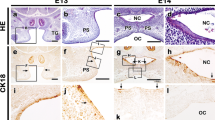
Abstract
Epithelial differentiation is thought to be determined by mesenchymal components during embryogenesis. In mice, palatal mucosa showed the region-specific keratinization pattern along antero-posterior axis. However, developmental mechanisms involved in oral mucosa differentiation with fine tuning of keratinization are not elucidated yet. To reveal this developmental mechanism, first, we conducted tissue recombination assay of the palate at E16 for 2 days which revealed that epithelial differentiation with specific localization of CK10 is modulated by mesenchymal components. Based on the results, we propose that mesenchymal signaling would determine the presumptive fate of developing palatal epithelium in spatiotemporal manner. Genome-wide screening analysis using laser micro-dissection to collect spatiotemporal specific molecules between anterior and posterior palate suggested Meox2 in the posterior mesenchymal tissue to be a candidate regulator controlling epithelial differentiation. To examine the detailed spatiotemporal function of Meox2, we employed in vitro organ cultivation with the loss- and gain-of-function studies at E14.5 for 2 and 4 days, respectively. Our results suggest that posteriorly expressed Meox2 modulates non-keratinized epithelial differentiation through complex signaling regulations in mice palatogenesis.





Similar content being viewed by others
References
Adhikari N, Neupane S, Gwon GJ, Kim JY, An CH, Lee S, Sohn WJ, Lee Y, Kim JY (2017) Grhl3 modulates epithelial structure formation of the circumvallate papilla during mouse development. Histochem Cell Biol 147:5–16
Arora R, del Alcazar CM, Morrisey EE, Naiche LA, Papaionnou VE (2012) Candidate gene approach identifies multiple genes and signaling pathways downstream of Tbx4 in the developing allantois. PLoS One 7(8):e43581
Biggs LC, Mikkola ML (2014) Early inductive events in ectodermal appendage morphogenesis. Semin Cell Dev Biol 25–26:11–21
Bongers EM, Duijf PH, van Beersum SE, Schoots J, Van Kampen A, Burckhardt A, Hamel BC, Losan F, Hoefsloot LH, Yntema HG, Knoers NV, van Bokhoven H (2004) Mutations in the human TBX4 gene cause small patella syndrome. Am J Hum Genet 74(6):1239–1248
Bush JO, Jiang R (2012) Palatogenesis: morphogenetic and molecular mechanisms of secondary palate development. Development 139:231–243
Candia AF, Hu J, Crosby J, Lalley PA, Noden D, Nadeau JH, Wright CV (1992) Mox-1 and Mox-2 define a novel homeobox gene subfamily and are differentially expressed during early mesodermal patterning in mouse embryos. Development 116:1123–1136
Deprez PM, Nichane MG, Lengele BG, Rezsohazy R, Nyssen-Behets C (2013) Molecular study of Hoxa2 gain of function in chondeogenesis: a model of idiopathic proportionate short stature. Int J Mol Sci 14(10):20386–20398
Ferguson WJ (1984) Epithelial–mesenchymal interactions during vertebrate palatogenesis. Curr Top Dev Biol 19:137–164
Fuchs E (2016) Epithelial skin biology: three decades of developmental biology, a hundred questions answered and a thousand new ones to address. Curr Top Dev Biol 116:357–374
Gorski DH, Leal AJ (2003) Inhibition of endothelial cell activation by the homeobox gene Gax. Oncology 111(1):91–99
Gritli-Linde A (2007) Molecular control of secondary palate development. Dev Biol 301:309–326
Han J, Mayo J, Xu X, Li J, Bringas P Jr, Maas RL, Rubenstein JL, Chai Y (2009) Indirect modulation of Shh signaling by Dlx5 affects the oral-nasal patterning of palate and rescues cleft palate in Msx1-null mice. Development 136:4225–4233
Hofmann C, Drossopoulou G, McMahon A, Balling R, Tickle C (1998) Inhibitory action of Bmps on Pax1 expression and on shoulder girdle formation during limb development. Dev Dyn 213(2):199–206
Ito Y, Yeo JY, Chytil A, Han J, Bringas P Jr, Nakajima A, Shuler CF, Moses HL, Chai Y (2003) Conditional inactivation of Tgfbr2 in cranial neural crest causes cleft palate and calvaria defects. Development 130:5269–5280
Jimenez-Rojo L, Granchi Z, Graf D, Mitsiadis TA (2012) Stem cell fate determination during development and regeneration of ectodermal organs. Front Physiol 3:107
Jin JZ, Ding J (2006) Analysis of Meox-2 mutant mice reveals a novel postfusion-based cleft palate. Dev Dyn 235(2):539–546
Lan Y, Xu J, Jiang R (2015) Cellular and molecular mechanisms of palatogenesis. Curr Top Dev Biol 115:59–84
Liu W, Lan Y, Pauws E, Meester-Smoor MA, Stanier P, Zwarthoff EC, Jiang R (2008) The Mn1 transcription factor acts upstream of Tbx22 and preferentially regulates posterior palate growth in mice. Development 135(23):3959–3968
Liu P, Kong F, Wang J, Lu Q, Xu H, Qi T, Meng J (2015) Involvement of IGF-1 and MEOX2 in PI3K/Akt1/2 and ERK1/2 pathways mediated proliferation and differentiation of perivascular adipocytes. Exp Cell Res 331(1):82–96
Mankoo BS, Skuntz S, Harrigan I, Grigorieva E, Candia A, Wright CV, Arnheiter H, Pachnis V (2003) The concerted action of Meox homeobox genes is required upstream of genetic pathways essential for the formation, patterning and differentiation of somites. Development 30(19):4655–4664
Nanci A (2012) Ten cate’s oral histology. Elsevier, St Louis, United States
Neupane S, Sohn WJ, Gwon GJ, Kim KR, Lee S, An CH, Suh JY, Shin HI, Yamamoto H, Cho SW, Lee Y, Kim JY (2015) The role of APCDD1 in epithelial rearrangement in tooth morphogenesis. Histochem Cell Biol 144(4):377–387
Ohshima J, Haruta M, Arai Y, Kasai F, Fujiwara Y, Ariga T, Okita H, Fukuzawa M, Hata J, Horie H, Kaneko Y (2009) Two candidate tumour suppressor genes, MEOX2 and SOSTDC1, identified in a 7p21 homozygous deletion region in Wilms tumor. Genes Chromosomes Cancer 48(12):1037–1050
Pantalacci S, Prochazka J, Martin A, Rothova M, Lambert A, Bernard L, Charles C, Viriot L, Peterkova R, Laudet V (2008) Patterning of palatal rugae through sequential addition reveals an anterior/posterior boundary in palate development. BMC Dev Biol 8:116
Patel S, Leal AD, Gorski DH (2005) The homeobox gene Gax inhibits angiogenesis through inhibition of nuclear factor-κB-dependent endothelial cell gene expression. Cancer Res 65(4):1414–1424
Pearson HB, McGlinn E, Phesse TJ, Schluter H, Srikumar A, Godde NJ, Woelwer CB, Ryan A, Phillips A, Ernst M, Kaur P, Humbert P (2015) The polarity protein Scrib mediates epidermal development and exerts a tumor suppressive function during skin carcinogenesis. Mol Cancer 14:169
Presland RB, Dale BA (2000) Epithelial structural proteins of the skin and oral cavity: function in health and disease. Crit Rev Oral Biol Med 11(4):383–408
Quinn LM, Latham SE, Kalionis B (2000) The homeobox genes MSX2 and MOX2 are candidates for regulating epithelial–mesenchymal cell interactions in the human placenta. Placenta 21(Suppl. A):S50–S54
Satokata I, Maas R (1994) Msx1 deficient mice exhibit cleft palate and abnormalities of craniofacial and tooth development. Nat Genet 6(4):348–356
Shetty S, Gokul S (2012) Keratinization and its disorders. Oman Med J 27(5):348–357
Shigetani Y, Nobusada Y, Kuratani S (2000) Ectodermally derived FGF8 defines the maxillomandibular region in the early chick embryo: epithelial–mesenchymal interactions in the specification of the craniofacial ectomesenchyme. Dev Biol 228:73–85
Smith TM, Lozanoff S, Iyyanar PP, Nazarali AJ (2012) Molecular signaling along the anterior-posterior axis of early palate development. Front Physiol 3:488
Sohn WJ, Yamamoto H, Shin HI, Ryoo ZY, Lee S, Bae YC, Jung HS, Kim JY (2011) Importance of region-specific epithelial rearrangements in mouse rugae development. Cell Tissue Res 344:271–277
Sohn WJ, Gwon KJ, Kim HS, Neupane S, Cho SJ, Lee JH, Yamamoto H, Choi JY, An CH, Lee Y, Shin HI, Lee S, Kim JY (2015) Mesenchymal signaling in dorsoventral differentiation of palatal epithelium. Cell Tissue Res 362(3):541–556
Sperber GH (2002) Palatogenesis: closure of the secondary palate. In: Wyszynski (ed) Cleft lip and palate: from origin to treatment. Oxford University Press, Oxford, pp 14–24
Stewart GA, Lowrey JA, Wakelin SJ, Fitch PA, Lindey S, Dallman MJ, Lamb JR, Howie SE (2002) Sonic hedgehog signaling modulates activation of and cytokine production by human peripheral CD4+ T cells. J Immunol 169(10):5451–5457
Szklarczyk D, Franceschini A, Wyder S, Forslund K, Heller D, Huerta-Cepas J, Simonovic M, Roth A, Santos A, Tsafou KP, Kuhn M, Bork P, Jensen LJ, von Mering C (2015) String v10: protein–protein interactions networks, integrated over the tree of life. Nucleic Acids Res 43(Database issue):D447–D452
Welsh IC, O’Brien TP (2009) Signaling intergration in the rugae zone directs sequential SHH signaling center formation during rostral outgrowth of the palate. Dev Biol 336:53–67
Welsh IC, Hagge-Greenberg A, O’Brien TP (2007) A dosage-dependent role for Spry2 in growth and patterning during palate development. Mech Dev 124(9–10):746–761
Xia S, Tai X, Wang Y, An X, Qian G, Dong J, Wang X, Sha B, Wang D, Murthi P, Kalionis B, Wang X, Bai C (2011) Involvement of Gax gene in hypoxia-induced pulmonary hypertension, proliferation, and apoptosis of arterial smooth muscle cells. Am J Respir Cell Mol Biol 44(1):66–73
Yu L, Gu S, Alappat S, Song Y, Yan M, Zhang X, Zhang G, Jiang Y, Zhang Z, Zhang Y, Chen Y (2005) Shox2-deficient mice exhibit a rare type of incomplete clefting of the secondary palate. Development 132:4397–4406
Zhang Z, Song Y, Zhao X, Zhang X, Fermin C, Chen Y (2002) Rescue of cleft palate in Msx1-deficient mice by transgenic Bmp4 reveals a network of BMP and Shh signaling in the regulation of mammalian palatogenesis. Development 129:4135–4146
Zhou J, Gao Y, Lan Y, Jia S, Jiang R (2013) Pax9 regulates a molecular network involving Bmp4, Fgf10, Shh signaling and the Osr2 transcription factor to control palate morphogenesis. Development 140(23):4709–4718
Acknowledgements
This study was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MEST) (Nos. 2017R1A5A2015391, 2010–0004243, 2012R1A1A2044113, 2014R1A1A3053711).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
418_2017_1620_MOESM1_ESM.tif
Figure S1. Scheme of LMD and putative interaction of signaling molecules. Schematic diagram of palate showing region of interest in the mesenchyme indicated by * and ** (a). Diagrammatic presentation of LMD (b). Numbers of genes regulated (c). Putative signaling interaction of up-regulated (>2 folds) genes (d). (TIF 2407 KB)
418_2017_1620_MOESM2_ESM.tif
Figure S2. Section in situ hybridization of Shox2 in the 2-day cultivated palate sections (a-f). The dotted lines indicate the basement membrane. Scale bars 50 µm. (TIF 2238 KB)
418_2017_1620_MOESM3_ESM.tif
Figure S3. Quantitative evaluation of CK10 in figure 5 (a) and the schematic diagram showing the region of histological sections presented in figure 5 (b). (TIF 5551 KB)
Rights and permissions
About this article
Cite this article
Neupane, S., Adhikari, N., Jung, JK. et al. Regulation of mesenchymal signaling in palatal mucosa differentiation. Histochem Cell Biol 149, 143–152 (2018). https://doi.org/10.1007/s00418-017-1620-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-017-1620-2