Abstract
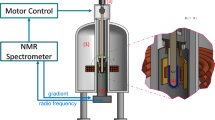
We discuss the potential insights gained from 1H nuclear magnetic resonance (NMR) spectroscopy experiments on polymeric systems under both shear and extension, and we show in particular that 1H spin-spin relaxation is sensitive to both molecular conformation and to molecular interactions. Rheo-NMR 1H spectroscopy studies on semi-dilute solutions of polyacrylamide demonstrate that the chain protons exhibit a marked T 2 reduction under shear and that the recovery on shear cessation is indicative of slow reorganisational dynamics. Studies of the wheat flour protein, gluten, indicate marked spectroscopic changes in the vicinity of the amidic resonances associated with glutamine residues, an effect we attribute to the disruption of hydrogen bonding.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 23 June 1999/Accepted: 23 August 1999
Rights and permissions
About this article
Cite this article
Callaghan, P., Gil, A. 1H NMR spectroscopy of polymers under shear and extensional flow. Rheol. Acta 38, 528–536 (1999). https://doi.org/10.1007/s003970050205
Issue Date:
DOI: https://doi.org/10.1007/s003970050205