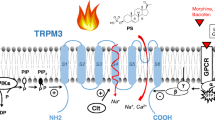
Abstract
Cooling is sensed by peripheral thermoreceptors, the main transduction mechanism of which is probably a cold- and menthol-activated ion channel, transient receptor potential (melastatin)-8 (TRPM8). Stronger cooling also activates another TRP channel, TRP (ankyrin-like)-1, (TRPA1), which has been suggested to underlie cold nociception. This review examines the roles of these two channels and other mechanisms in thermal transduction. TRPM8 is activated directly by gentle cooling and depolarises sensory neurones; its threshold temperature (normally ~26–31°C in native neurones) is very flexible and it can adapt to long-term variations in baseline temperature to sensitively detect small temperature changes. This modulation is enabled by TRPM8’s low intrinsic thermal sensitivity: it is sensitised to varying degrees by its cellular context. TRPM8 is not the only thermosensitive element in cold receptors and interacts with other ionic currents to shape cold receptor activity. Cold can also cause pain: the transduction mechanism is uncertain, possibly involving TRPM8 in some neurones, but another candidate is TRPA1 which is activated in expression systems by strong cooling. However, native neurones that appear to express TRPA1 respond very slowly to cold, and TRPA1 alone cannot account readily for cold nociceptor activity or cold pain in humans. Other, as yet unknown, mechanisms of cold nociception are likely.








Similar content being viewed by others
References
Andersson DA, Chase HW, Bevan S (2004) TRPM8 activation by menthol, icilin, and cold is differentially modulated by intracellular pH. J Neurosci 24:5364–5369
Askwith CC, Benson CJ, Welsh MJ, Snyder PM (2001) DEG/ENaC ion channels involved in sensory transduction are modulated by cold temperature. Proc Natl Acad Sci USA 98:6459–6463
Babes A, Zorzon D, Reid G (2004) Two populations of cold-sensitive neurons in rat dorsal root ganglia and their modulation by nerve growth factor. Eur J Neurosci 20:2276–2282
Babes A, Zorzon D, Reid G (2005) A novel type of cold-sensitive neurone in rat dorsal root ganglia (DRG) with rapid adaptation to cooling (abstract). J Physiol (Lond) (Proceedings) Bristol meeting July 2005
Baccaglini PI, Hogan PG (1983) Some rat sensory neurons in culture express characteristics of differentiated pain sensory cells. Proc Natl Acad Sci USA 80:594–598
Bandell M, Story GM, Hwang SW, Viswanath V, Eid SR, Petrus MJ, Earley TJ, Patapoutian A (2004) Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron 41:849–857
Behrendt HJ, Germann T, Gillen C, Hatt H, Jostock R (2004) Characterization of the mouse cold-menthol receptor TRPM8 and vanilloid receptor type-1 VR1 using a fluorometric imaging plate reader (FLIPR) assay. Br J Pharmacol 141:737–745
Bessou P, Perl ER (1969) Response of cutaneous sensory units with unmyelinated fibers to noxious stimuli. J Neurophysiol 32:1025–1043
Bonnington JK, McNaughton PA (2003) Signalling pathways involved in the sensitisation of mouse nociceptive neurones by nerve growth factor. J Physiol (Lond) 551:433–446
Brauchi S, Orio P, Latorre R (2004) Clues to understanding cold sensation: thermodynamics and electrophysiological analysis of the cold receptor TRPM8. Proc Natl Acad Sci USA 101:15494–15499
Braun HA, Bade H, Hensel H (1980) Static and dynamic discharge patterns of bursting cold fibers related to hypothetical receptor mechanisms. Arch 386:1–9
Braun HA, Wissing H, Schäfer K, Hirsch MC (1994) Oscillation and noise determine signal transduction in shark multimodal sensory cells. Nature 367:270–273
Brock J, Pianova S, Belmonte C (2001) Differences between nerve terminal impulses of polymodal nociceptors and cold sensory receptors of the guinea-pig cornea. J Physiol (Lond) 533:493–501
Burgess PR, Perl ER (1967) Myelinated afferent fibres responding specifically to noxious stimulation of the skin. J Physiol (Lond) 190:541–562
Campero M, Serra J, Bostock H, Ochoa JL (2001) Slowly conducting afferents activated by innocuous low temperature in human skin. J Physiol (Lond) 535:855–865
Carr RW, Pianova S, Brock JA (2002) The effects of polarizing current on nerve terminal impulses recorded from polymodal and cold receptors in the guinea-pig cornea. J Gen Physiol 120:395–405
Carr RW, Pianova S, Fernandez J, Fallon JB, Belmonte C, Brock JA (2003) Effects of heating and cooling on nerve terminal impulses recorded from cold-sensitive receptors in the guinea-pig cornea. J Gen Physiol 121:427–439
Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389:816–824
Cesare P, McNaughton P (1996) A novel heat-activated current in nociceptive neurons and its sensitization by bradykinin. Proc Natl Acad Sci USA 93:15435–15439
Chuang HH, Neuhausser WM, Julius D (2004) The super-cooling agent icilin reveals a mechanism of coincidence detection by a temperature-sensitive TRP channel. Neuron 43:859–869
Chuang HH, Prescott ED, Kong HY, Shields S, Jordt SE, Basbaum AI, Chao MV, Julius D (2001) Bradykinin and nerve growth factor release the capsaicin receptor from PtdIns(4,5)P2-mediated inhibition. Nature 411:957–962
Craig AD (2002) Opinion: How do you feel? Interoception: the sense of the physiological condition of the body. Nat Rev Neurosci 3:655–666
Craig AD, Bushnell MC (1994) The thermal grill illusion: unmasking the burn of cold pain. Science 265:252–255
Darian-Smith I (1984) Thermal sensibility. In: Darian-Smith I (ed) The nervous system (Handbook of physiology series, section 1). American Physiological Society, Bethesda, Maryland, pp 879–913
Darian-Smith I, Johnson KO, Dykes R (1973) “Cold” fiber population innervating palmar and digital skin of the monkey: responses to cooling pulses. J Neurophysiol 36:325–346
Davis KD (1998) Cold-induced pain and prickle in the glabrous and hairy skin. Pain 75:47–57
Davis KD, Pope GE (2002) Noxious cold evokes multiple sensations with distinct time courses. Pain 98:179–185
Djouhri L, Bleazard L, Lawson SN (1998) Association of somatic action potential shape with sensory receptive properties in guinea-pig dorsal root ganglion neurones. J Physiol (Lond) 513:857–872
Dodt E, Zotterman Y (1952) The discharge of specific cold fibres at high temperatures (the paradoxical cold). Acta Physiol Scand 26:358–365
Drummond HA, Abboud FM, Welsh MJ (2000) Localization of β and γ subunits of ENaC in sensory nerve endings in the rat foot pad. Brain Res 884:1–12
Fowler CJ, Sitzoglou K, Ali Z, Halonen P (1988) The conduction velocities of peripheral nerve fibres conveying sensations of warming and cooling. J Neurol Neurosurg Psych 51:1164–1170
Fruhstorfer H (1984) Thermal sensibility changes during ischemic nerve block. Pain 20:355–361
Georgopoulos AP (1976) Functional properties of primary afferent units probably related to pain mechanisms in primate glabrous skin. J Neurophysiol 39:71–83
Gingl E, Tichy H (2001) Infrared sensitivity of thermoreceptors. J Comp Physiol [A] 187:467–475
Hensel H (1981) Thermoreception and temperature regulation. Academic Press, London
Hensel H, Boman KKA (1960) Afferent impulses in cutaneous sensory nerves in human subjects. J Neurophysiol 23:564–578
Hensel H, Iggo A (1971) Analysis of cutaneous warm and cold fibres in primates. Pflügers Arch 329:1–8
Hensel H, Schäfer K (1974) Effects of calcium on warm and cold receptors. Pflügers Arch 352:87–90
Hensel H, Zotterman Y (1951) The effect of menthol on the thermoreceptors. Acta Physiol Scand 24:27–34
Hensel H, Zotterman Y (1951) The response of the cold receptors to constant cooling. Acta Physiol Scand 22:96–105
Hille B (2001) Ionic channels of excitable cells, 3rd ed. Sinauer Associates, Sunderland, MA
Johnson KO, Darian-Smith I, LaMotte C (1973) Peripheral neural determinants of temperature discrimination in man: a correlative study of responses to cooling skin. J Neurophysiol 36:347–370
Jordt SE, Bautista DM, Chuang HH, McKemy DD, Zygmunt PM, Hogestatt ED, Meng ID, Julius D (2004) Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 427:260–265
Jordt SE, McKemy DD, Julius D (2003) Lessons from peppers and peppermint: the molecular logic of thermosensation. Curr Opin Neurobiol 13:487–492
Kenshalo DR, Duclaux R (1977) Response characteristics of cutaneous cold receptors in the monkey. J Neurophysiol 40:319–332
Kenshalo DR, Scott HA Jr (1966) Temporal course of thermal adaptation. Science 151:1095–1096
Kim HC, Chung MK (1999) Voltage-dependent sodium and calcium currents in acutely isolated adult rat trigeminal root ganglion neurons. J Neurophysiol 81:1123–1134
Klement W, Arndt JO (1992) The role of nociceptors of cutaneous veins in the mediation of cold pain in man. J Physiol (Lond) 449:73–83
Kress M, Koltzenburg M, Reeh PW, Handwerker HO (1992) Responsiveness and functional attributes of electrically localized terminals of cutaneous C-fibers in vivo and in vitro. J Neurophysiol 68:581–595
LaMotte RH, Thalhammer JG (1982) Response properties of high-threshold cutaneous cold receptors in the primate. Brain Res 244:279–287
Levitan ES, Kramer RH (1990) Neuropeptide modulation of single calcium and potassium channels detected with a new patch clamp configuration. Nature 348:545–547
Linte R, Babes A, Reid G (2005) The increase in cold sensitivity in rat dorsal root ganglion (DRG) neurones induced by nerve growth factor (NGF) is mediated by the high affinity NGF receptor (Abstract). J Physiol (Lond) (Proceedings) Bristol meeting July 2005
Liu B, Qin F (2005) Functional control of cold- and menthol-sensitive TRPM8 ion channels by phosphatidylinositol 4,5-bisphosphate. J Neurosci 25:1674–1681
Long RR (1977) Sensitivity of cutaneous cold fibers to noxious heat: paradoxical cold discharge. J Neurophysiol 40:489–502
Mackenzie RA, Burke D, Skuse NF, Lethlean AK (1975) Fibre function and perception during cutaneous nerve block. J Neurol Neurosurg Psych 38:865–873
Maingret F, Lauritzen I, Patel AJ, Heurteaux C, Reyes R, Lesage F, Lazdunski M, Honoré E (2000) TREK-1 is a heat-activated background K+ channel. EMBO J 19:2483–2491
McKemy DD, Neuhausser WM, Julius D (2002) Identification of a cold receptor reveals a general role for TRP channels in thermosensation. Nature 416:52–58
Nealen ML, Gold MS, Thut PD, Caterina MJ (2003) TRPM8 mRNA is expressed in a subset of cold-responsive trigeminal neurons from rat. J Neurophysiol 90:515–520
Norrsell U, Finger S, Lajonchere C (1999) Cutaneous sensory spots and the “law of specific nerve energies”: history and development of ideas. Brain Res Bull 48:457–465
Okazawa M, Inoue W, Hori A, Hosokawa H, Matsumura K, Kobayashi S (2004) Noxious heat receptors present in cold-sensory cells in rats. Neurosci Lett 359:33–36
Okazawa M, Takao K, Hori A, Shiraki T, Matsumura K, Kobayashi S (2002) Ionic basis of cold receptors acting as thermostats. J Neurosci 22:3994–4001
Okazawa M, Terauchi T, Shiraki T, Matsumura K, Kobayashi S (2000) l-menthol-induced [Ca2+]i increase and impulses in cultured sensory neurons. Neuroreport 11:2151–2155
Patapoutian A, Peier AM, Story GM, Viswanath V (2003) ThermoTRP channels and beyond: mechanisms of temperature sensation. Nat Rev Neurosci 4:529–539
Peier AM, Moqrich A, Hergarden AC, Reeve AJ, Andersson DA, Story GM, Earley TJ, Dragoni I, McIntyre P, Bevan S, Patapoutian A (2002) A TRP channel that senses cold stimuli and menthol. Cell 108:705–715
Perl ER (1996) Pain and the discovery of nociceptors. In: Belmonte C, Cervero F (eds) Neurobiology of nociceptors. Oxford University Press, Oxford, pp 5–36
Petruska JC, Napaporn J, Johnson RD, Gu JG, Cooper BY (2000) Subclassified acutely dissociated cells of rat DRG: histochemistry and patterns of capsaicin-, proton-, and ATP-activated currents. J Neurophysiol 84:2365–2379
Pierau F, Torrey P, Carpenter D (1974) Mammalian cold receptor afferents: role of an electrogenic sodium pump in sensory transduction. Brain Res 73:156–160
Prescott ED, Julius D (2003) A modular PIP2 binding site as a determinant of capsaicin receptor sensitivity. Science 300:1284–1288
Reid G (2003) Doing physiology in Romania. Physiology News Summer 2003:30–32 (http://www.physoc.org/publications/pn/issuepdf/51/30.pdf)
Reid G (2004) Temperature adaptation of the cold and menthol receptor TRPM8 depends on a membrane-delimited mechanism (abstract). J Physiol (Lond) 557P:C102
Reid G, Amuzescu B, Zech E, Flonta M-L (2001) A system for applying rapid warming or cooling stimuli to cells during patch clamp recording or ion imaging. J Neurosci Methods 111:1–8
Reid G, Babes A, Pluteanu F (2002) A cold- and menthol-activated current in rat dorsal root ganglion neurones: properties and role in cold transduction. J Physiol (Lond) 545:595–614
Reid G, Flonta M-L (2001) Cold current in thermoreceptive neurons. Nature 413:480
Reid G, Flonta M-L (2001) Cold transduction by inhibition of a background potassium conductance in rat primary sensory neurones. Neurosci Lett 297:171–174
Reid G, Flonta M-L (2002) Ion channels activated by cold and menthol in cultured rat dorsal root ganglion neurones. Neurosci Lett 324:164–168
Reid G, Zorzon D (2005) A rapid system for applying thermal stimuli during patch clamp and [Ca2+]i imaging experiments (abstract). J Physiol (Lond) (Proceedings) Bristol meeting July 2005
Rosenzweig M, Brennan KM, Tayler TD, Phelps PO, Patapoutian A, Garrity PA (2005) The Drosophila ortholog of vertebrate TRPA1 regulates thermotaxis. Genes Dev 19:419–424
Schäfer K, Braun H, Isenberg C (1986) Effect of menthol on cold receptor activity. Analysis of receptor processes. J Gen Physiol 88:757–776
Scroggs RS, Fox AP (1992) Calcium current variation between acutely isolated adult rat dorsal root ganglion neurons of different size. J Physiol (Lond) 445:639–658
Serra J, Campero M, Ochoa J, Bostock H (1999) Activity-dependent slowing of conduction differentiates functional subtypes of C fibres innervating human skin. J Physiol (Lond) 515:799–811
Simone DA, Kajander KC (1996) Excitation of rat cutaneous nociceptors by noxious cold. Neurosci Lett 213:53–56
Simone DA, Kajander KC (1997) Responses of cutaneous A-fiber nociceptors to noxious cold. J Neurophysiol 77:2049–2060
Smith MP, Beacham D, Ensor E, Koltzenburg M (2004) Cold-sensitive, menthol-insensitive neurons in the murine sympathetic nervous system. Neuroreport 15:1399–1403
Spray DC (1974) Characteristics, specificity, and efferent control of frog cutaneous cold receptors. J Physiol (Lond) 237:15–38
Spray DC (1974) Metabolic dependence of frog cold receptor sensitivity. Brain Res 72:354–359
Spray DC (1986) Cutaneous temperature receptors. Annu Rev Physiol 48:625–638
Story GM, Peier AM, Reeve AJ, Eid SR, Mosbacher J, Hricik TR, Earley TJ, Hergarden AC, Andersson DA, Hwang SW, McIntyre P, Jegla T, Bevan S, Patapoutian A (2003) ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell 112:819–829
Suto K, Gotoh H (1999) Calcium signaling in cold cells studied in cultured dorsal root ganglion neurons. Neuroscience 92:1131–1135
Thut PD, Wrigley D, Gold MS (2003) Cold transduction in rat trigeminal ganglia neurons in vitro. Neuroscience 119:1071–1083
Todorovic SM, Lingle CJ (1998) Pharmacological properties of T-type Ca2+ current in adult rat sensory neurons: effects of anticonvulsant and anesthetic agents. J Neurophysiol 79:240–252
Tracey WD, Wilson RI, Laurent G, Benzer S (2003) painless, a Drosophila gene essential for nociception. Cell 113:261–273
Viana F, de la Peña E, Belmonte C (2002) Specificity of cold thermotransduction is determined by differential ionic channel expression. Nat Neurosci 5:254–260
Viana F, de la Peña E, Mälkiä A, Cabedo H, Belmonte C (2004) TRPM8-dependent and -independent mechanisms in neuronal cold sensing (abstract). Program No. 599.7, 2004 Abstract Viewer/Itinerary Planner. Society for Neuroscience, Washington. http://sfn.scholarone.com/itin2004/index.html
Viswanath V, Story GM, Peier AM, Petrus MJ, Lee VM, Hwang SW, Patapoutian A, Jegla T (2003) Opposite thermosensor in fruitfly and mouse. Nature 423:822–823
Voets T, Droogmans G, Wissenbach U, Janssens A, Flockerzi V, Nilius B (2004) The principle of temperature-dependent gating in cold- and heat-sensitive TRP channels. Nature 430:748–754
Wang Z, van den Berg RJ, Ypey DL (1994) Resting membrane potentials and excitability at different regions of rat dorsal root ganglion neurons in culture. Neuroscience 60:245–254
Yarnitsky D, Ochoa JL (1990) Release of cold-induced burning pain by block of cold-specific afferent input. Brain 113:893–902
Zars T (2001) Two thermosensors in Drosophila have different behavioral functions. J Comp Physiol [A] 187:235–242
Zotterman Y (1978) How it started: a personal review. In: Kenshalo DR (ed) Sensory functions of the skin in humans. Plenum Press, New York, pp 5–22
Acknowledgements
Work in the author’s laboratory is supported by the Volkswagen Foundation, the Physiological Society and the Wellcome Trust. I am grateful to Alexandru Babes and Sally Lawson for comments on the manuscript, Felix Viana and Hans Braun for thought-provoking discussions, David Julius and David McKemy for rat TRPM8 used in the experiments shown in Fig. 4, and to Catriona Reid for help with preparing the figures.
Author information
Authors and Affiliations
Corresponding author
Note added in the proof
Note added in the proof
A very recent study (Rohacs et al., Nature Neurosci 8:626-634, 2005) has considerably strengthened the evidence for involvement of PIP2 in Ca2+-dependent adaptation and modulation of TRPM8, making it likely that PIP2 has a dominant role in these processes.
Rights and permissions
About this article
Cite this article
Reid, G. ThermoTRP channels and cold sensing: what are they really up to?. Pflugers Arch - Eur J Physiol 451, 250–263 (2005). https://doi.org/10.1007/s00424-005-1437-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-005-1437-z