Abstract
Background
Dabigatran etexilate, a direct thrombin inhibitor and non-vitamin K antagonist oral anticoagulant (NOAC), has been shown to effectively prevent thromboembolic events in patients with non-valvular atrial fibrillation (AF). However, there is a paucity of data on the antithrombotic efficacy and safety of dabigatran in the resolution of left atrial appendage (LAA) thrombi in AF patients.
Objective
The primary objective of the RE-LATED AF trial is to assess whether dabigatran results in a faster complete LAA thrombus resolution as compared to vitamin K antagonist phenprocoumon. Secondary objectives are to assess the impact of dabigatran on complete LAA thrombus resolution rate within 6 weeks of treatment and change in LAA thrombus volume under treatment. Furthermore, this study aims to assess and compare safety and tolerability of dabigatran vs. phenprocoumon.
Methods
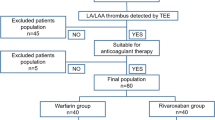
The study is designed as a prospective, randomized, open-label, controlled, explorative, blinded endpoint (PROBE) trial. Patients with AF and left atrial appendage thrombus confirmed by transoesophageal echocardiography (TEE) will be randomized to receive either dabigatran (150 mg bid) or phenprocoumon (INR 2–3) for the resolution of LAA thrombus formation for at least 21 days. Thrombus resolution will be determined by TEE 3 weeks after treatment initiation and subsequently at weeks 4 and 6, if the LAA thrombus has not been resolved before. A total of 110 patients are planned to be randomized.
Conclusion
This is the first prospective, multicentre, randomized controlled clinical trial investigating safety and efficacy of a NOAC for the resolution of LAA thrombi in patients with non-valvular AF.

Similar content being viewed by others
References
Wallace TW, Atwater BD, Daubert JP et al (2010) Prevalence and clinical characteristics associated with left atrial appendage thrombus in fully anticoagulated patients undergoing catheter-directed atrial fibrillation ablation. J Cardiovasc Electrophysiol 21:849–852
Khan MN, Usmani A, Noor S et al (2008) Low incidence of left atrial or left atrial appendage thrombus in patients with paroxysmal atrial fibrillation and normal EF who present for pulmonary vein antrum isolation procedure. J Cardiovasc Electrophysiol 19:356–358
Klein AL, Grimm RA, Murray RD et al (2001) Use of transesophageal echocardiography to guide cardioversion in patients with atrial fibrillation. N Engl J Med 344:1411–1420
Wasmer K, Kobe J, Dechering D et al (2013) CHADS(2) and CHA(2)DS (2)-VASc score of patients with atrial fibrillation or flutter and newly detected left atrial thrombus. Clin Res Cardiol Off J German Cardiac Soc 102:139–144
Stoddard MF, Singh P, Dawn B, Longaker RA (2003) Left atrial thrombus predicts transient ischemic attack in patients with atrial fibrillation. Am Heart J 145:676–682
European Heart Rhythm A, European Association for Cardio-Thoracic S, Camm AJ et al (2010) Guidelines for the management of atrial fibrillation: the task force for the management of atrial fibrillation of the European society of Cardiology (ESC). Eur Heart J 31:2369–2429
Hohnloser SH, Oldgren J, Yang S et al (2012) Myocardial ischemic events in patients with atrial fibrillation treated with dabigatran or warfarin in the RE-LY (randomized evaluation of long-term anticoagulation therapy) trial. Circulation 125:669–676
Hirsh J, Fuster V, Ansell J, Halperin JL (2003) American Heart Association/American College of Cardiology F. American Heart Association/American College of Cardiology Foundation guide to warfarin therapy. J Am Coll Cardiol 41:1633–1652
Lip GY, Ramsay SG (2012) Insights from the RCPE UK consensus conference on approaching the comprehensive management of atrial fibrillation. Exp Rev Cardiovasc Ther 10:697–700
Skeppholm M, Friberg L (2014) Adherence to warfarin treatment among patients with atrial fibrillation. Clin Res Cardiol Off J German Cardiac Soc 103:998–1005
Camm AJ, Lip GY, De Caterina R et al (2012) 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation. Developed with the special contribution of the European Heart Rhythm Association. Eur Heart J 33:2719–2747
Caldeira D, Costa J, Ferreira JJ, Lip GY, Pinto FJ (2015) Non-vitamin K antagonist oral anticoagulants in the cardioversion of patients with atrial fibrillation: systematic review and meta-analysis. Clin Res Cardiol Off J German Cardiac Soc. doi:10.1007/s00392-015-0821-8
Nielsen PB, Lane DA, Rasmussen LH, Lip GY, Larsen TB (2015) Renal function and non-vitamin K oral anticoagulants in comparison with warfarin on safety and efficacy outcomes in atrial fibrillation patients: a systemic review and meta-regression analysis. Clin Res Cardiol Off J German Cardiac Soc 104:418–429
Ruff CT, Giugliano RP, Braunwald E et al (2014) Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet 383:955–962
Zylla MM, Pohlmeier M, Hess A et al (2015) Prevalence of intracardiac thrombi under phenprocoumon, direct oral anticoagulants (dabigatran and rivaroxaban), and bridging therapy in patients with atrial fibrillation and flutter. Am J Cardiol 115:635–640
Seidl K, Rameken M, Drogemuller A et al (2002) Embolic events in patients with atrial fibrillation and effective anticoagulation: value of transesophageal echocardiography to guide direct-current cardioversion. Final results of the Ludwigshafen Observational Cardioversion Study. J Am Coll Cardiol 39:1436–1442
Collins LJ, Silverman DI, Douglas PS, Manning WJ (1995) Cardioversion of nonrheumatic atrial fibrillation. Reduced thromboembolic complications with 4 weeks of precardioversion anticoagulation are related to atrial thrombus resolution. Circulation 92:160–163
Corrado G, Tadeo G, Beretta S et al (1999) Atrial thrombi resolution after prolonged anticoagulation in patients with atrial fibrillation. Chest 115:140–143
Jaber WA, Prior DL, Thamilarasan M et al (2000) Efficacy of anticoagulation in resolving left atrial and left atrial appendage thrombi: a transesophageal echocardiographic study. Am Heart J 140:150–156
Saeed M, Rahman A, Afzal A et al (2006) Role of transesophageal echocardiography guided cardioversion in patients with atrial fibrillation, previous left atrial thrombus and effective anticoagulation. Int J Cardiol 113:401–405
Fukuda S, Watanabe H, Shimada K et al (2011) Left atrial thrombus and prognosis after anticoagulation therapy in patients with atrial fibrillation. J Cardiol 58:266–277
Husted S, de Caterina R, Andreotti F et al (2014) Non-vitamin K antagonist oral anticoagulants (NOACs): no longer new or novel. Thromb Haemost 111:781–782
Vidal A, Vanerio G (2012) Dabigatran and left atrial appendage thrombus. J Thromb Thrombolysis 34:545–547
Nagamoto Y, Shiomi T, Sadamatsu K (2013) Thrombolytic action of dabigatran in patients with acute pre-existing atrial thrombus. Europace European pacing, arrhythmias, and cardiac electrophysiology. J Working Groups Cardiac Pacing Arrhythmias Cardiac Cell Electrophysiol Eur Soc Cardiol 15:1608
Morita S, Ajiro Y, Uchida Y, Iwade K (2013) Dabigatran for left atrial thrombus. Eur Heart J 34:2745
Hammerstingl C, Potzsch B, Nickenig G (2013) Resolution of giant left atrial appendage thrombus with rivaroxaban. Thromb Haemost 109:583–584
Kawakami T, Kobayakawa H, Ohno H, Tanaka N, Ishihara H (2013) Resolution of left atrial appendage thrombus with apixaban. Thrombosis J 11:26
Dobashi S, Fujino T, Ikeda T (2014) Use of apixaban for an elderly patient with left atrial thrombus. BMJ Case Rep 2014
Takasugi J, Yamagami H, Okata T, Toyoda K, Nagatsuka K (2013) Dissolution of the left atrial appendage thrombus with rivaroxaban therapy. Cerebrovasc Dis 36:322–323
Nagarakanti R, Ezekowitz MD, Oldgren J et al (2011) Dabigatran versus warfarin in patients with atrial fibrillation: an analysis of patients undergoing cardioversion. Circulation 123:131–136
Lip GY, Hammerstingl C, Marin F et al (2015) Rationale and design of a study exploring the efficacy of once-daily oral rivaroxaban (X-TRA) on the outcome of left atrial/left atrial appendage thrombus in nonvalvular atrial fibrillation or atrial flutter and a retrospective observational registry providing baseline data (CLOT-AF). Am Heart J 169(464–71):e2
Bernhardt P, Schmidt H, Hammerstingl C et al (2004) Fate of left atrial thrombi in patients with atrial fibrillation determined by transesophageal echocardiography and cerebral magnetic resonance imaging. Am J Cardiol 94:801–804
Ammollo CT, Semeraro F, Incampo F, Semeraro N, Colucci M (2010) Dabigatran enhances clot susceptibility to fibrinolysis by mechanisms dependent on and independent of thrombin-activatable fibrinolysis inhibitor. JTH 8:790–798
Schulman S, Kearon C (2005) Subcommittee on control of anticoagulation of the S, Standardization Committee of the International Society on T, Haemostasis. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients. JTH 3:692–694
Otto C (2002) The practice of clinical echocardiography, 2nd edn. WB Saunders Co, Philadelphia
Fatkin D, Loupas T, Jacobs N, Feneley MP (1995) Quantification of blood echogenicity: evaluation of a semiquantitative method of grading spontaneous echo contrast. Ultrasound Med Biol 21:1191–1198
Schulman S, Kearon C, Kakkar AK et al (2009) Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med 361:2342–2352
Connolly SJ, Ezekowitz MD, Yusuf S et al (2009) Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 361:1139–1151
Acknowledgments
RE-LATED AF is an investigator-initiated trial (IIT) and supported by a grant of Boehringer Ingelheim Pharma GmbH & Co. KG to and by an additional research grant provided by the University Medical Center of the Johannes Gutenberg-University Mainz. This clinical trial is performed under the patronage of the German Competence Network on Atrial Fibrillation (AFNET e.V.; http://www.kompetenznetz-vorhofflimmern.de/en/home) as RE-LATED AF – AFNET 7 and in close collaboration with AFNET e.V. members and its clinical trial sites. Principal investigator: Thomas Münzel, Mainz. Members of the Steering Committee: Günter Breithardt, Münster (Chairman), Lars Eckardt, Münster, Thomas Rostock, Mainz, Monika Seibert-Grafe, Mainz. Independent Data Monitoring Committee: Dietrich Andresen, Berlin, Wolfgang Köpcke, Münster. Core Laboratories: Andreas Hagendorff, Leipzig, Stephan von Bardeleben, Mainz. We would like to thank all members of these committees as well as all investigators for their contribution to the trial. We would also like to thank members of the BMBF-funded IZKS (Funding No.: 01KN1103) for regulatory work, monitoring, data and safety management.
Conflict of interest
G. Breithardt has previously received educational funds for the AFNET from Boehringer Ingelheim, and he has been on advisory boards of this company. In addition, he has been on advisory boards of Bayer Health Care, Johnson & Johnson, BMS/Pfizer, MSD, Portola, and has received research grants from Meda Pharma, Biosense, St. Jude Medical, Sanofi-Aventis, BMS/Pfizer, and Daiichi-Sankyo, all either provided to the Westfälische Wilhelms-Universität for work done by AFNET or directly to AFNET e.V., Münster, Germany.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ferner, M., Wachtlin, D., Konrad, T. et al. Rationale and design of the RE-LATED AF—AFNET 7 trial: REsolution of Left atrial-Appendage Thrombus—Effects of Dabigatran in patients with Atrial Fibrillation. Clin Res Cardiol 105, 29–36 (2016). https://doi.org/10.1007/s00392-015-0883-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-015-0883-7