Abstract
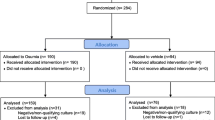
The objective of the study was to determine if the addition of topical antibiotic increases the efficacy of topical steroid in controlling otitis externa. A double-blind randomised controlled trial was performed from February 2003 to April 2005 in an otolaryngology emergency clinic (acute urban teaching hospital) in the United Kingdom. Patients were followed up for 2 weeks. Forty-five adults with otitis externa based on the presence of oedema, discharge or debris in the outer ear canal were recruited. The patients were randomised to one of the two treatment groups, namely using betamethasone sodium phosphate 0.1% (Vista-Methasone) or betamethasone sodium phosphate 0.1% with neomycin sulphate 0.5% (Vista-Methasone N), and were instructed to use the trial medication at three drops three times a day for 2 weeks. Subjects’ visual analogue symptom scores (blockage, pain, discharge, and itching) for otitis externa pre-treatment (day 0) and post-treatment (day 15), percentage changes in visual analogue symptom scores as a result of treatment, proportion of patients whose symptom scores failed to improve or deteriorated on treatment were analysed. The two experimental arms demonstrated statistically similar presenting symptom scores at recruitment (mean symptom scores of 19.2 for betamethasone group and 28.7 for betamethasone-neomycin group). The mean symptom score change in response to treatment was 82.8 and 47.8% in the betamethasone–neomycin and betamethasone-alone groups, respectively. There was no statistically significant difference between the groups in median percentage symptom score change in response to treatment. All patients in the betamethasone–neomycin group showed symptom improvement but in the betamethasone alone group, five patients got worse (Fishers exact, P = 0.05). Topical antibiotic–steroid combination therapy is superior to steroid-alone treatment for symptomatic control of otitis externa.

Similar content being viewed by others
References
Russell JD, Donnelly M, McShane DP, Alun-Jones T, Walsh M (1993) What causes acute otitis externa? J Laryngol Otol 107:898–901
Rowlands S, Devalia H, Smith C, Hubbard R, Dean A (2001) Otitis externa in UK general practice: a survey using the UK General Practice Research Database. Br J Gen Pract 51:533–538
Ruddy J, Bickerton RC (1992) Optimum management of the discharging ear. Drugs 43:219–235
Jinn TH, Kim PD, Russell PT et al (2001) Determination of ototoxicity of common otic drops using isolated cochlear outer hair cells. Laryngoscope 111:2105–2108
Smith IM, Keay DG, Buxton PK (1990) Contact hypersensitivity in patients with chronic otitis externa. Clin Otolaryngol 15:155–158
Tsikuodas A, Jasser P, England RJ (2002) Are topical antibiotics necessary in the management of otitis externa? Clin Otolaryngol 27:260–262
Alper CM, Dohar JE, Gulham M et al (2000) Treatment of chronic suppurative otitis media with topical tobramycin and dexamethasone. Arch Otolaryngol Head Neck Surg 126:165–173
Johnston MN, Flook EP, Mehta D, Mortimore S (2006) Prospective randomised single-blind controlled trial of glacial acetic acid versus glacial acetic acid, neomycin sulphate and dexamethasone spray in otitis externa and infected mastoid cavities. Clin Otolaryngol 31(6):504–507
Altman DG (1991) Practical statistics for medical research. Chapman and Hall, London, pp 440–474
Rosenfeld R, Singer M, Wasserman J, Stinnett S (2006) Systematic review of topical antimicrobial therapy for acute otitis externa. Otolaryngol-Head Neck Surg 134:S24–S48
Marais J, Rutka JA (1998) Ototoxicity and topical eardrops. Clin Otolaryngol Allied Sci 23:360–367
Joint Formulary Committee (2006) British national formulary, 51st edn edn. British Medical Association and Royal Pharmaceutical Society of Great Britain, London, p 550
Emgard P, Hellstrom S (2005) A group III steroid solution without antibiotic components: an effective cure for external otitis. J Laryngol Otol 119(5):342–347
Schwartz RH (2006) Once-daily ofloxacin otic solution versus neomycin sulfate/polymyxin B sulfate/hydrocortisone otic suspension four times a day: a multicenter, randomized, evaluator-blinded trial to compare the efficacy, safety, and pain relief in pediatric patients with otitis externa. Curr Med Res Opin 22(9):1725–1736
Acknowledgments
We are grateful to all the study participants for their cooperation. We thank Mr. V. Sankar and Mr. C. Repanos (Department of Otolaryngology, North Bristol NHS Trust, Southmead Hospital) for patient recruitment and data collection and Ms. Chaloner (Pharmacy Department, North Bristol NHS Trust, Southmead Hospital) for randomisation and pharmacy resources. D.A.N. (principal investigator) had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Conflict of interest statement
The authors have no financial obligations to disclose related to this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented as poster in 2007 AAO-HNS Annual Meeting, September 16–19, 2007, Washington DC Convention Center.
Rights and permissions
About this article
Cite this article
Abelardo, E., Pope, L., Rajkumar, K. et al. A double-blind randomised clinical trial of the treatment of otitis externa using topical steroid alone versus topical steroid–antibiotic therapy. Eur Arch Otorhinolaryngol 266, 41–45 (2009). https://doi.org/10.1007/s00405-008-0712-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-008-0712-y