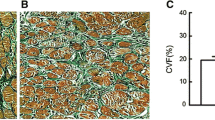
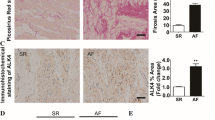
Abstract
Atrial fibrillation (AF) is associated with atrial fibrosis. Inhibition of atrial fibrosis might be a plausible approach for AF prevention and therapy. This study is designed to evaluate the potential role of CD44, a membrane receptor known to regulate fibrosis, and its related signaling in the pathogenesis of atrial fibrosis and AF. Treatment of cultured rat atrial fibroblasts with transforming growth factor-β (TGF-β, a key mediator of atrial fibrosis) led to a higher expression of hyaluronan (HA), CD44, STAT3, and collagen (a principal marker of fibrosis) than that of ventricular fibroblasts. In vivo, TGF-β transgenic mice and AF patients exhibited a greater expression of HA, CD44, STAT3, and collagen in their atria than wild-type mice and sinus rhythm subjects, respectively. Treating TGF-β transgenic mice with an anti-CD44 blocking antibody resulted in a lower expression of STAT3 and collagen in their atria than those with control IgG antibody. Programmed stimulation triggered less AF episodes in TGF-β transgenic mice treated with anti-CD44 blocking antibody than in those with control IgG. Blocking CD44 signaling with anti-CD44 antibody and mutated CD44 plasmids attenuated TGF-β-induced STAT3 activation and collagen expression in cultured atrial fibroblasts. Deletion and mutational analysis of the collagen promoter along with chromatin immunoprecipitation demonstrated that STAT3 served as a vital transcription factor in collagen expression. TGF-β-mediated HA/CD44/STAT3 pathway plays a crucial role in the development of atrial fibrosis and AF. Blocking CD44-dependent signaling may be a feasible way for AF management.








Similar content being viewed by others
References
Bourguignon LY, Singleton PA, Zhu H, Zhou B (2002) Hyaluronan promotes signaling interaction between CD44 and the transforming growth factor beta receptor I in metastatic breast tumor cells. J Biol Chem 277:39703–39712. doi:10.1074/jbc.M204320200
Burstein B, Libby E, Calderone A, Nattel S (2008) Differential behaviors of atrial versus ventricular fibroblasts. A potential role for platelet-derived growth factor in atrial-ventricular remodeling differences. Circulation 117:1630–1641. doi:10.1161/CIRCULATIONAHA.107.748053
Burstein B, Nattel S (2008) Atrial fibrosis: mechanism and clinical relevance in atrial fibrillation. J Am Coll Cardiol 51:802–809. doi:10.1016/j.jacc.2007.09.064
Chen Y, Surinkaew S, Naud P, Qi XY, Gillis MA, Shi YF, Tardif JC, Dobrev D, Nattel S (2017) JAK-STAT signalling and the atrial fibrillation promoting fibrotic substrate. Cardiovasc Res 113:310–320. doi:10.1093/cvr/cvx004
Hanna N, Cardin S, Leung TK, Nattel S (2004) Differences in atrial versus ventricular remodeling in dogs with ventricular tachypacing-induced congestive heart failure. Cardiovasc Res 63:236–244. doi:10.1016/j.cardiores.2004.03.026
Hoffmann S, Clauss S, Berger IM, Weiß B, Montalbano A, Röth R, Bucher M, Klier I, Wakili R, Seitz H, Schulze-Bahr E, Katus HA, Flachsbart F, Nebel A, Guenther SP, Bagaev E, Rottbauer W, Kääb S, Just S, Rappold GA (2016) Coding and non-coding variants in the SHOX2 gene in patients with early-onset atrial fibrillation. Basic Res Cardiol 111:36. doi:10.1007/s00395-016-0557-2
Hong CS, Cho MC, Kwak YG, Song CH, Lee YH, Lim JS, Kwon YK, Chae SW, Kim DH (2002) Cardiac remodeling and atrial fibrillation in transgenic mice overexpressing junction. FASEB J 16:1310–1312. doi:10.1096/fj.01-0908fje
Huebener P, Abou-Khamis T, Zymek P, Bujak M, Ying X, Chatila K, Haudek S, Thakker G, Frangogiannis NG (2008) CD44 is critically involved in infarct healing by regulating the inflammatory and fibrotic response. J Immunol 180:2625–2633. doi:10.4049/jimmunol.180.4.2625
Ito T, Williams JD, Fraser D, Phillips AO (2004) Hyaluronan attenuates transforming growth factor-beta1-mediated signaling in renal proximal tubular epithelial cells. Am J Pathol 164:1979–1988. doi:10.1016/S0002-9440(10)63758-3
Ito T, Williams JD, Fraser DJ, Phillips AO (2004) Hyaluronan regulates transforming growth factor-beta1 receptor compartmentalization. J Biol Chem 279:25326–25332. doi:10.1074/jbc.M403135200
Jin L, Hope KJ, Zhai Q, Smadja-Joffe F, Dick JE (2006) Targeting of CD44 eradicates human acute myeloid leukemic stem cells. Nat Med 12:1167–1174. doi:10.1038/nm1483
Kers J, Xu-Dubois YC, Rondeau E, Claessen N, Idu MM, Roelofs JJ, Bemelman FJ, ten Berge IJ, Florquin S (2010) Intragraft tubular vimentin and CD44 expression correlate with long-term renal allograft function and interstitial fibrosis and tubular atrophy. Transplantation 90:502–509. doi:10.1097/TP.0b013e3181e86b42
Khurana SS, Riehl TE, Moore BD, Fassan M, Rugge M, Romero-Gallo J, Noto J, Peek RM Jr, Stenson WF, Mills JC (2013) The hyaluronic acid receptor CD44 coordinates normal and metaplastic gastric epithelial progenitor cell proliferation. J Biol Chem 288:16085–16097. doi:10.1074/jbc.M112.445551
Krause DS, Lazarides K, von Andrian UH, Van Etten RA (2006) Requirement for CD44 in homing and engraftment of BCR-ABL-expressing leukemic stem cells. Nat Med 12:1175–1180. doi:10.1038/nm1489
Lee JL, Wang MJ, Chen JY (2009) Acetylation and activation of STAT3 mediated nuclear translocation of CD44. J Cell Biol 185:949–957. doi:10.1083/jcb.200812060
Levy DE, Darnell JE Jr (2002) Stats: transcriptional control and biological impact. Nat Rev Mol Cell Biol 3:651–662. doi:10.1038/nrm909
Li J, Gorski DJ, Anemaet W, Velasco J, Takeuchi J, Sandy JD, Plaas A (2012) Hyaluronan injection in murine osteoarthritis prevents TGFbeta 1-induced synovial neovascularization and fibrosis and maintains articular cartilage integrity by a CD44-dependent mechanism. Arthr Res Ther 14:R151. doi:10.1186/ar3887
Li Y, Jiang D, Liang J, Meltzer EB, Gray A, Miura R, Wogensen L, Yamaguchi Y, Noble PW (2011) Severe lung fibrosis requires an invasive fibroblast phenotype regulated by hyaluronan and CD44. J Exp Med 208:1459–1471. doi:10.1084/jem.20102510
Meran S, Luo DD, Simpson R, Martin J, Wells A, Steadman R, Phillips AO (2011) Hyaluronan facilitates transforming growth factor-β1-dependent proliferation via CD44 and epidermal growth factor receptor interaction. J Biol Chem 286:17618–17630. doi:10.1074/jbc.M111.226563
Meran S, Thomas DW, Stephens P, Enoch S, Martin J, Steadman R, Phillips AO (2008) Hyaluronan facilitates transforming growth factor-beta1-mediated fibroblast proliferation. J Biol Chem 283:6530–6545. doi:10.1074/jbc.M704819200
Mikecz K, Brennan FR, Kim JH, Glant TT (1995) Anti-CD44 treatment abrogates tissue oedema and leukocyte infiltration in murine arthritis. Nat Med 1:558–563
Misra S, Hascalll VC, Markwald RR, Ghatak S (2015) Interactions between hyaluronan and its receptors (CD44, RHAMM) regulate the activities of inflammation and cancer. Front Immunol 6:201. doi:10.3389/fimmu.2015.00201
Müller J, Gorressen S, Grandoch M, Feldmann K, Kretschmer I, Lehr S, Ding Z, Schmitt JP, Schrader J, Garbers C, Heusch G, Kelm M, Scheller J, Fischer JW (2014) Interleukin-6-dependent phenotypic modulation of cardiac fibroblasts after acute myocardial infarction. Basic Res Cardiol 109:440. doi:10.1007/s00395-014-0440-y
Nakajima H, Nakajima HO, Salcher O, Dittie AS, Dembowsky K, Jing S, Field LJ (2000) Atrial but not ventricular fibrosis in mice expressing a mutant transforming growth factor-beta(1) transgene in the heart. Circ Res 86:571–579. doi:10.1161/01.RES.86.5.571
Nataatmadja M, West J, West M (2006) Overexpression of transforming growth factor-beta is associated with increased hyaluronan content and impairment of repair in Marfan syndrome aortic aneurysm. Circulation 114(Suppl I):I371–I377. doi:10.1161/CIRCULATIONAHA.105.000927
Ponta H, Sherman L, Herrlich PA (2003) CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol 4:33–45. doi:10.1038/nrm1004
Rameshwar P, Chang VT, Gascón P (1996) Implication of CD44 in adhesion-mediated overproduction of TGF-beta and IL-1 in monocytes from patients with bone marrow fibrosis. Br J Haematol 93:22–29. doi:10.1046/j.1365-2141.1996.4631004.x
Riechelmann H, Sauter A, Golze W, Hanft G, Schroen C, Hoermann K, Erhardt T, Gronau S (2008) Phase I trial with the CD44v6-targeting immunoconjugate bivatuzumab mertansine in head and neck squamous cell carcinoma. Oral Oncol 44:823–829. doi:10.1016/j.oraloncology.2007.10.009
Rupp U, Schoendorf-Holland E, Eichbaum M, Schuetz F, Lauschner I, Schmidt P, Staab A, Hanft G, Huober J, Sinn HP, Sohn C, Schneeweiss A (2007) Safety and pharmacokinetics of bivatuzumab mertansine in patients with CD44v6-positive metastatic breast cancer: final results of a phase I study. Anticancer Drugs 18:477–485. doi:10.1097/CAD.0b013e32801403f4
Schotten U, Verheule S, Kirchhof P, Goette A (2011) Pathophysiological mechanisms of atrial fibrillation: a translational appraisal. Physiol Rev 91:265–325. doi:10.1152/physrev.00031.2009
Sherman LS, Matsumoto S, Su W, Srivastava T, Back SA (2015) Hyaluronan synthesis, catabolism, and signaling in neurodegenerative diseases. Int J Cell Biol 2015:368584. doi:10.1155/2015/368584
So JY, Smolarek AK, Salerno DM, Maehr H, Uskokovic M, Liu F, Suh N (2013) Targeting CD44-STAT3 signaling by Gemini vitamin D analog leads to inhibition of invasion in basal-like breast cancer. PLoS ONE 8:e54020. doi:10.1371/journal.pone.0054020
Stuhlmeier KM, Pollaschek C (2004) Differential effect of transforming growth factor beta (TGF-beta) on the genes encoding hyaluronan synthases and utilization of the p38 MAPK pathway in TGF-beta-induced hyaluronan synthase 1 activation. J Biol Chem 279:8753–8760. doi:10.1074/jbc.M303945200
Su YJ, Lai HM, Chang YW, Chen GY, Lee JL (2011) Direct reprogramming of stem cell properties in colon cancer cells by CD44. EMBO J 30:3186–3199. doi:10.1038/emboj.2011.211
Sun Z, Zhou D, Xie X, Wang S, Wang Z, Zhao W, Xu H, Zheng L (2016) Cross-talk between macrophages and atrial myocytes in atrial fibrillation. Basic Res Cardiol 111:63
Toole BP (2009) Hyaluronan-CD44 interactions in cancer: paradoxes and possibilities. Clin Cancer Res 15:7462–7468. doi:10.1158/1078-0432.CCR-09-0479
Tsai CT, Lai LP, Kuo KT, Hwang JJ, Hsieh CS, Hsu KL, Tseng CD, Tseng YZ, Chiang FT, Lin JL (2008) Angiotensin II activates signal transducer and activators of transcription 3 via Rac1 in atrial myocytes and fibroblasts. Circulation 117:344–355. doi:10.1161/CIRCULATIONAHA.107.695346
Verheule S, Sato T, Everett T, Engle SK, Otten D, der Rubart-von LM, Nakajima HO, Nakajima H, Field LJ, Olgin JE (2004) Increased vulnerability to atrial fibrillation in transgenic mice with selective atrial fibrosis caused by overexpression of TGF-beta1. Circ Res 94:1458–1465. doi:10.1161/01.RES.0000129579.59664.9d
Wang Q, Yu Y, Zhang P, Chen Y, Li C, Chen J, Wang Y, Li Y (2017) The crucial role of activin A/ALK4 pathway in the pathogenesis of Ang-II-induced atrial fibrosis and vulnerability to atrial fibrillation. Basic Res Cardiol 112:47. doi:10.1007/s00395-017-0634-1
Xiao HD, Fuchs S, Campbell DJ, Lewis W, Dudley SC Jr, Kasi VS, Hoit BD, KeshelavaG Zhao H, Capecchi MR, Bernstein KE (2004) Mice with cardiac restricted angiotensin-converting enzyme (ACE) have atrial enlargement, cardiac arrhythmia, and sudden death. Am J Pathol 165:1019–1032. doi:10.1016/S0002-9440(10)63363-9
Yeh YH, Hsu LA, Chen YH, Kuo CT, Chang GJ, Chen WJ (2016) Protective role of heme oxygenase-1 in atrial remodeling. Basic Res Cardiol 111:58. doi:10.1007/s00395-016-0577-y
Yeh YH, Kuo CT, Chang GJ, Chen YH, Lai YJ, Cheng ML, Chen WJ (2015) Rosuvastatin suppresses atrial tachycardia-induced cellular remodeling via Akt/Nrf2/heme oxygenase-1 pathway. J Mol Cell Cardiol 82:84–92. doi:10.1016/j.yjmcc.2015.03.004
Yeh YH, Kuo CT, Chang GJ, Qi XY, Nattel S, Chen WJ (2013) Nicotinamide adenine dinucleotide phosphate oxidase 4 mediates the differential responsiveness of atrial versus ventricular fibroblasts to transforming growth factor-β. Circ Arrhythm Electrophysiol 6:790–798. doi:10.1161/CIRCEP.113.000338
Zheng Z, Katoh S, He Q, Oritani K, Miyake K, Lesley J, Hyman R, Hamik A, Parkhouse RM, Farr AG, Kincade PW (1995) Monoclonal antibodies to CD44 and their influence on hyaluronan recognition. J Cell Biol 130:485–495. doi:10.1083/jcb.130.2.485
Acknowledgements
We thank Mr. Chih-Chun Chen for his technical assistance in confocal microscopy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by grants from Chang Gung Research Grant Foundation (CMRPG 3B1691-3, 3D1331-3, 3D1631-3, 3F0991-3, and 3F2281) and from Ministry of Science and Technology, Taiwan (102-2628-B-182-011-MY3, 104-2314-B-182A-135-MY2, and 104-2314-B-182-052-MY3).
Conflict of interest
US patent is issued for the method in treating and/or preventing AF patients with a pharmaceutical composition containing an anti-CD44 neutralizing antibody or an antigen binding portion.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chang, SH., Yeh, YH., Lee, JL. et al. Transforming growth factor-β-mediated CD44/STAT3 signaling contributes to the development of atrial fibrosis and fibrillation. Basic Res Cardiol 112, 58 (2017). https://doi.org/10.1007/s00395-017-0647-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-017-0647-9