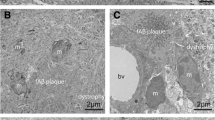
Abstract
Rapidly progressive Alzheimer’s disease (rpAD) is a particularly aggressive form of Alzheimer’s disease, with a median survival time of 7–10 months after diagnosis. Why these patients have such a rapid progression of Alzheimer’s disease is currently unknown. To further understand pathological differences between rpAD and typical sporadic Alzheimer’s disease (sAD) we used localized proteomics to analyze the protein differences in amyloid plaques in rpAD and sAD. Label-free quantitative LC–MS/MS was performed on amyloid plaques microdissected from rpAD and sAD patients (n = 22 for each patient group) and protein expression differences were quantified. On average, 913 ± 30 (mean ± SEM) proteins were quantified in plaques from each patient and 279 of these proteins were consistently found in plaques from every patient. We found significant differences in protein composition between rpAD and sAD plaques. We found that rpAD plaques contained significantly higher levels of neuronal proteins (p = 0.0017) and significantly lower levels of astrocytic proteins (p = 1.08 × 10−6). Unexpectedly, cumulative protein differences in rpAD plaques did not suggest accelerated typical sAD. Plaques from patients with rpAD were particularly abundant in synaptic proteins, especially those involved in synaptic vesicle release, highlighting the potential importance of synaptic dysfunction in the accelerated development of plaque pathology in rpAD. Combined, our data provide new direct evidence that amyloid plaques do not all have the same protein composition and that the proteomic differences in plaques could provide important insight into the factors that contribute to plaque development. The cumulative protein differences in rpAD plaques suggest rpAD may be a novel subtype of Alzheimer’s disease.




Similar content being viewed by others
References
Andreev VP, Petyuk VA, Brewer HM, Karpievitch YV, Xie F, Clarke J, Camp D, Smith RD, Lieberman AP, Albin RL et al (2012) Label-free quantitative LC-MS proteomics of Alzheimer’s disease and normally aged human brains. J Proteom Res 11:3053–3067
Bai B, Hales CM, Chen PC, Gozal Y, Dammer EB, Fritz JJ, Wang X, Xia Q, Duong DM, Street C et al (2013) U1 small nuclear ribonucleoprotein complex and RNA splicing alterations in Alzheimer’s disease. Proc Natl Acad Sci USA 110:16562–16567
Beck K, Brodsky B (1998) Supercoiled protein motifs: the collagen triple-helix and the alpha-helical coiled coil. J Struct Biol 122:17–29. doi:10.1006/jsbi.1998.3965
Begcevic I, Kosanam H, Martinez-Morillo E, Dimitromanolakis A, Diamandis P, Kuzmanov U, Hazrati LN, Diamandis EP (2013) Semiquantitative proteomic analysis of human hippocampal tissues from Alzheimer’s disease and age-matched control brains. Clin Proteom 10:5
Bera TK, Huynh N, Maeda H, Sathyanarayana BK, Lee B, Pastan I (2004) Five POTE paralogs and their splice variants are expressed in human prostate and encode proteins of different lengths. Gene 337:45–53. doi:10.1016/j.gene.2004.05.009
Bera TK, Zimonjic DB, Popescu NC, Sathyanarayana BK, Kumar V, Lee B, Pastan I (2002) POTE, a highly homologous gene family located on numerous chromosomes and expressed in prostate, ovary, testis, placenta, and prostate cancer. Proc Natl Acad Sci USA 99:16975–16980. doi:10.1073/pnas.262655399
Bignami A, LeBlanc A, Perides G (1994) A role for extracellular matrix degradation and matrix metalloproteinases in senile dementia? Acta Neuropathol 87:308–312
Bilousova T, Miller CA, Poon WW, Vinters HV, Corrada M, Kawas C, Hayden EY, Teplow DB, Glabe C, Albay R (2016) Synaptic amyloid-beta oligomers precede p-tau and differentiate high pathology control cases. Am J Pathol 186:185–198. doi:10.1016/j.ajpath.2015.09.018
Blanchard V, Moussaoui S, Czech C, Touchet N, Bonici B, Planche M, Canton T, Jedidi I, Gohin M, Wirths O et al (2003) Time sequence of maturation of dystrophic neurites associated with Abeta deposits in APP/PS1 transgenic mice. Exp Neurol 184:247–263
Bronsert P, Weisser J, Biniossek ML, Kuehs M, Mayer B, Drendel V, Timme S, Shahinian H, Kusters S, Wellner UF et al (2014) Impact of routinely employed procedures for tissue processing on the proteomic analysis of formalin-fixed paraffin-embedded tissue. Proteom Clin Appl 8:796–804
Castano EM, Maarouf CL, Wu T, Leal MC, Whiteside CM, Lue LF, Kokjohn TA, Sabbagh MN, Beach TG, Roher AE (2013) Alzheimer disease periventricular white matter lesions exhibit specific proteomic profile alterations. Neurochem Int 62:145–156
Chang KW, Yang PY, Lai HY, Yeh TS, Chen TC, Yeh CT (2006) Identification of a novel actin isoform in hepatocellular carcinoma. Hepatol Res 36:33–39. doi:10.1016/j.hepres.2006.05.003
Chang RY, Etheridge N, Nouwens AS, Dodd PR (2015) SWATH analysis of the synaptic proteome in Alzheimer’s disease. Neurochem Int 87:1–12. doi:10.1016/j.neuint.2015.04.004
Chen S, Lu FF, Seeman P, Liu F (2012) Quantitative proteomic analysis of human substantia nigra in Alzheimer’s disease, Huntington’s disease and multiple sclerosis. Neurochem Res 37:2805–2813
Chuang JY, Lee CW, Shih YH, Yang T, Yu L, Kuo YM (2012) Interactions between amyloid-beta and hemoglobin: implications for amyloid plaque formation in Alzheimer’s disease. PLoS One 7:e33120. doi:10.1371/journal.pone.0033120
Cine N, Baykal AT, Sunnetci D, Canturk Z, Serhatli M, Savli H (2014) Identification of ApoA1, HPX and POTEE genes by omic analysis in breast cancer. Oncol Rep 32:1078–1086. doi:10.3892/or.2014.3277
Cohen M, Appleby B, Safar JG (2016) Distinct prion-like strains of amyloid beta implicated in phenotypic diversity of Alzheimer’s disease. Prion 10:9–17. doi:10.1080/19336896.2015.1123371
Cohen ML, Kim C, Haldiman T, ElHag M, Mehndiratta P, Pichet T, Lissemore F, Shea M, Cohen Y, Chen W et al (2015) Rapidly progressive Alzheimer’s disease features distinct structures of amyloid-beta. Brain 138:1009–1022. doi:10.1093/brain/awv006
Cox J, Hein MY, Luber CA, Paron I, Nagaraj N, Mann M (2014) Accurate proteome-wide label-free quantification by delayed normalization and maximal peptide ratio extraction, termed MaxLFQ. Mol Cell Proteomics 13:2513–2526. doi:10.1074/mcp.M113.031591
Dickey CA, Gordon MN, Wilcock DM, Herber DL, Freeman MJ, Morgan D (2005) Dysregulation of Na +/K + ATPase by amyloid in APP + PS1 transgenic mice. BMC Neurosci 6:7. doi:10.1186/1471-2202-6-7
Dickson TC, Chuckowree JA, Chuah MI, West AK, Vickers JC (2005) alpha-Internexin immunoreactivity reflects variable neuronal vulnerability in Alzheimer’s disease and supports the role of the beta-amyloid plaques in inducing neuronal injury. Neurobiol Dis 18:286–295. doi:10.1016/j.nbd.2004.10.001
Donovan LE, Higginbotham L, Dammer EB, Gearing M, Rees HD, Xia Q, Duong DM, Seyfried NT, Lah JJ, Levey AI (2012) Analysis of a membrane-enriched proteome from postmortem human brain tissue in Alzheimer’s disease. Proteom Clin Appl 6:201–211
Drummond E, Nayak S, Ueberheide B, Wisniewski T (2015) Proteomic analysis of individual neurons isolated from formalin-fixed paraffin embedded brain sections using laser microdissection. Sci Rep 5:15456
Duong T, Pommier EC, Scheibel AB (1989) Immunodetection of the amyloid P component in Alzheimer’s disease. Acta Neuropathol 78:429–437
Ferreira ST, Lourenco MV, Oliveira MM, De Felice FG (2015) Soluble amyloid-beta oligomers as synaptotoxins leading to cognitive impairment in Alzheimer’s disease. Front Cell Neurosci 9:191. doi:10.3389/fncel.2015.00191
Finnegan S, Robson J, Hocking PM, Ali M, Inglehearn CF, Stitt A, Curry WJ (2010) Proteomic profiling of the retinal dysplasia and degeneration chick retina. Mol Vis 16:7–17
Fowler CB, O’Leary TJ, Mason JT (2013) Toward improving the proteomic analysis of formalin-fixed, paraffin-embedded tissue. Expert Rev Proteom 10:389–400
Freer R, Sormanni P, Vecchi G, Ciryam P, Dobson CM, Vendruscolo M (2016) A protein homeostasis signature in healthy brains recapitulates tissue vulnerability to Alzheimer’s disease. Sci Adv 2:e1600947. doi:10.1126/sciadv.1600947
George AJ, Holsinger RM, McLean CA, Tan SS, Scott HS, Cardamone T, Cappai R, Masters CL, Li QX (2006) Decreased phosphatidylethanolamine binding protein expression correlates with Abeta accumulation in the Tg2576 mouse model of Alzheimer’s disease. Neurobiol Aging 27:614–623. doi:10.1016/j.neurobiolaging.2005.03.014
Glenner GG, Wong CW (1984) Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem Biophys Res Commun 120:885–890
Gozal YM, Duong DM, Cheng D, Hanfelt JJ, Funderburk C, Peng J, Lah JJ, Levey AI (2009) Proteomics analysis reveals novel components in the detergent-insoluble subproteome in Alzheimer’s disease. J Proteome Res 8:5069–5079
Hadley KC, Rakhit R, Guo H, Sun Y, Jonkman JE, McLaurin J, Hazrati LN, Emili A, Chakrabartty A (2015) Determining composition of micron-scale protein deposits in neurodegenerative disease by spatially targeted optical microproteomics. Elife. doi:10.7554/eLife.09579
Hashimoto M, Bogdanovic N, Volkmann I, Aoki M, Winblad B, Tjernberg LO (2010) Analysis of microdissected human neurons by a sensitive ELISA reveals a correlation between elevated intracellular concentrations of Abeta42 and Alzheimer’s disease neuropathology. Acta Neuropathol 119:543–554
Higuchi M, Iwata N, Matsuba Y, Takano J, Suemoto T, Maeda J, Ji B, Ono M, Staufenbiel M, Suhara T et al (2012) Mechanistic involvement of the calpain-calpastatin system in Alzheimer neuropathology. FASEB J 26:1204–1217. doi:10.1096/fj.11-187740
Hirokawa N, Sobue K, Kanda K, Harada A, Yorifuji H (1989) The cytoskeletal architecture of the presynaptic terminal and molecular structure of synapsin 1. J Cell Biol 108:111–126
Ho Kim J, Franck J, Kang T, Heinsen H, Ravid R, Ferrer I, Hee Cheon M, Lee JY, Shin Yoo J, Steinbusch HW et al (2015) Proteome-wide characterization of signalling interactions in the hippocampal CA4/DG subfield of patients with Alzheimer’s disease. Sci Rep 5:11138. doi:10.1038/srep11138
Hondius DC, van Nierop P, Li KW, Hoozemans JJ, van der Schors RC, van Haastert ES, van der Vies SM, Rozemuller AJ, Smit AB (2016) Profiling the human hippocampal proteome at all pathologic stages of Alzheimer’s disease. Alzheimers Dement 12:654–668. doi:10.1016/j.jalz.2015.11.002
Kakimura J, Kitamura Y, Takata K, Umeki M, Suzuki S, Shibagaki K, Taniguchi T, Nomura Y, Gebicke-Haerter PJ, Smith MA et al (2002) Microglial activation and amyloid-beta clearance induced by exogenous heat-shock proteins. FASEB J 16:601–603
Kovacs GG, Laszlo L, Kovacs J, Jensen PH, Lindersson E, Botond G, Molnar T, Perczel A, Hudecz F, Mezo G et al (2004) Natively unfolded tubulin polymerization promoting protein TPPP/p25 is a common marker of alpha-synucleinopathies. Neurobiol Dis 17:155–162. doi:10.1016/j.nbd.2004.06.006
Kraft AW, Hu X, Yoon H, Yan P, Xiao Q, Wang Y, Gil SC, Brown J, Wilhelmsson U, Restivo JL et al (2013) Attenuating astrocyte activation accelerates plaque pathogenesis in APP/PS1 mice. FASEB J 27:187–198. doi:10.1096/fj.12-208660
Kramer A, Green J, Pollard J Jr, Tugendreich S (2014) Causal analysis approaches in ingenuity pathway analysis. Bioinformatics 30:523–530. doi:10.1093/bioinformatics/btt703
Lee Y, Ise T, Ha D, Saint Fleur A, Hahn Y, Liu XF, Nagata S, Lee B, Bera TK, Pastan I (2006) Evolution and expression of chimeric POTE-actin genes in the human genome. Proc Natl Acad Sci USA 103:17885–17890. doi:10.1073/pnas.0608344103
Liao L, Cheng D, Wang J, Duong DM, Losik TG, Gearing M, Rees HD, Lah JJ, Levey AI, Peng J (2004) Proteomic characterization of postmortem amyloid plaques isolated by laser capture microdissection. J Biol Chem 279:37061–37068
Liu XF, Bera TK, Liu LJ, Pastan I (2009) A primate-specific POTE-actin fusion protein plays a role in apoptosis. Apoptosis 14:1237–1244. doi:10.1007/s10495-009-0392-0
Manavalan A, Mishra M, Sze SK, Heese K (2013) Brain-site-specific proteome changes induced by neuronal P60TRP expression. Neurosignals 21:129–149
Masliah E, Cole GM, Hansen LA, Mallory M, Albright T, Terry RD, Saitoh T (1991) Protein kinase C alteration is an early biochemical marker in Alzheimer’s disease. J Neurosci 11:2759–2767
Mathur R, Ince PG, Minett T, Garwood CJ, Shaw PJ, Matthews FE, Brayne C, Simpson JE, Wharton SB, Function MRCC et al (2015) A reduced astrocyte response to beta-amyloid plaques in the ageing brain associates with cognitive impairment. PLoS One 10:e0118463. doi:10.1371/journal.pone.0118463
Matus A, Ackermann M, Pehling G, Byers HR, Fujiwara K (1982) High actin concentrations in brain dendritic spines and postsynaptic densities. Proc Natl Acad Sci USA 79:7590–7594
Mi Z, Halfter W, Abrahamson EE, Klunk WE, Mathis CA, Mufson EJ, Ikonomovic MD (2016) Tenascin-C Is associated with cored amyloid-beta plaques in Alzheimer disease and pathology burdened cognitively normal elderly. J Neuropathol Exp Neurol 75:868–876. doi:10.1093/jnen/nlw062
Minjarez B, Valero Rustarazo ML, Sanchez del Pino MM, Gonzalez-Robles A, Sosa-Melgarejo JA, Luna-Munoz J, Mena R, Luna-Arias JP (2013) Identification of polypeptides in neurofibrillary tangles and total homogenates of brains with Alzheimer’s disease by tandem mass spectrometry. J Alzheimers Dis 34:239–262
Montine TJ, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Dickson DW, Duyckaerts C, Frosch MP, Masliah E, Mirra SS et al (2012) National Institute on Aging-Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease: a practical approach. Acta Neuropathol 123:1–11
Morawski M, Bruckner G, Jager C, Seeger G, Matthews RT, Arendt T (2012) Involvement of perineuronal and perisynaptic extracellular matrix in Alzheimer’s disease neuropathology. Brain Pathol 22:547–561. doi:10.1111/j.1750-3639.2011.00557.x
Morris JC (1993) The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology 43:2412–2414
Musunuri S, Kultima K, Richard BC, Ingelsson M, Lannfelt L, Bergquist J, Shevchenko G (2015) Micellar extraction possesses a new advantage for the analysis of Alzheimer’s disease brain proteome. Anal Bioanal Chem 407:1041–1057. doi:10.1007/s00216-014-8320-8
Musunuri S, Wetterhall M, Ingelsson M, Lannfelt L, Artemenko K, Bergquist J, Kultima K, Shevchenko G (2014) Quantification of the brain proteome in Alzheimer’s disease using multiplexed mass spectrometry. J Proteom Res 13:2056–2068
Nakamura S, Kawamata T, Akiguchi I, Kameyama M, Nakamura N, Kimura H (1990) Expression of monoamine oxidase B activity in astrocytes of senile plaques. Acta Neuropathol 80:419–425
Ono K, Takahashi R, Ikeda T, Mizuguchi M, Hamaguchi T, Yamada M (2014) Exogenous amyloidogenic proteins function as seeds in amyloid beta-protein aggregation. Biochim Biophys Acta 1842:646–653. doi:10.1016/j.bbadis.2014.01.002
Osborn LM, Kamphuis W, Wadman WJ, Hol EM (2016) Astrogliosis: an integral player in the pathogenesis of Alzheimer’s disease. Prog Neurobiol 144:121–141. doi:10.1016/j.pneurobio.2016.01.001
Perry G, Friedman R, Shaw G, Chau V (1987) Ubiquitin is detected in neurofibrillary tangles and senile plaque neurites of Alzheimer disease brains. Proc Natl Acad Sci USA 84:3033–3036
Pickett EK, Koffie RM, Wegmann S, Henstridge CM, Herrmann AG, Colom-Cadena M, Lleo A, Kay KR, Vaught M, Soberman R et al (2016) Non-fibrillar oligomeric amyloid-beta within synapses. J Alzheimers Dis 53:787–800. doi:10.3233/JAD-160007
Picklo MJ, Olson SJ, Markesbery WR, Montine TJ (2001) Expression and activities of aldo-keto oxidoreductases in Alzheimer disease. J Neuropathol Exp Neurol 60:686–695
Potter H, Wisniewski T (2012) Apolipoprotein E: essential catalyst of the Alzheimer amyloid cascade. Int J Alz Dis 2012:489428
Power JH, Asad S, Chataway TK, Chegini F, Manavis J, Temlett JA, Jensen PH, Blumbergs PC, Gai WP (2008) Peroxiredoxin 6 in human brain: molecular forms, cellular distribution and association with Alzheimer’s disease pathology. Acta Neuropathol 115:611–622. doi:10.1007/s00401-008-0373-3
Reisberg B, Ferris SH, De Leon MJ, Crook T (1982) The global deterioration scale for assessment of primary degenerative dementia. Am J Psychiatry 139:1136–1139
Rosenberger AF, Rozemuller AJ, van der Flier WM, Scheltens P, van der Vies SM, Hoozemans JJ (2014) Altered distribution of the EphA4 kinase in hippocampal brain tissue of patients with Alzheimer’s disease correlates with pathology. Acta Neuropathol Commun 2:79. doi:10.1186/s40478-014-0079-9
Rudrabhatla P, Jaffe H, Pant HC (2011) Direct evidence of phosphorylated neuronal intermediate filament proteins in neurofibrillary tangles (NFTs): phosphoproteomics of Alzheimer’s NFTs. FASEB J 25:3896–3905
Sadleir KR, Kandalepas PC, Buggia-Prevot V, Nicholson DA, Thinakaran G, Vassar R (2016) Presynaptic dystrophic neurites surrounding amyloid plaques are sites of microtubule disruption, BACE1 elevation, and increased Abeta generation in Alzheimer’s disease. Acta Neuropathol 132:235–256. doi:10.1007/s00401-016-1558-9
Schmidt C, Haik S, Satoh K, Rabano A, Martinez-Martin P, Roeber S, Brandel JP, Calero-Lara M, de Pedro-Cuesta J, Laplanche JL et al (2012) Rapidly progressive Alzheimer’s disease: a multicenter update. J Alzheimer’s Dis JAD 30:751–756. doi:10.3233/JAD-2012-120007
Schnurra I, Bernstein HG, Riederer P, Braunewell KH (2001) The neuronal calcium sensor protein VILIP-1 is associated with amyloid plaques and extracellular tangles in Alzheimer’s disease and promotes cell death and tau phosphorylation in vitro: a link between calcium sensors and Alzheimer’s disease? Neurobiol Dis 8:900–909. doi:10.1006/nbdi.2001.0432
Serang O, Noble W (2012) A review of statistical methods for protein identification using tandem mass spectrometry. Stat Interface 5:3–20
Sergeant N, Wattez A, Galvan-valencia M, Ghestem A, David JP, Lemoine J, Sautiere PE, Dachary J, Mazat JP, Michalski JC et al (2003) Association of ATP synthase alpha-chain with neurofibrillary degeneration in Alzheimer’s disease. Neuroscience 117:293–303
Serrano-Pozo A, Betensky RA, Frosch MP, Hyman BT (2016) Plaque-associated local toxicity increases over the clinical course of Alzheimer disease. Am J Pathol 186:375–384. doi:10.1016/j.ajpath.2015.10.010
Shinohara M, Fujioka S, Murray ME, Wojtas A, Baker M, Rovelet-Lecrux A, Rademakers R, Das P, Parisi JE, Graff-Radford NR et al (2014) Regional distribution of synaptic markers and APP correlate with distinct clinicopathological features in sporadic and familial Alzheimer’s disease. Brain 137:1533–1549. doi:10.1093/brain/awu046
Sihag RK, Cataldo AM (1996) Brain beta-spectrin is a component of senile plaques in Alzheimer’s disease. Brain Res 743:249–257
Stoeck K, Schmitz M, Ebert E, Schmidt C, Zerr I (2014) Immune responses in rapidly progressive dementia: a comparative study of neuroinflammatory markers in Creutzfeldt-Jakob disease, Alzheimer’s disease and multiple sclerosis. J Neuroinflammation 11:170. doi:10.1186/s12974-014-0170-y
Stoltzner SE, Grenfell TJ, Mori C, Wisniewski KE, Wisniewski TM, Selkoe DJ, Lemere CA (2000) Temporal accrual of complement proteins in amyloid plaques in Down’s syndrome with Alzheimer’s disease. Am J Pathol 156:489–499. doi:10.1016/S0002-9440(10)64753-0
Sultana R, Boyd-Kimball D, Cai J, Pierce WM, Klein JB, Merchant M, Butterfield DA (2007) Proteomics analysis of the Alzheimer’s disease hippocampal proteome. J Alzheimers Dis 11:153–164
Sweet RA, MacDonald ML, Kirkwood CM, Ding Y, Schempf T, Jones-Laughner J, Kofler J, Ikonomovic MD, Lopez OL, Garver ME et al (2016) Apolipoprotein E*4 (APOE*4) genotype is associated with altered levels of glutamate signaling proteins and synaptic coexpression networks in the prefrontal cortex in mild to moderate Alzheimer disease. Mol Cell Proteom 15:2252–2262. doi:10.1074/mcp.M115.056580
Szklarczyk D, Jensen LJ (2015) Protein-protein interaction databases. Methods Mol Biol 1278:39–56. doi:10.1007/978-1-4939-2425-7_3
Takata K, Kitamura Y, Nakata Y, Matsuoka Y, Tomimoto H, Taniguchi T, Shimohama S (2009) Involvement of WAVE accumulation in Abeta/APP pathology-dependent tangle modification in Alzheimer’s disease. Am J Pathol 175:17–24. doi:10.2353/ajpath.2009.080908
Tanca A, Pagnozzi D, Addis MF (2012) Setting proteins free: progresses and achievements in proteomics of formalin-fixed, paraffin-embedded tissues. Proteom Clin Appl 6:7–21
Wang Q, Woltjer RL, Cimino PJ, Pan C, Montine KS, Zhang J, Montine TJ (2005) Proteomic analysis of neurofibrillary tangles in Alzheimer disease identifies GAPDH as a detergent-insoluble paired helical filament tau binding protein. FASEB J 19:869–871
Wang YJ, Chen GH, Hu XY, Lu YP, Zhou JN, Liu RY (2005) The expression of calcium/calmodulin-dependent protein kinase II-alpha in the hippocampus of patients with Alzheimer’s disease and its links with AD-related pathology. Brain Res 1031:101–108. doi:10.1016/j.brainres.2004.10.061
Wisniewski T, Frangione B (1992) Apolipoprotein E: a pathological chaperone protein in patients with cerebral and systemic amyloid. Neurosci Lett 135:235–238
Xing SL, Chen B, Shen DZ, Zhu CQ (2012) beta-amyloid peptide binds and regulates ectopic ATP synthase alpha-chain on neural surface. Int J Neurosci 122:290–297. doi:10.3109/00207454.2011.649867
Yamada T, Kawamata T, Walker DG, McGeer PL (1992) Vimentin immunoreactivity in normal and pathological human brain tissue. Acta Neuropathol 84:157–162
Yang Y, Kim J, Kim HY, Ryoo N, Lee S, Kim Y, Rhim H, Shin YK (2015) Amyloid-beta oligomers may impair SNARE-mediated exocytosis by direct binding to Syntaxin 1a. Cell Rep 12:1244–1251. doi:10.1016/j.celrep.2015.07.044
Yu JT, Tan L, Hardy J (2014) Apolipoprotein E in Alzheimer’s disease: an update. Annu Rev Neurosci 37:79–100
Zahid S, Oellerich M, Asif AR, Ahmed N (2014) Differential expression of proteins in brain regions of Alzheimers disease patients. Neurochem Res 39:208–215. doi:10.1007/s11064-013-1210-1
Zelaya MV, Perez-Valderrama E, de Morentin XM, Tunon T, Ferrer I, Luquin MR, Fernandez-Irigoyen J, Santamaria E (2015) Olfactory bulb proteome dynamics during the progression of sporadic Alzheimer’s disease: identification of common and distinct olfactory targets across Alzheimer-related co-pathologies. Oncotarget 6:39437–39456. doi:10.18632/oncotarget.6254
Zhan X, Jickling GC, Ander BP, Stamova B, Liu D, Kao PF, Zelin MA, Jin LW, DeCarli C, Sharp FR (2015) Myelin basic protein associates with AbetaPP, Abeta1-42, and amyloid plaques in cortex of Alzheimer’s disease brain. J Alzheimers Dis 44:1213–1229. doi:10.3233/JAD-142013
Zhang LH, Wang X, Stoltenberg M, Danscher G, Huang L, Wang ZY (2008) Abundant expression of zinc transporters in the amyloid plaques of Alzheimer’s disease brain. Brain Res Bull 77:55–60. doi:10.1016/j.brainresbull.2008.03.014
Zhang Y, Muller M, Xu B, Yoshida Y, Horlacher O, Nikitin F, Garessus S, Magdeldin S, Kinoshita N, Fujinaka H et al (2015) Unrestricted modification search reveals lysine methylation as major modification induced by tissue formalin fixation and paraffin embedding. Proteomics 15:2568–2579
Zhang Y, Sloan SA, Clarke LE, Caneda C, Plaza CA, Blumenthal PD, Vogel H, Steinberg GK, Edwards MS, Li G et al (2016) Purification and characterization of progenitor and mature human astrocytes reveals transcriptional and functional differences with mouse. Neuron 89:37–53. doi:10.1016/j.neuron.2015.11.013
Zhao J, Fu Y, Yasvoina M, Shao P, Hitt B, O’Connor T, Logan S, Maus E, Citron M, Berry R et al (2007) Beta-site amyloid precursor protein cleaving enzyme 1 levels become elevated in neurons around amyloid plaques: implications for Alzheimer’s disease pathogenesis. J Neurosci 27:3639–3649. doi:10.1523/JNEUROSCI.4396-06.2007
Zhou J, Jones DR, Duong DM, Levey AI, Lah JJ, Peng J (2013) Proteomic analysis of postsynaptic density in Alzheimer’s disease. Clin Chim Acta 420:62–68
Acknowledgements
The authors are grateful to the patient’s families, the CJD Foundation, and all the members of the National Prion Disease Pathology Surveillance Center for their help. This study was supported by the following Grants: NS074317, U51 CK000309, AG08051, AG20245, NS073502, the Spitz Family Foundation Grant and a Seix Dow Foundation Grant.
Author contributions
ED and TW conceived and supervised the project. ED, SN, AF, GP and RH performed the experiments. BU supervised the proteomics experiments. MA, SN and BU performed the proteomics data analysis, TW, JS, MC, TH and CK performed neuropathology, characterized and classified all cases, and sampled the brain tissue. ED, SN, MA, XH, YS, BU and TW analyzed the data. ED and TW wrote the paper with input from all authors.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
401_2017_1691_MOESM1_ESM.pdf
Supplementary material 1 (PDF 224 kb). Supplementary Figure 1: Sequence alignment of detected peptides mapped to POTEE and ACTBM. Sequence alignment was performed using Clustal Omega. Detected peptides are highlighted in yellow. Amino acid differences between POTEE and ACTBM are shown in red. The table lists identified peptides mapped to POTEE, whether each peptide is also found in ACTBM or any other actin isoforms and their corresponding average LFQ intensity values in rpAD and sAD plaques.
401_2017_1691_MOESM2_ESM.xlsx
Supplementary material 2 (XLSX 2312 kb). Supplementary Table 1: LC-MS/MS results of all proteins present in plaques. The first tab shows imputed data. All results discussed in this paper were generated using this dataset. Relative protein abundance was quantified using MaxQuant. Data shows Log2 transformed LFQ intensities with missing values imputed from normal distribution. This also contains comparison with the in-house developed Alzheimer’s protein database. ALZproteins column identifies the number of proteins in the protein group that are also present in the Alzheimer’s protein database. ALZscore lists the total number of previous studies that a protein group was significantly associated with Alzheimer’s disease in the Alzheimer’s disease database (i.e. the higher the number, the more associated with Alzheimer’s disease the protein group). ALZdetails shows the breakdown of how a protein group has been previously associated with Alzheimer’s disease; U – up-regulated in previous Alzheimer’s disease studies; D – down-regulated in previous Alzheimer’s disease studies; N – enriched in neurofibrillary tangles; P – enriched in plaques. ALZdirection gives the overall annotation for the whole protein group as either up (U) or down-regulated (D) in Alzheimer’s disease. Data in the second tab (labeled unimputed data) shows LFQ intensities of all the proteins identified with 2 or more unique + razor peptides per proteins.
401_2017_1691_MOESM3_ESM.xlsx
Supplementary material 3 (XLSX 228 kb). Supplementary Table 2: Proteins present in plaques in all 44 cases examined. The list of all quantified proteins was filtered to include only proteins that had measurable expression in all plaques before imputation. Proteins are sorted by average expression in rpAD plaques. Proteins with novel involvement in AD have an “ALZproteins” score of 0.
401_2017_1691_MOESM4_ESM.xlsx
Supplementary material 4 (XLSX 121 kb). Supplementary Table 3: Proteins with significantly altered expression in rpAD plaques. The list of all proteins with significantly altered expression in rpAD plaques, sorted by –Log Welch’s t-test p-value rpAD_sAD. Proteins with a positive Welch’s t-test difference have significantly higher expression in rpAD plaques. This also contains comparison with the in-house developed Alzheimer’s protein database (see Supplementary Table 1 figure legend for details).
401_2017_1691_MOESM5_ESM.xlsx
Supplementary material 5 (XLSX 260 kb). Supplementary Table 4: Protein expression in formic acid treated tissue blocks vs non-formic acid treated tissue blocks. Data shows Log2 transformed LFQ intensities with missing values imputed from normal distribution.
Rights and permissions
About this article
Cite this article
Drummond, E., Nayak, S., Faustin, A. et al. Proteomic differences in amyloid plaques in rapidly progressive and sporadic Alzheimer’s disease. Acta Neuropathol 133, 933–954 (2017). https://doi.org/10.1007/s00401-017-1691-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-017-1691-0