J, Lee HE, Kim DS, Kang BC (2020) Genome editing of eIF4E1 in tomato confers resistance to pepper mottle virus. Front Plant Sci 11:1098. https://doi.org/10.3398/fpls.2020.01098
Yu H, Lin T, Meng X, Du H, Zhang J et al (2021) A route to de novo domestication of wild allotetraploid rice. Cell 184(5):1156–1170. https://doi.org/10.1016/j.cell.2021.01.013
Yuste-Lisbona FJ, Fernandez-Lozano A, Pineda B, Bretones S, Ortíz-Atienza A et al (2020) ENO regulates tomato fruit size through the floral meristem development network. PNAS 117(14):8187–8195. https://doi.org/10.1073/pnas.1913688117
Zeng Y, Wen J, Zhao W, Wang Q, Huang W (2019) Rational improvement of rice yield and cold tolerance by editing the three genes OsPIN5b, GS3, and OsMYB30 with the CRISPR-Cas9 system. Front Plant Sci 10:1663. https://doi.org/10.3389/fpls.2019.01663
Zeng D, Liu T, Ma X, Wang B, Zheng Z, Zhang Y, Xie X, Yang B, Zhao Z, Zhu Q, Liu Y-G (2020) Quantitative regulation of Waxy expression by CRISPR/Cas9-based promoter and 5’UTR-intron editing improvesgrain quality in rice. Plant Biotechnol J 18:2385–2387. https://doi.org/10.1111/pbi.13427
Zhai Y, Yu K, Cai S, Hu L, Amoo O et al (2020) Targeted mutagenesis of BnTT8 homologs controls yellow seed coat development for effective oil production in Brassica napus L. Plant Biotechnol J 18:1153–1168. https://doi.org/10.1111/pbi.13281
Zhan X, Zhang F, Zhong Z, Chen R, Wang Y, Chang L, Bock R, Nie B, Zhang J (2019) Generation of virus-resistant potato plants by RNA genome targeting. Plant Biotechnol J 17:1814–1822. https://doi.org/10.1111/pbi.13102
Zhang Y, Zhang F, Li X, Baller JA, Qi Y, Starker CG, Bogdanove AJ, Voytas DF (2013) Transcription activator-like effector nucleases enable efficient plant genome engineering. Plant Physiol 161:20–27. https://doi.org/10.1104/pp.112.205179
Zhang H, Zhang J, Wei P, Zhang B, Gou F, Feng Z, Mao Y, Yang L, Zhang H, Xu N, Zhu JK (2014) The CRISPR/Cas9 system produces specific and homozygous targeted gene editing in rice in one generation. Plant Biotechnol J 12:797–807. https://doi.org/10.1111/pbi.12200
Zhang M, Cao Y, Wang Z, Wang ZQ, Shi J, Liang X, Song W, Chen Q, Lai J, Jiang C (2018a) A retrotransposon in an HKT1 family sodium transporter causesvariation of leaf Na+exclusion and salt tolerance in maize. New Phytol 217:1161–1176. https://doi.org/10.1111/nph.14882
Zhang Y, Li D, Zhang D, Zhao X, Cao X, Dong L, Liu J, Chen K, Zhang H, Gao C, Wang D (2018b) Analysis of the functions of TaGW2 homoeologs in wheat grain weight and protein content traits. Plant J 94:857–866. https://doi.org/10.1111/tpj.13903
Zhang A, Liu Y, Wang F, Li T, Chen Z, Kong D, Bi J, Zhang F, Luo X, Wang J, Tang J, Yu X, Liu G, Luo L (2019a) Enhanced rice salinity tolerance via CRISPR/Cas9-targeted mutagenesis of the OsRR22 gene. Mol Breed 39:47. https://doi.org/10.1007/s11032-019-0954-y
Zhang Z, Hua L, Gupta A, Tricoli A, Edwards KJ, Yang B, Li W (2019b) Development of an agrobacterium delivered CRISPR/Cas9 system for wheat genome editing. Plant Biotechnol j 17:1623–1635. https://doi.org/10.1111/pbi.13088
Zhang M, Liu Q, Yang X, Xu J, Liu G, Yao X, Ren R, Xu J, Lou L (2020a) CRISPR/Cas9-mediated mutagenesis of Clpsk1 in watermelon to confer resistance to Fusarium oxysporum f. sp. niveum. Plant Cell Rep 39:589–595. https://doi.org/10.1007/s00299-020-02516-0
Zhang Y, Pribil M, Palmgren M, Gao C (2020b) A CRISPR way for accelerating improvement of food crops. Nat Food 1:200–205. https://doi.org/10.1038/s43016-020-0051-8
Zhong Y, Blennow A, Kofoed-Enevoldsen O, Jiang D, Hebelstrup KH (2019a) Protein Targeting to Starch 1 is essential for starchy endosperm development in barley. J Exp Bot 70(2):485–496. https://doi.org/10.1093/jxb/ery398
Zhong Y, Liu C, Qi X, Jiao Y, Wang D, Wang Y, Liu Z, Chen C, Chen B, Tian X, Li J, Chen M, Dong X, Xu X, Li L, Li W, Liu W, Jin W, Lai J, Chen S (2019b) Mutation of ZmDMP enhances haploid induction in maize. Nat Plant 5:575–580. https://doi.org/10.1038/s41477-019-0443-7
Zhou H, Liu B, Weeks DP, Spalding MH, Yang B (2014) Large chromosomal deletions and heritable small genetic changes induced by CRISPR/Cas9 in rice. Nucleic Acids Res 42:10903–10914. https://doi.org/10.1093/nar/gku806
Zhou J, Xin X, He Y, Chen H, Li Q, Tang X, Zhong Z, Deng K, Zheng X, Akher SA, Cai G, Qi Y, Zhang Y (2019) Multiplex QTL editing of grain-related genes improves yield in elite rice varieties. Plant Cell Rep 38:475–485. https://doi.org/10.1007/s00299-018-2340-3
Zhu C, Bortesi L, Baysal C, Twyman RM, Fischer S, Capell T, Schillberg S, Christou P (2017) Characteristics of genome editing mutations in cereal crops. Trends Plant Sci 22:38–52. https://doi.org/10.1016/j.tplants.2016.08.009
Zong Y, Wang Y, Li C, Zhang R, Chen K, Ran Y, Qiu JL, Wang D, Gao C (2017) Precise base editing in rice, wheat and maize with a Cas9-cytidine deaminase fusion. Nat Biotechnol 35:438–440. https://doi.org/10.1038/nbt.3811
Zsögön A, Čermák T, Naves ER, Notini MM, Edel KH, Weinl S, Freschi L, Voytas DF, Kudla J, Peres LEP (2018) De novo domestication of wild tomato using genome editing. Nat Biotechnol 36:1211–1216. https://doi.org/10.1038/nbt.4272
Acknowledgements
This work was partially supported by grants from the China National Transgenic Plant Special Fund (2016ZX08012-002, 2017ZX08013-001) and the Programme of Introducing Talents of Discipline to Universities (111 Project, B14016).
Author information
Authors and Affiliations
Contributions
SJ designed the project, supervised the project, and revised the manuscript. BS collected information, did analysis, and wrote the first draft of the manuscript. ZD participated in the discussion.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Editorial Responsibility: Sukhpreet Sandhu.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Biswas, S., Zhang, D. & Shi, J. CRISPR/Cas systems: opportunities and challenges for crop breeding. Plant Cell Rep 40, 979–998 (2021). https://doi.org/10.1007/s00299-021-02708-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-021-02708-2