Abstract
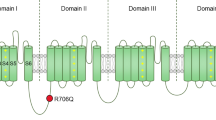
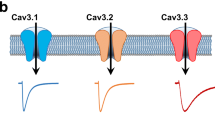
We report expression system-dependent effects of heterozygous mutations (P769L and A1059S) in the Cav3.2 CACNA1H gene identified in a pediatric patient with chronic pain and absence seizures. The mutations were introduced individually into recombinant channels and then analyzed by means of electrophysiology. When both mutants were co-expressed in tsA-201 cells, we observed a loss of channel function, with significantly smaller current densities across a wide range of voltages (−40 to +20 mV). In addition, when both mutant channels were co-expressed, the channels opened at a more depolarizing potential with a ~5-mV right shift in the half-activation potential, with no changes in half-inactivation potential and the rate of recovery from inactivation. Interestingly, when both mutants were co-expressed in the neuronal-derived CAD cells in a different extracellular milieu, the effect was remarkably different. Although not statistically significant (p < 0.07), current densities appeared augmented compared to wild-type channels and the difference in the half-activation potential was lost. This could be attributed to the replacement of extracellular sodium and potassium with tetraethylammonium chloride. Our results show that experimental conditions can be a confounding factor in the biophysical effects of T-type calcium channel mutations found in certain neurological disorders.




Similar content being viewed by others
References
Perez-Reyes E (2003) Molecular physiology of low-voltage-activated t-type calcium channels. Physiol Rev 83:117–161. doi:10.1152/physrev.00018.2002
Cain SM, Snutch TP (2013) T-type calcium channels in burst-firing, network synchrony, and epilepsy. Biochimica et Biophysica Acta (BBA) - Biomembranes 1828:1572-1578. doi:http://dx.doi.org/10.1016/j.bbamem.2012.07.028
Huguenard J, Prince D (1992) A novel T-type current underlies prolonged Ca (2+)-dependent burst firing in GABAergic neurons of rat thalamic reticular nucleus. The Journal of Neuroscience 12:3804–3817
Eckle VS, Shcheglovitov A, Vitko I, Dey D, Yap CC, Winckler B, Perez-Reyes E (2014) Mechanisms by which a CACNA1H mutation in epilepsy patients increases seizure susceptibility. J Physiol 592:795–809. doi:10.1113/jphysiol.2013.264176
Chen Y, Lu J, Pan H, Zhang Y, Wu H, Xu K, Liu X, Jiang Y, Bao X, Yao Z (2003) Association between genetic variation of CACNA1H and childhood absence epilepsy. Annals of neurology 54:239–243
Heron SE, Khosravani H, Varela D, Bladen C, Williams TC, Newman MR, Scheffer IE, Berkovic SF, Mulley JC, Zamponi GW (2007) Extended spectrum of idiopathic generalized epilepsies associated with CACNA1H functional variants. Ann Neurol 62:560–568. doi:10.1002/ana.21169
Khosravani H, Altier C, Simms B, Hamming KS, Snutch TP, Mezeyova J, McRory JE, Zamponi GW (2004) Gating effects of mutations in the Cav3.2 T-type calcium channel associated with childhood absence epilepsy. J Biol Chem 279:9681–9684. doi:10.1074/jbc.C400006200
Khosravani H, Bladen C, Parker DB, Snutch TP, McRory JE, Zamponi GW (2005) Effects of Cav3.2 channel mutations linked to idiopathic generalized epilepsy. Ann Neurol 57:745–749. doi:10.1002/ana.20458
Bourinet E, Altier C, Hildebrand ME, Trang T, Salter MW, Zamponi GW (2014) Calcium-permeable ion channels in pain signaling. Physiol Rev 94:81–140. doi:10.1152/physrev.00023.2013
Zamponi GW, Striessnig J, Koschak A, Dolphin AC (2015) The physiology, pathology, and pharmacology of voltage-gated calcium channels and their future therapeutic potential. Pharmacol Rev 67:821–870. doi:10.1124/pr.114.009654
Bourinet E, Alloui A, Monteil A, Barrere C, Couette B, Poirot O, Pages A, McRory J, Snutch TP, Eschalier A, Nargeot J (2005) Silencing of the Cav3.2 T-type calcium channel gene in sensory neurons demonstrates its major role in nociception. EMBO J 24:315–324. doi:10.1038/sj.emboj.7600515
Francois A, Kerckhove N, Meleine M, Alloui A, Barrere C, Gelot A, Uebele VN, Renger JJ, Eschalier A, Ardid D, Bourinet E (2013) State-dependent properties of a new T-type calcium channel blocker enhance Ca(V)3.2 selectivity and support analgesic effects. Pain 154:283–293. doi:10.1016/j.pain.2012.10.023
Garcia-Caballero A, Gadotti VM, Stemkowski P, Weiss N, Souza IA, Hodgkinson V, Bladen C, Chen L, Hamid J, Pizzoccaro A, Deage M, Francois A, Bourinet E, Zamponi GW (2014) The deubiquitinating enzyme USP5 modulates neuropathic and inflammatory pain by enhancing Cav3.2 channel activity. Neuron 83:1144–1158. doi:10.1016/j.neuron.2014.07.036
Berger ND, Gadotti VM, Petrov RR, Chapman K, Diaz P, Zamponi GW (2014) NMP-7 inhibits chronic inflammatory and neuropathic pain via block of Cav3.2 T-type calcium channels and activation of CB2 receptors. Mol Pain 10:77. doi:10.1186/1744-8069-10-77
Cao XH, Byun HS, Chen SR, Pan HL (2011) Diabetic neuropathy enhances voltage-activated Ca2+ channel activity and its control by M4 muscarinic receptors in primary sensory neurons. J Neurochem 119:594–603. doi:10.1111/j.1471-4159.2011.07456.x
Wen XJ, Li ZJ, Chen ZX, Fang ZY, Yang CX, Li H, Zeng YM (2006) Intrathecal administration of Cav3.2 and Cav3.3 antisense oligonucleotide reverses tactile allodynia and thermal hyperalgesia in rats following chronic compression of dorsal root of ganglion. Acta Pharmacol Sin 27:1547–1552. doi:10.1111/j.1745-7254.2006.00461.x
Weiss N, Black SA, Bladen C, Chen L, Zamponi GW (2013) Surface expression and function of Cav3.2 T-type calcium channels are controlled by asparagine-linked glycosylation. Pflugers Arch 465:1159–1170. doi:10.1007/s00424-013-1259-3
Qi Y, Wang JK, McMillian M, Chikaraishi DM (1997) Characterization of a CNS cell line, CAD, in which morphological differentiation is initiated by serum deprivation. The Journal of Neuroscience 17:1217–1225
Wang H, Oxford GS (2000) Voltage-dependent ion channels in CAD cells: a catecholaminergic neuronal line that exhibits inducible differentiation. Journal of Neurophysiology 84:2888–2895
Khosravani H, Zamponi GW (2006) Voltage-gated calcium channels and idiopathic generalized epilepsies. Physiol Rev 86:941–966. doi:10.1152/physrev.00002.2006
Waxman SG, Zamponi GW (2014) Regulating excitability of peripheral afferents: emerging ion channel targets. Nat Neurosci 17:153–163. doi:10.1038/nn.3602
Hans M, Luvisetto S, Williams ME, Spagnolo M, Urrutia A, Tottene A, Brust PF, Johnson EC, Harpold MM, Stauderman KA (1999) Functional consequences of mutations in the human α1A calcium channel subunit linked to familial hemiplegic migraine. The Journal of Neuroscience 19:1610–1619
Tottene A, Fellin T, Pagnutti S, Luvisetto S, Striessnig J, Fletcher C, Pietrobon D (2002) Familial hemiplegic migraine mutations increase Ca(2+) influx through single human CaV2.1 channels and decrease maximal CaV2.1 current density in neurons. Proc Natl Acad Sci U S A 99:13284–13289. doi:10.1073/pnas.192242399
Lewis TM, Harkness PC, Sivilotti LG, Colquhoun D, Millar NS (1997) The ion channel properties of a rat recombinant neuronal nicotinic receptor are dependent on the host cell type. The Journal of Physiology 505:299–306. doi:10.1111/j.1469-7793.1997.299bb.x
Cummins TR, Aglieco F, Renganathan M, Herzog RI, Dib-Hajj SD, Waxman SG (2001) Nav1. 3 sodium channels: rapid repriming and slow closed-state inactivation display quantitative differences after expression in a mammalian cell line and in spinal sensory neurons. The Journal of Neuroscience 21:5952–5961
Zamponi GW (2015) Targeting voltage-gated calcium channels in neurological and psychiatric diseases. Nat Rev Drug Discov. doi:10.1038/nrd.2015.5
Peloquin JB, Khosravani H, Barr W, Bladen C, Evans R, Mezeyova J, Parker D, Snutch TP, McRory JE, Zamponi GW (2006) Functional analysis of Ca3.2 T-type calcium channel mutations linked to childhood absence epilepsy. Epilepsia 47:655–658. doi:10.1111/j.1528-1167.2006.00482.x
Iftinca MC, Zamponi GW (2009) Regulation of neuronal T-type calcium channels. Trends Pharmacol Sci 30:32–40. doi:10.1016/j.tips.2008.10.004
Dibbens LM, Heron SE, Mulley JC (2007) A polygenic heterogeneity model for common epilepsies with complex genetics. Genes Brain Behav 6:593–597. doi:10.1111/j.1601-183X.2007.00333.x
Powell KL, Cain SM, Ng C, Sirdesai S, David LS, Kyi M, Garcia E, Tyson JR, Reid CA, Bahlo M, Foote SJ, Snutch TP, O'Brien TJ (2009) A Cav3.2 T-type calcium channel point mutation has splice-variant-specific effects on function and segregates with seizure expression in a polygenic rat model of absence epilepsy. J Neurosci 29:371–380. doi:10.1523/JNEUROSCI.5295-08.2009
Lennon P, Crotty M, Fenton JE (2015) Infectious mononucleosis. BMJ 350:h1825. doi:10.1136/bmj.h1825
Connelly KP, DeWitt LD (1994) Neurologic complications of infectious mononucleosis. Pediatric Neurology 10:181–184. doi:10.1016/0887-8994(94)90021-3
Silverstein A, Steinberg G, Nathanson M (1972) Nervous system involvement in infectious mononucleosis: the heralding and/or major manifestation. Archives of Neurology 26:353–358
Acknowledgments
We thank the patient and her family for their cooperation with our research team. We are grateful to Lina Chen for her technical support. We thank Dr. Francois Bernier for the ExAC database search.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Financial support
This work was supported by a grant from the Canadian Institutes of Health Research to GWZ. MAG is supported by a Consejo Nacional de Ciencia y Tecnologia (Mexico) postdoctoral fellowship, and MMW was supported by an Alberta Innovates—Health Solutions summer studentship.
Conflict of interest
The authors declare that they have no conflict of interest.
Consent
Informed consent was obtained from the mother for the publication of this article.
Additional information
Ivana A. Souza and Maria A. Gandini contributed equally to this work.
Rights and permissions
About this article
Cite this article
Souza, I.A., Gandini, M.A., Wan, M.M. et al. Two heterozygous Cav3.2 channel mutations in a pediatric chronic pain patient: recording condition-dependent biophysical effects. Pflugers Arch - Eur J Physiol 468, 635–642 (2016). https://doi.org/10.1007/s00424-015-1776-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-015-1776-3