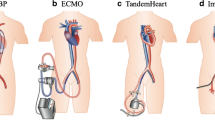
Abstract
Cardiogenic shock due to acute myocardial infarction, postcardiotomy syndrome following cardiac surgery, or manifestation of heart failure remains a clinical challenge with high mortality rates, despite ongoing advances in surgical techniques, widespread use of primary percutaneous interventions, and medical treatment. Clinicians have, therefore, turned to mechanical means of circulatory support. At present, a broad range of devices are available, which may be extracorporeal, implantable, or percutaneous; temporary or long term. Although counter pulsation provided by intra-aortic balloon pump (IABP) and comprehensive mechanical support for both the systemic and the pulmonary circulation through extracorporeal membrane oxygenation (ECMO) remain a major tool of acute care in patients with cardiogenic shock, both before and after surgical or percutaneous intervention, the development of devices such as the Impella or the Tandemheart allows less invasive forms of temporary support. On the other hand, concerning mid-, or long-term support, left ventricular assist devices have evolved from a last resort life-saving therapy to a well-established viable alternative for thousands of heart failure patients caused by the shortage of donor organs available for transplantation. The optimal selection of the assist device is based on the initial consideration according to hemodynamic situation, comorbidities, intended time of use and therapeutic options. The present article offers an update on currently available mechanical circulatory support systems (MCSS) for short and long-term use as well as an insight into future perspectives.





Similar content being viewed by others
References
Trost JC, Hillis LD (2006) Intra-aortic balloon counterpulsation. Am J Cardiol 97:1391–1398
DiGiorgi PL, Rao V, Naka Y, Mehmet CO (2003) Which patient, which pump? J Heart Lung Transplant 22:221–235
Mielck F, Quintel M (2005) Extracorporeal membrane oxygenation. Curr Opin Crit Care 11:87–93
Zhang T, Cheng G, Koert A et al (2009) Functional and biocompatibility performance of an integrated maglev pump-oxygenator. Artif Organs 33:36–45
Gibbon JH (1954) Application of a mechanical heart and lung apparatus to cardiac surgery. Minn Med 37:171–185
DeBakey ME (2005) Development of mechanical heart devices. Ann Thorac Surg 79:S2228–S2231
Gemmato CJ, Forrester MD, Myers TJ, Frazier OH, Cooley DA (2005) Thirty-five years of mechanical circulatory support at the Texas Heart Institute—an updated overview. Tex Heart Inst J 32:168–177
DeVries WC, Anderson JL, Joyce LD et al (1984) Clinical use of the total artificial heart. N Engl J Med 310:273–278
Fraser KH, Taskin ME, Griffith BP, Wu ZJ (2011) The use of computational fluid dynamics in the development of ventricular assist devices. Med Eng Phys 33:263–280
Caccamo M, Eckman P, John R (2011) Current state of ventricular assist devices. Curr Heart Fail Rep 8:91–98
Strüber M, Meyer AL, Malehsa D, Kugler C, Simon AR, Haverich A (2009) The current status of heart transplantation and the development of artificial heart systems. Dtsch Arztebl Int 106:471–477
Kirklin JK, Naftel DC, Kormos RL et al (2012) The fourth INTERMACS annual report: 4,000 implants and counting. J Heart Lung Trans 31:117–126
Spiliopoulos K, Giamouzis G, Karayannis G et al (2012) Current status of mechanical circulatory support: a systematic review. Cardiol Res Pract 2012:574198
Frazier OH, Rose EA, McCarthy P et al (1995) Improved mortality and rehabilitation of transplant candidates treated with a long-term implantable left ventricular assist system. Ann Surg 222:327–338
McCarthy PM, James KB, Savage RM et al (1994) Implantable left ventricular assist device: approaching an alternative for end-stage heart failure. Circulation 90:II83–II86
Goldstein DJ, Oz MC, Rose EA (1998) Implantable left ventricular assist devices. N Engl J Med 339:1522–1533
Slaughter MS, Rogers JG, Milano CA et al (2009) Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med 361:2241–2251
Rogers JG, Aaronson KD, Boyle AJ et al (2010) Continuous flow left ventricular assist device improves functional capacity and quality of life of advanced heart failure patients. J Am Coll Cardiol 55:1826–1834
Kirklin JK, Naftel DC, Kormos RL et al (2010) Second INTERMACS annual report: more than 1,000 primary left ventricular assist device implants. J Heart Lung Transplant 29:1–10
Pagani FD, Miller LW, Russell SD et al (2009) Extended mechanical circulatory support with a continuous-flow rotary left ventricular assist device. J Am Coll Cardiol 54:312–321
Hoshi H, Shinshi T, Takatani S (2006) Third-generation blood pumps with mechanical noncontact magnetic bearings. Artif Organs 30:324–338
Wieselthaler GM, O’Driscoll G, Jansz P, Khaghani A, Strueber M (2010) Initial clinical experience with a novel left ventricular assist device with a magnetically levitated rotor in a multi-institutional trial. J Heart Lung Transpl 29:1218–1225
Nishinaka T, Schima H, Roethy W et al (2006) The DuraHeart VAD, a magnetically levitated centrifugal pump: The University of Vienna bridge-to-transplant experience. Circulation 70:1421–1425
Farrar DJ, Bourque K, Dague CP et al (2007) Design features, developmental status, and experimental results with the Heartmate III centrifugal left ventricular assist system with a magnetically levitated rotor. ASAIO J 53:310–315
Gazzoli F, Alloni A, Pagani F et al (2007) Arrow CorAide left ventricular assist system: initial experience of the cardio-thoracic surgery center in Pavia. Ann Thorac Surg 83:279–282
Goldowsky M (2005) Lafaro, Reed G. Magnevad status of design improvements human blood results and preliminary sheep trial. Artif Organs 29:855–857
Masuzawa T, Ohta A, Tanaka N et al (2009) Estimation of changes in dynamic hydraulic force in a magnetically suspended centrifugal blood pump with transient computational fluid dynamics analysis. J Artif Organs 12:150–159
Ren Z, Jahanmir S, Heshmat H et al (2009) Design analysis and performance assessment of hybrid magnetic bearings for a rotary centrifugal blood pump. ASAIO J 55:340–347
Rose EA, Gelijns AC, Moskowitz AJ et al (2001) Long-term use of a left ventricular assist device for end-stage heart failure. N Engl J Med 345:1435–1443
Long JW, Healy AH, Rasmusson BY et al (2008) Improving outcomes with long-term”destination” therapy using left ventricular assist devices. J Thorac Cardiovasc Surg 135:1353–1360
Aaronson KD, Slaughter MS, Miller LW et al (2012) Use of an intrapericardial, continuous-flow, centrifugal pump in patients awaiting heart transplantation. Circulation 26(125):3191–3200
Miller LW, Pagani FD, Russell SD et al (2007) Use of a continuous-flow device in patients awaiting heart transplantation. N Engl J Med 357:885–896
Kirklin JK, Naftel DC, Kormos RL et al (2013) Fifth INTERMACS annual report: risk factor analysis from more than 6,000 mechanical circulatory support patients. J Heart Lung Transplant 32:141–156
Copeland JG, Smith RG, Arabia FA et al (2001) Comparison of the CardioWest total artificial heart, the Novacor left ventricular assist system, and the Thoratec ventricular assist system in bridge to transplantation. Ann Thorac Surg 71:S92–S97
El-Banayosy A, Arusoglu L, Morshuis M et al (2005) CardioWest total artificial heart: bad Oeynhausen experience. Ann Thorac Surg 80:548–552
Kung RT, Yu LS, Ochs B et al (1993) An atrial hydraulic shunt in a total artificial heart. A balance mechanism for the bronchial shunt. ASAIO J 39:M213–M217
Samuels L (2003) The AbioCor totally implantable replacement heart. Am Heart Hosp J 1:91–96
Dowling RD, Gray LA Jr, Etoch SW et al (2004) Initial experience with the AbioCor implantable replacement heart system. J Thorac Cardiovasc Surg 127:131–141
Frazier OH, Dowling RD, Gray LA, Shah NA, Pool T, Gregoric I (2004) The total artificial heart: where do we stand. Cardiology 101:117–121
Lederman D, Kung RT, McNair DS (2002) Therapeutic potential of implantable replacement hearts. Am J Cardiovasc Drug 2:297–301
Von Herrick JB (1912) Clinical features of sudden obstruction of the coronary arteries. JAMA 250:1757–1765
Thiele H, Zeymer U, Neumann FJ et al (2012) Intraaortic balloon support for myocardial infarction with cardiogenic shock. N Engl J Med 367:1287–1296
Werdan K, Ruß M, Buerke M et al (2012) Cardiogenic shock due to myocardial infarction: diagnosis, monitoring and treatment: a German-Austrian S3 Guideline. Dtsch Arztebl Int 109:343–351
Vogel RA, Shawl F, Tommaso C et al (1990) Initial report of the national registry of elective cardiopulmonary bypass supported coronary angioplasty. J Am Coll Cardiol 15:23–29
Putzer G, Braun P, Zimmermann A et al (2013) LUCAS compared to manual cardiopulmonary resuscitation is more effective during helicopter rescue-a prospective, randomized, cross-over manikin study. Am J Emerg Med 31:384–389
Marrin CA, Rose EA, Spotnitz HM, Bregman D (1982) Mechanical circulatory support via the left ventricular vent: the concept of left ventricular copulsation. J Thorac Cardiovasc Surg 84:426–429
Jung C, Ferrari M, Rödiger C, Fritzenwanger M, Figulla HR (2008) Combined Impella and intra-aortic balloon pump support to improve macro- and microcirculation: a clinical case. Clin Res Cardiol 97:849–850
Ferrari M, Poerner TC, Brehm BR et al (2008) First use of a novel plug-and-play percutaneous circulatory assist device for high-risk coronary angioplasty. Acute Card Care 10:111–115
Vranckx P, Foley DP, de Feijter PJ, Vos J, Smits P, Serruys PW (2003) Clinical introduction of the Tandemheart, a percutaneous left ventricular assist device, for circulatory support during high-risk percutaneous coronary intervention. Int J Cardiovasc Interv 5:35–39
Smith EJ, Reitan O, Keeble T, Dixon K, Rothman MT (2009) A first-in-man study of the Reitan catheter pump for circulatory support in patients undergoing high-risk percutaneous coronary intervention. Catheter Cardiovasc Interv 73:859–865
Klotz S, Meyns B, Simon A et al (2010) Partial mechanical long-term support with the circulite synergy® pump as bridge-to-transplant in congestive heart failure. Thorac Cardiov Surg 58:173–178
Umeki A, Nishimura T, Ando M et al (2012) Alteration of LV end-diastolic volume by controlling the power of the continuous-flow LVAD, so it is synchronized with cardiac beat: development of a native heart load control system (NHLCS). J Artif Organs 15:128–133
Ando M, Takewa Y, Nishimura T et al (2011) A novel counterpulsation mode of rotary left ventricular assist devices can enhance myocardial perfusion. J Artif Organs 14:185–191
Timms D, Fraser J, Hayne M, Dunning J, McNeil K, Pearcy M (2008) The BiVACOR rotary biventricular assist device: concept and in vitro investigation. Artif Organs 32:816–827
Greatrex NA, Timms DL, Kurita N, Palmer EW, Masuzawa T (2011) Axial magnetic bearing development for the BiVACOR rotary BiVAD/TAH. Ann Biomed Eng 39:2313–2328
Abe Y, Isoyama T, Saito I et al (2007) Development of mechanical circulatory support devices at the University of Tokyo. J Artif Organs. 10:60–70
Abe Y, Isoyama T, Saito I et al (2011) Results of animal experiments with the fourth model of the undulation pump total artificial heart. Artif Organs 35:781–790
Jansen P, van Oeveren W, Capel A, Carpentier A (2012) In vitro haemocompatibility of a novel bioprosthetic total artificial heart. Eur J Cardiothorac Surg 41:e166–e172
Boyle AJ, Ascheim DD, Russo MJ et al (2011) Clinical outcomes for continuous-flow left ventricular assist device patients stratified by pre-operative INTERMACS classification. J Heart Lung Transplant 30:402–407
Acknowledgments
This study was elaborated within the grant of European Regional Development Fund—Project FNUSA-ICRC (No. CZ.1.05/1.1.00/02.0123).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ferrari, M., Kruzliak, P. & Spiliopoulos, K. An insight into short- and long-term mechanical circulatory support systems. Clin Res Cardiol 104, 95–111 (2015). https://doi.org/10.1007/s00392-014-0771-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-014-0771-6