Abstract
Purpose
This study aimed to investigate the association between asthma and risk of myasthenia gravis (MG) using the method of systematic review and meta-analysis.
Methods
Potentially eligible studies were identified from Medline and EMBASE databases from inception to July 2020 using search strategy that comprised terms for “Asthma” and “Myasthenia Gravis”. Eligible cohort study must consist of one cohort of individuals with asthma and another cohort of individuals without asthma. Then, the study must report relative risk (RR) with 95% confidence intervals (95% CIs) of incident MG between the groups. Eligible case–control studies must include cases with MG and controls without MG. Then, the study must explore their history of asthma. Odds ratio (OR) with 95% CIs of the association between asthma status and MG must be reported. Point estimates with standard errors were retrieved from each study and were combined together using the generic inverse variance method.
Results
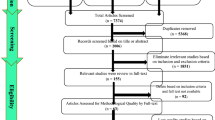
A total of 6,835 articles were identified. After two rounds of independent review by five investigators, two cohort studies and three case–control studies met the eligibility criteria and were included into the meta-analysis. Pooled analysis showed that asthma was significantly associated with risk of MG with the pooled risk ratio of 1.38 (95% CI 1.02–1.86). Funnel plot was symmetric, which was not suggestive of publication bias.
Conclusion
The current study found a significant association between asthma and increased risk of MG.



Similar content being viewed by others
Data Availability
All data and materials support the published claims and comply with field standards.
References
Gilhus NE (2016) Myasthenia gravis. N Engl J Med 375(26):2570–2581
Gilhus NE, Verschuuren JJ (2015) Myasthenia gravis: subgroup classification and therapeutic strategies. Lancet Neurol 14(10):1023–1036
Meriggioli MN, Sanders DB (2012) Muscle autoantibodies in myasthenia gravis: beyond diagnosis? Expert Rev Clin Immunol 8(5):427–438
Hehir MK, Silvestri NJ (2018) Generalized myasthenia gravis: classification, clinical presentation, natural history, and epidemiology. Neurol Clin 36(2):253–260
Berrih-Aknin S, Le Panse R (2014) Myasthenia gravis: a comprehensive review of immune dysregulation and etiological mechanisms. J Autoimmun 52:90–100
Mims JW (2015) Asthma: definitions and pathophysiology. Int Forum Allergy Rhinol 5(Suppl 1):S2–6
Maddox L, Schwartz DA (2002) The pathophysiology of asthma. Annu Rev Med 53(1):477–498
Croisant S (2014) Epidemiology of asthma: prevalence and burden of disease. Heterogeneity in asthma. Springer, Boston, pp 17–29
Sze E, Bhalla A, Nair P (2020) Mechanisms and therapeutic strategies for non-T2 asthma. Allergy 75(2):311–325
Charoenngam N et al (2021) Patients with asthma have a higher risk of systemic lupus erythematosus: a systematic review and meta-analysis. Clin Rheumatol 40(2):529–536
Shen T-C et al (2016) Risk of asthma in patients with primary Sjögren’s syndrome: a retrospective cohort study. BMC Pulm Med 16(1):152–152
Charoenngam N et al (2020) Patients with asthma have a higher risk of rheumatoid arthritis: a systematic review and meta-analysis. Semin Arthritis Rheum 50(5):968–976
Hsiao YP et al (2014) Atopic diseases and systemic lupus erythematosus: an epidemiological study of the risks and correlations. Int J Environ Res Public Health 11(8):8112–8122
Jeong HE, Jung SM, Cho SI (2018) Association between rheumatoid arthritis and respiratory allergic diseases in Korean adults: a propensity score matched case-control study. Int J Rheumatol 2018:3798124
Sheen YH et al (2018) Association of asthma with rheumatoid arthritis: a population-based case-control study. J Allergy Clin Immunol Pract 6(1):219–226
Hemminki K et al (2010) Subsequent autoimmune or related disease in asthma patients: clustering of diseases or medical care? Ann Epidemiol 20(3):217–222
Krishna MT et al (2019) Allergic diseases and long-term risk of autoimmune disorders: longitudinal cohort study and cluster analysis. Eur Respir J 54(5):1900476
Murai H et al (2004) High frequency of allergic conjunctivitis in myasthenia gravis without thymoma. J Neurol Sci 225(1–2):27–31
Tsai JD et al (2014) Increased subsequent risk of myasthenia gravis in children with allergic diseases. J Neuroimmunol 276(1–2):202–206
Yeh JH et al (2015) Higher risk of myasthenia gravis in patients with thyroid and allergic diseases: a national population-based study. Medicine 94(21):e835
Gong P-H et al (2017) Acute severe asthma with thyroid crisis and myasthenia: a case report and literature review. Clin Respir J 11(6):671–676
Satkunam N, Siddiqi ZA, Vethanayagam D (2014) Severe asthma associated with myasthenia gravis. Can Respir J. https://doi.org/10.1155/2014/914719
Souza-Machado A, Ponte E, Cruz AA (2007) Severe asthma associated with myasthenia gravis and upper airway obstruction. J Investig Allergol Clin Immunol 17(4):267–270
Wells GA et al (2000) The Newcastle–Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Oxford
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7(3):177–188
Higgins JP et al (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560
Murai H et al (1997) Expression of CD23 in the germinal center of thymus from myasthenia gravis patients. J Neuroimmunol 76(1–2):61–69
Bansal AS et al (1993) Variations in serum sCD23 in conditions with either enhanced humoral or cell-mediated immunity. Immunology 79(2):285–289
Gordon J (1992) CD23 and B cell activation. Clin Exp Allergy 22(2):199–204
Balbino B et al (2018) Approaches to target IgE antibodies in allergic diseases. Pharmacol Ther 191:50–64
Galli SJ, Tsai M (2012) IgE and mast cells in allergic disease. Nat Med 18(5):693–704
Robinson DS (2010) The role of the T cell in asthma. J Allergy Clin Immunol 126(6):1081–1091
Zhong H, Zhao C, Luo S (2019) HLA in myasthenia gravis: from superficial correlation to underlying mechanism. Autoimmun Rev 18(9):102349
Muñiz-Castrillo S, Vogrig A, Honnorat J (2020) Associations between HLA and autoimmune neurological diseases with autoantibodies. Auto Immun Highlights 11(1):2
Schoettler N et al (2019) Advances in asthma and allergic disease genetics: Is bigger always better? J Allergy Clin Immunol 144(6):1495–1506
Shiina T, Inoko H, Kulski JK (2004) An update of the HLA genomic region, locus information and disease associations: 2004. Tissue Antigens 64(6):631–649
Kontakioti E et al (2014) HLA and asthma phenotypes/endotypes: a review. Hum Immunol 75(8):930–939
Kang SW et al (2019) Promoter polymorphism (− 308G/A) of tumor necrosis factor-alpha (TNF-α) gene and asthma risk: an updated meta-analysis. Genet Test Mol Biomark 23(6):363–372
Sun H et al (2018) Associations of tumor necrosis factor-α polymorphisms with the risk of asthma: a meta-analysis. Exp Mol Pathol 105(3):411–416
Mikhail I, Grayson MH (2019) Asthma and viral infections: an intricate relationship. Ann Allergy Asthma Immunol 123(4):352–358
Le Panse R, Berrih-Aknin S (2013) Autoimmune myasthenia gravis: autoantibody mechanisms and new developments on immune regulation. Curr Opin Neurol 26(5):569–576
Gilhus NE et al (2018) Myasthenia gravis and infectious disease. J Neurol 265(6):1251–1258
Funding
Nipith Charoenngam receives the institutional research training grant from the Ruth L. Kirchstein National Research Service Award program from the National Institutes of Health (2 T32 DK 7201-42).
Author information
Authors and Affiliations
Contributions
All authors had access to the data and a role in writing the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
We declare that there is no financial or nonfinancial potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yingchoncharoen, P., Charoenngam, N., Ponvilawan, B. et al. The Association Between Asthma and Risk of Myasthenia Gravis: A Systematic Review and Meta-analysis. Lung 199, 273–280 (2021). https://doi.org/10.1007/s00408-021-00444-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-021-00444-8