Abstract
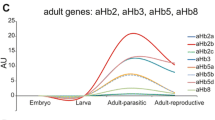
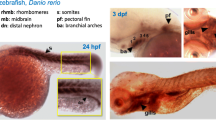
Respiratory proteins are responsible for transport and storage of oxygen. It is well established that specific requirements for oxygen during vertebrate ontogeny cause switches of hemoglobin chain expression. Here, we characterize the developmental profiles of zebrafish (Danio rerio) globins by means of quantitative real-time reverse transcription PCR. The total mRNA levels of the hemoglobin chains, including a newly identified embryonic α-chain, as well as myoglobin, neuroglobin, cytoglobin 1 and 2, and globin X were estimated. mRNAs of all globins were detectable in unfertilized eggs, suggesting maternal storage. Embryonic α- and β-hemoglobin mRNA peaked at hatching and the switch to adult hemoglobin expression occurred 16 dpf. Enhanced myoglobin mRNA levels were detected ~31 h post-fertilization (hpf), coinciding with the heart and the muscle development, while neuroglobin mRNA expression pattern correlates with the formation of the nervous system. Amounts of myoglobin and neuroglobin mRNA were similar within an order of magnitude throughout the ontogeny, tentatively supporting a respiratory role of neuroglobin. Cytoglobin 2 mRNA levels increased gradually, whereas cytoglobin 1 mRNA levels increased strongly after ~31 hpf, which is in agreement with a function in cell proliferation. Globin X mRNA level was highest in oocytes, but low in later stages. Together, these data suggest a specific role for each globin, which are also associated with certain events in fish development.




Similar content being viewed by others
Abbreviations
- ARP:
-
Acidic ribosomal protein
- Cygb:
-
Cytoglobin
- Ef1α:
-
Elongation factor 1 α
- GbX:
-
Globin X
- Dpf:
-
Days post-fertilization
- Hb:
-
Hemoglobin
- His 2h3c1:
-
Histone 2h3c1
- Hpf:
-
Hours post-fertilization
- Ldha:
-
Lactate dehydrogenase A
- Mb:
-
Myoglobin
- Ngb:
-
Neuroglobin
- Ppia:
-
Peptidylprolyl isomerase A (cyclophilin A)
- ROS:
-
Reactive oxygen species
References
Abascal F, Zardoya R, Posada D (2005) Prottest: selection of best-fit models of protein evolution. Bioinformatics 21:2104–2105
Bauer C, Tamm R, Petschow D, Bartels R, Bartels H (1975) Oxygen affinity and allosteric effects of embryonic mouse haemolglobins. Nature 257:333–334
Bentmann A, Schmidt M, Reuss S, Wolfrum U, Hankeln T, Burmester T (2005) Divergent distribution in vascular and avascular mammalian retinae links neuroglobin to cellular respiration. J Biol Chem 280:20660–20665
Brownlie A, Hersey C, Oates AC, Paw BH, Falick AM, Witkowska HE, Flint J, Higgs D, Jessen J, Bahary N, Zhu H, Lin S, Zon L (2003) Characterization of embryonic globin genes of the zebrafish. Dev Biol 255:48–61
Brunori M (1975) Molecular adaptation to physiological requirements: the hemoglobin system of trout. Curr Top Cell Regul 9:1–39
Brunori M, Giuffre A, Nienhaus K, Nienhaus GU, Scandurra FM, Vallone B (2005) Neuroglobin, nitric oxide, and oxygen: functional pathways and conformational changes. Proc Natl Acad Sci U S A 102:8483–8488
Burmester T, Hankeln T (2009) What is the function of neuroglobin? J Exp Biol 212:1423–1428
Burmester T, Weich B, Reinhardt S, Hankeln T (2000) A vertebrate globin expressed in the brain. Nature 407:520–523
Burmester T, Ebner B, Weich B, Hankeln T (2002) Cytoglobin: a novel globin type ubiquitously expressed in vertebrate tissues. Mol Biol Evol 19:416–421
Bustin S (2002) Quantification of mRNA using real-time reverse transcription PCR (RT-PCR): trends and problems. J Mol Endocrin 29:23–39
Chan FY, Robinson J, Brownlie A, Shivdasani RA, Donovan A, Brugnara C, Kim J, Lau BC, Witkowska HE, Zon LI (1997) Characterization of adult alpha- and beta-globin genes in the zebrafish. Blood 89:688–700
Coates ML (1975) Hemoglobin function in the vertebrates: an evolutionary model. J Mol Evol 6:285–307
Dickerson RE, Geis I (1983) Hemoglobin. The Benjamin/Cummings, Menlo Park, California
Fago A, Hundahl C, Malte H, Weber RE (2004) Functional properties of neuroglobin and cytoglobin. Insights into the ancestral physiological roles of globins. IUBMB Life 56:689–696
Fraser J, de Mello LV, Ward D, Rees HH, Williams DR, Fang Y, Brass A, Gracey AY, Cossins AR (2006) Hypoxia-inducible myoglobin expression in nonmuscle tissues. Proc Natl Acad Sci USA 103:2977–2981
Fuchs C, Heib V, Kiger L, Haberkamp M, Roesner A, Schmidt M, Hamdane D, Marden MC, Hankeln T, Burmester T (2004) Zebrafish reveals different and conserved features of vertebrate neuroglobin gene structure, expression pattern, and ligand binding. J Biol Chem 279:24116–24122
Fuchs C, Luckhardt A, Gerlach F, Burmester T, Hankeln T (2005) Duplicated cytoglobin genes in teleost fishes. Biochem Biophys Res Commun 337:216–223
Grillitsch S, Medgyesy N, Schwerte T, Pelster B (2005) The influence of environmental P(O2) on hemoglobin oxygen saturation in developing zebrafish Danio rerio. J Exp Biol 208:309–316
Hankeln T, Ebner B, Fuchs C, Gerlach F, Haberkamp M, Laufs TL, Roesner A, Schmidt M, Weich B, Wystub S, Saaler-Reinhardt S, Reuss S, Bolognesi M, De Sanctis D, Marden MC, Kiger L, Moens L, Dewilde S, Nevo E, Avivi A, Weber RE, Fago A, Burmester T (2005) Neuroglobin and cytoglobin in search of their role in the vertebrate globin family. J Inorg Biochem 99:110–119
Hartley SE, Horne MT (1984) Chromosome polymorphism and constitutive heterochromatin in the Atlantic salmon, Salmo salar. Chromosoma 89:377–380
Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17:754–755
Iuchi I (1973) Chemical and physiological properties of the larval and the adult hemoglobins in rainbow trout, Salmo gairdnerii irideus. Comp Biochem Physiol B 44:1087–1101
Johnson KR, Wright JE Jr, May B (1987) Linkage relationships reflecting ancestral tetraploidy in salmonid fish. Genetics 116:579–591
Jonz MG, Nurse CA (2005) Development of oxygen sensing in the gills of zebrafish. J Exp Biol 208:1537–1549
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203:253–310
Larhammar D, Risinger C (1994) Molecular genetic aspects of tetraploidy in the common carp Cyprinus carpio. Mol Phylogenet Evol 3:59–68
Malicki J, Neuhauss SCF, Schier AFS-K L, Stemple DL, Didier Y, Stainier R, Abdelilah S, Zwartkruis F, Rangini Z, Driever W (2005) Mutations affecting development of the zebrafish retina. Development 123:263–273
Mammen PP, Shelton JM, Ye Q, Kanatous SB, McGrath AJ, Richardson JA, Garry DJ (2006) Cytoglobin is a stress-responsive hemoprotein expressed in the developing and adult brain. J Histochem Cytochem 54:1349–1361
Maruyama K, Yasumasu S, Iuchi I (1999) Characterization and expression of embryonic globin in the rainbow trout, Oncorhynchus mykiss: intra-embryonic initiation of erythropoiesis. Dev Growth Differ 41:589–599
Meeson AP, Radford N, Shelton JM, Mammen PP, DiMaio JM, Hutcheson K, Kong Y, Elterman J, Williams RS, Garry DJ (2001) Adaptive mechanisms that preserve cardiac function in mice without myoglobin. Circ Res 88:713–720
Mitz SA, Reuss S, Folkow LP, Blix AS, Ramirez JM, Hankeln T, Burmester T (2009) When the brain goes diving: glial oxidative metabolism may confer hypoxia tolerance to the seal brain. Neuroscience 163:552–560
Miyamura Y, Nakayasu H (2001) Zonal distribution of Purkinje cells in the zebrafish cerebellum: analysis by means of a specific monoclonal antibody. Cell Tissue Res 305:299–305
Nicholas KB, Nicholas HBJ (1997) GeneDoc: analysis and visualization of genetic variation. http://wwwpscedu/biomed/genedoc/
Pelster B, Burggren WW (1996) Disruption of hemoglobin oxygen transport does not impact oxygen-dependent physiological processes in developing embryos of zebra fish (Danio rerio). Circ Res 79:358–362
Perutz MF (1960) Structure of hemoglobin. Brookhaven Symp Biol 13:165–183
Ransom DG, Haffter P, Odenthal J, Brownlie A, Vogelsang E, Kelsh RN, Brand M, van Eeden FJ, Furutani-Seiki M, Granato M, Hammerschmidt M, Heisenberg CP, Jiang YJ, Kane DA, Mullins MC, Nusslein-Volhard C (1996) Characterization of zebrafish mutants with defects in embryonic hematopoiesis. Development 123:311–319
Reuss S, Saaler-Reinhardt S, Weich B, Wystub S, Reuss MH, Burmester T, Hankeln T (2002) Expression analysis of neuroglobin mRNA in rodent tissues. Neuroscience 115:645–656
Roesner A, Fuchs C, Hankeln T, Burmester T (2005) A globin gene of ancient evolutionary origin in lower vertebrates: evidence for two distinct globin families in animals. Mol Biol Evol 22:12–20
Roesner A, Hankeln T, Burmester T (2006) Hypoxia induces a complex response of globin expression in zebrafish (Danio rerio). J Exp Biol 209:2129–2137
Roesner A, Mitz SA, Hankeln T, Burmester T (2008) Globins and hypoxia adaptation in the goldfish, Carassius auratus. FEBS J 275:3633–3643
Rombough P, Drader H (2009) Hemoglobin enhances oxygen uptake in larval zebrafish (Danio rerio) but only under conditions of extreme hypoxia. J Exp Biol 212:778–784
Schmidt M, Gerlach F, Avivi A, Laufs T, Wystub S, Simpson JC, Nevo E, Saaler-Reinhardt S, Reuss S, Hankeln T, Burmester T (2004) Cytoglobin is a respiratory protein in connective tissue and neurons, which is up-regulated by hypoxia. J Biol Chem 279:8063–8069
Sprague J, Bayraktaroglu L, Clements D, Conlin T, Fashena D, Frazer K, Haendel M, Howe D, Mani P, Ramachandran S, Schaper K, Segerdell E, Song P, Sprunger B, Taylor S, Van Slyke C, Westerfield M (2006) The zebrafish information network: the zebrafish model organism database. Nucl Acids Res 34:D581–D585
Stainier DY, Lee RK, Fishman MC (1993) Cardiovascular development in the zebrafish. I. Myocardial fate map and heart tube formation. Development 119:31–40
Stamatoyannopoulos G (1991) Human hemoglobin switching. Science 252:383
Van Blerkom J, Davis PW, Lee J (1995) ATP content of human oocytes and developmental potential and outcome after in vitro fertilization and embryo transfer. Hum Reprod 10:415–424
van der Meer DL, van den Thillart GE, Witte F, de Bakker MA, Besser J, Richardson MK, Spaink HP, Leito JT, Bagowski CP (2005) Gene expression profiling of the long-term adaptive response to hypoxia in the gills of adult zebrafish. Am J Physiol Regul Integr Comp Physiol 289:R1512–R1519
Vlecken DH, Testerink J, Ott EB, Sakalis PA, Jaspers RT, Bagowski CP (2009) A critical role for myoglobin in zebrafish development. Int J Dev Biol 53:517–524
Weller PA, Price M, Isenberg H, Edwards YH, Jeffreys AJ (1986) Myoglobin expression: early induction and subsequent modulation of myoglobin and myoglobin mRNA during myogenesis. Mol Cell Biol 6:4539–4547
Wilkins NP (1985) Ontogeny and evolution of salmonid hemoglobins. Int Rev Cytol 94:269–298
Wittenberg JB, Wittenberg BA (2003) Myoglobin function reassessed. J Exp Biol 206:2011–2020
Acknowledgments
We thank Dr. Brigitte Walderich (Max-Planck Institute for Developmental Biology, Tübingen, Germany) for providing inbred zebrafish, Dr. Anja Roesner (Mainz) for supplying primer sequences and Detlef Teege for animal care. This work has been supported by grants of the Deutsche Forschungsgemeinschaft (Bu956/10 and Ha2103/3-4) and the Fonds der Chemischen Industrie.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tiedke, J., Gerlach, F., Mitz, S.A. et al. Ontogeny of globin expression in zebrafish (Danio rerio). J Comp Physiol B 181, 1011–1021 (2011). https://doi.org/10.1007/s00360-011-0588-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-011-0588-9