Abstract
Main conclusion
Root antioxidant defense, restricted root-to-shoot Cu translocation, altered nutrient partition, and leaf gas exchange adjustments occurred as tolerance mechanisms of soybean plants to increasing soil Cu levels.
Abstract
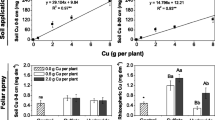
The intensive application of copper (Cu) fungicides has been related to the accumulation of this metal in agricultural soils. This study aimed to evaluate the effects of increasing soil Cu levels on soybean (Glycine max) plants. Soybean was cultivated under greenhouse conditions in soils containing different Cu concentrations (11.2, 52.3, 79.4, 133.5, 164.0, 205.1, or 243.8 mg kg−1), and biochemical and morphophysiological plant responses were analyzed through linear and nonlinear regression models. Although Cu concentrations around 50 mg kg−1 promoted some positive effects on the initial development of soybean plants (e.g., increased root length and dry weight), these Cu concentrations also induced root oxidative stress and activated defense mechanisms (such as the induction of antioxidant response, N and S accumulation in the roots). At higher concentrations, Cu led to growth inhibition (mainly of the root), nutritional imbalance, and damage to the photosynthetic apparatus of soybean plants, resulting in decreased CO2 assimilation and stomatal conductance. In contrast, low translocation of Cu to the leaves, conservative water use, and increased carboxylation efficiency contributed to the partial mitigation of Cu-induced stress. These responses allowed soybean plants treated with Cu levels in the soil as high as 90 mg kg−1 to maintain growth parameters higher than or similar to those of plants in the non-contaminated soil. These data provide a warning for the potentially deleterious consequences of the increasing use of Cu-based fungicides. However, it is necessary to verify how the responses to Cu contamination are affected by different types of soil and soybean cultivars.








Similar content being viewed by others
Abbreviations
- A :
-
Net photosynthetic rate
- APX:
-
Ascorbate peroxidase
- CAT:
-
Catalase
- F 0 :
-
Basal fluorescence
- F v/F 0 :
-
Potential activity of photosystem II
- k :
-
Instantaneous carboxylation efficiency
- POD:
-
Peroxidase
- rETR:
-
Relative rate of linear electron transport of photosystem II
- SOD:
-
Superoxide dismutase
- WUE:
-
Water use efficiency
References
Adrees M, Ali S, Rizwan M, Ibrahim M, Abbas F, Farid M, Zia-ur-Rehman M, Irshad MK, Bharwana SA (2015) The effect of excess copper on growth and physiology of important food crops: a review. Environ Sci Pollut Res 22:8148–8162. https://doi.org/10.1007/s11356-015-4496-5
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:114–121. https://doi.org/10.1016/s0076-6879(84)05016-3
Alexieva V, Sergiev I, Mapelli S, Karanov E (2001) The effect of drought and ultraviolet radiation on growth and stress markers in pea and wheat. Plant Cell Environ 24:1337–1344. https://doi.org/10.1046/j.1365-3040.2001.00778.x
Ambrosini VG, Rosa DJ, Basso A, Borghezan M, Pescador R, Miotto A, Melo GWB, Soares CRFS, Comin JJ, Brunetto G (2016) Liming as an ameliorator of copper toxicity in black oat (Avena strigosa Schreb.). J Plant Nutr 40:404–416. https://doi.org/10.1080/01904167.2016.1240203
Amlal F, Drissi S, Makroum K, Dhassi K, Er-rezza H, Houssa AA (2020) Influence of soil characteristics and leaching rate on copper migration: column test. Heliyon 6:e03375. https://doi.org/10.1016/j.heliyon.2020.e03375
Anderson MD, Prasad TK, Stewart CR (1995) Changes in isozyme profiles of catalase, peroxidase and gluthione reductase during acclimation to chilling in mesocotylus of maize seedlings. Plant Physiol 109:1247–1257. https://doi.org/10.1104/pp.109.4.1247
Archontoulis SV, Miguez FE (2015) Nonlinear regression models and applications in agricultural research. Agron J 107:786–798. https://doi.org/10.2134/agronj2012.0506
Baker NR (2008) Chlorophyll fluorescence: a probe of photosynthesis in vivo. Annu Rev Plant Biol 59:89–113. https://doi.org/10.1146/annurev.arplant.59.032607.092759
Balestrasse KB, Benavides MP, Gallego SM, Tomaro ML (2003) Effect of cadmium stress on nitrogen metabolism in nodules and roots of soybean plants. Funct Plant Biol 30:57–64. https://doi.org/10.1071/fp02074
Bayer (2020) Soybean growth and development. Dekalb, Asgrow and Deltapine com. https://www.dekalbasgrowdeltapine.com/en-us/agronomy/soybean-growth-stages.html. Accessed 25 Nov 2020
Boerema A, Peeters A, Swolfs S, Vandevenne F, Jacobs S, Staes J, Meire P (2016) Soybean trade: balancing environmental and socio-economic impacts of an intercontinental market. PLoS ONE 11:e0155222. https://doi.org/10.1371/journal.pone.0155222
Boveris A, Cadenas E, Chance B (1980) Low-level chemi-luminescence of the lipoxygenase reaction. Photobiochem Photobiophys 1:175–182
Brunetto G, Melo GWB, Terzano R, Buono DD, Astolfi S, Tomasi N, Pii Y, Mimmo T, Cesco S (2016) Copper accumulation in vineyard soils: rhizosphere processes and agronomic practices to limit its toxicity. Chemosphere 162:293–307. https://doi.org/10.1016/j.chemosphere.2016.07.104
Brunetto G, Ferreira PAA, Melo GW, Ceretta CA, Toselli M (2017) Heavy metals in vineyards and orchard soils. Rev Bras Frutic 39:e-263. https://doi.org/10.1590/0100-29452017263
Cambrollé J, García JL, Figueroa ME, Cantos M (2015) Evaluating wild grapevine tolerance to copper toxicity. Chemosphere 120:171–178. https://doi.org/10.1016/j.chemosphere.2014.06.044
Campos DVB, Teixeira PC (2017) Microelementos. In: Teixeira PC, Donagema GK, Fontana A, Teixeira WGM (eds) Manual de métodos de análise de solo, 3rd edn. Embrapa, Brasília, pp 328–333
Chiou WY, Hsu FC (2019) Copper toxicity and prediction models of copper content in leafy vegetables. Sustainability 11:6215. https://doi.org/10.3390/su11226215
Couder E, Mattielli N, Drouet T, Smolders E, Delvaux B, Iserentant A, Meeus C, Maerschalk C, Opfergelt S, Houben D (2015) Transpiration flow controls Zn transport in Brassica napus and Lolium multiflorum under toxic levels as evidenced from isotopic fractionation. C R Geosci 347:386–396. https://doi.org/10.1016/j.crte.2015.05.004
Embrapa (2020) Soluções tecnológicas. Soja-BRS 257. https://www.embrapa.br/busca-de-solucoes-tecnologicas/-/produto-servico/401/soja---brs-257. Accessed 25 Nov 2020
Fan J, He Z, Ma LQ, Stoffella PJ (2011) Accumulation and availability of copper in citrus grove soils as affected by fungicide application. J Soil Sediment 11:639–648. https://doi.org/10.1007/s11368-011-0349-0
Finger-Teixeira A, Ferrarese MLL, Soares AR, Silva D, Ferrarese-Filho O (2010) Cadmium-induced lignification restricts soybean root growth. Ecotoxicol Environ Safe 73:1959–1964. https://doi.org/10.1016/j.ecoenv.2010.08.021
Fu L, Chen C, Wang B, Zhou X, Li S, Guo P, Shen Z, Wang G, Chen Y (2015) Differences in copper absorption and accumulation between copper-exclusion and copper-enrichment plants: a comparison of structure and physiological responses. PLoS ONE 10:e0133424. https://doi.org/10.1371/journal.pone.0133424
Giannopolitis CN, Ries SK (1977) Superoxide dismutases. I. occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.1104/pp.59.2.309
Gomes DG, da Cunha LS, Oliveira HC (2020) Dose-dependent dual effect of soil copper on the initial development of Glycine max (L.) Merr. cv. BRS 257 seedlings. Bull Environ Contam Tox 105:553–558. https://doi.org/10.1007/s00128-020-02985-9
Husak V (2015) Copper and copper-containing pesticides: metabolism, toxicity and oxidative stress. JPNU 2:38–50. https://doi.org/10.15330/jpnu.2.1.38-50
Juliatti FC, Azevedo LAS, Juliatti FC (2017) Strategies of chemical protection for controlling soybean rust. In: Kasai M (ed) Soybean: the basis of yield, biomass and productivity. IntechOpen, London, pp 35–62
Kolbert Z, Petö A, Lehotai N, Feigl G, Erdei L (2012) Long-term copper (Cu2+) exposure impacts on auxin, nitric oxide (NO) metabolism and morphology of Arabidopsis thaliana L. Plant Growth Regul 68:151–159. https://doi.org/10.1007/s10725-012-9701-7
Kulikova AL, Kuznetsova NA, Burmistrova NA (2015) Change in growth and physiological parameters in soybean seedlings in response to toxic action of copper. Russ J Plant Physiol 62:488–498. https://doi.org/10.1134/S1021443715040111
Leng X, Mu Q, Wang X, Li X, Zhu X, Shangguan L, Fang J (2015) Transporters, chaperones, and P-type ATPases controlling grapevine copper homeostasis. Funct Integr Genomics 15:673–684. https://doi.org/10.1007/s10142-015-0444-1
Lequeux H, Hermans C, Lutts S, Verbruggen N (2010) Response to copper excess in Arabidopsis thaliana: impact on the root system architecture, hormone distribution, lignin accumulation and mineral profile. Plant Physiol Biochem 48:673–682. https://doi.org/10.1016/j.plaphy.2010.05.005
Lichtenthaler HK, Buschmann C (2001) Chlorophylls and carotenoids: measurement and characterization by UV-VIS spectroscopy. Curr Protocols Food Anal Chem 1:F4.3.1-F.4.3.8. https://doi.org/10.1002/0471142913.faf0403s01
Liu Q, Luo L, Zheng L (2018) Lignins: biosynthesis and biological functions in plants. Int J Mol Sci 19:335. https://doi.org/10.3390/ijms19020335
Malavolta E, Vitti GC, Oliveira AS (1997) Avaliação do estado nutricional das plantas: princípios e aplicações, 2nd edn. Potafos, Piracicaba
Małkowski E, Sitko K, Zieleźnik-Rusinowska P, Gieroń Ż, Szopiński M (2019) Heavy metal toxicity: physiological implications of metal toxicity in plants. In: Sablok G (ed) Plant metallomics and functional omics. Springer, Cham, pp 253–301
Marques DM, Júnior VV, Silva AB, Mantovani JR, Magalhães PC, Souza TC (2018) Copper toxicity on photosynthetic responses and root morphology of Hymenaea courbaril L (Caesalpinioideae). Water Air Soil Pollut 229:138. https://doi.org/10.1007/s11270-018-3769-2
Moreira A, Moraes LAC (2019) Soybean response to copper applied to two soild with different levels of organic matter and clay. J Plant Nut 42:2247–2258. https://doi.org/10.1080/01904167.2019.1655039
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Napoli M, Cecchi S, Grassi C, Baldi A, Zanchi CA, Orlandini S (2019) Phytoextraction of copper from a contaminated soil using arable and vegetable crops. Chemosphere 219:122–129. https://doi.org/10.1016/j.chemosphere.2018.12.017
Parveen A, Saleem MH, Kamran M, Haider MZ, Chen J-T, Malik Z, Rana MS, Hassan A, Hu G, Javed MT, Azeem M (2020) Effect of citric acid on growth, ecophysiology, chloroplast ultrastructure, and phytoremediation potential of jute (Corchorus capsularis L.) seedlings exposed to copper stress. Biomolecules 10:592. https://doi.org/10.3390/biom10040592
Peixoto HPP, Cambraia J, San’tana R, Mosquim PR, Moreira AM (1999) Aluminium effects on lipid peroxidation and the activities of enzymes of oxidative metabolism in sorghum. Rev Bras Fisiol Veg 11:137–143
Pérez-Chaca MV, Vigliocco A, Reinoso H, Molina A, Abdala G, Zirulnik F, Pedranzani H (2014) Effects of cádmium stress on growth, anatomy and hormone contents in Glycine max (L.) Merr. Acta Physiol Plant 36:2815–2826. https://doi.org/10.1007/s11738-014-1656-z
R Development Core Team (2020) R: A language and environment for statistical computing. http://www.R-project.org/. Accessed 13 Aug 2020
Rascio N, Navari-Izzo F (2011) Heavy metal hyperaccumulating plants: How and why do they do it? And what makes them so interesting? Plant Sci 180:169–181. https://doi.org/10.1016/j.plantsci.2010.08.016
Rucinska-Sobkowiak R (2016) Water relations in plants subjected to heavy metal stresses. Acta Physiol Plant 38:257. https://doi.org/10.1007/s11738-016-2277-5
Saleem MH, Fahad S, Khan SU, Din M, Ullah A, Sabagh AE, Hossain A, Llanes A, Liu L (2019) Copper-induced oxidative stress, initiation of antioxidants and phytoremediation potential of flax (Linum usitatissimum L.) seedlings grown under the mixing of two different soils of China. Environ Sci Pollut Res 27:5211–5221. https://doi.org/10.1007/s11356-019-07264-7
Saleem MH, Ali S, Rehman M, Rana MS, Rizwan M, Kamran M, Imran M, Riaz M, Soliman MH, Elkelish A, Liu L (2020a) Influence of phosphorus on copper phytoextraction via modulating cellular organelles in two jute (Corchorus capsularis L.) varieties grown in a copper mining soil of Hubei Province, China. Chemosphere 248:126032. https://doi.org/10.1016/j.chemosphere.2020.126032
Saleem MH, Fahad S, Khan SU, Ahmar S, Ullah Khan MH, Rehman M, Maqbool Z, Liu L (2020b) Morpho-physiological traits, gaseous exchange attributes, and phytoremediation potential of jute (Corchorus capsularis L.) grown in different concentrations of copper-contaminated soil. Ecotoxicol Environ Saf 189:109915. https://doi.org/10.1016/j.ecoenv.2019.109915
Saleem MH, Kamran M, Zhou Y, Parveen A, Rehman M, Ahmar S, Malik Z, Mustafa A, Ahmad Anjum RM, Wang B, Liu L (2020c) Appraising growth, oxidative stress and copper phytoextraction potential of flax (Linum usitatissimum L.) grown in soil differentially spiked with copper. J Environ Manage 257:109994. https://doi.org/10.1016/j.jenvman.2019.109994
Schabenberger O, Pierce FJ (2002) Contemporary statistical models for the plant and soil Sciences, 1st ed. CRC, Boca Raton
Sharma R, Bhardwaj R, Handa N, Gautam V, Kohli SK, Bali S, Kaur P, Thukral AK, Arora S, Ohri P, Vig AP (2015) Responses of phytochelatins and metallothioneins in alleviation of heavy metal stress in plants: an overview. In: Ahmad P (ed) Plant metal interaction: emerging remediation techniques. Elsevier, Amsterdam, pp 263–283
Singh S, Parihar P, Singh R, Singh VP, Prasad SM (2016) Heavy metal tolerance in plants: role of transcriptomics, proteomics, metabolomics, and ionomics. Front Plant Sci 6:1143. https://doi.org/10.3389/fpls.2015.01143
Srivastava S, Mishra S, Tripathi RD, Dwivedi S, Gupta DK (2006) Copper-induced stress and responses of antioxidants and phytochelatins in Hydrilla verticillata (L.) Royle. Aquat Toxicol 80:405–415. https://doi.org/10.1016/j.aquatox.2006.10.006
Srivastava R, Khan R, Manzoor N, Uzzafar M (2011) Responses of cadmium exposures on growth, physio-biochemical characteristics and the antioxidative defence system of soybean (Glycine max L.). J Phytol 3:20–25
Štork F, Bačkor M, Klejdus B, Hedbavny J, Kováčik J (2013) Changes of metal-induced toxicity by H2O2/NO modulators in Scenedesmus quadricauda (Chlorophyceae). Environ Sci Pollut Res 20:5502–5511. https://doi.org/10.1007/s11356-013-15410
Sun Q, Li T, Alva AK, Li YC (2019) Mobility and fractionation of copper in sandy soils. Environ Pollut Bioavail 31:18–23. https://doi.org/10.1080/09542299.2018.1558114
Tiwari S, Sarangi BK (2015) Arsenic and chromium-induced oxidative stress in metal accumulator and non-accumulator plants and detoxification mechanisms. In: Gupta DK, Palma JM, Corpas FJ (eds) Reactive oxygen species and oxidative damage in plants under stress. Springer, Switzerland, pp 165–189
Toselli M, Schiatti P, Ara D, Bertacchini A, Quartieri M (2009) The accumulation of copper in soils of the Italian region Emilia-Romagna. Plant Soil Environ 55:74–79. https://doi.org/10.17221/317-PSE
United States Department of Agriculture, USDA (2020) Oilseeds: World markets and trade. https://www.fas.usda.gov/data/oilseeds-world-markets-and-trade. Accessed 13 Aug 2020
Verma S, Dubey RS (2003) Lead toxicity induces lipid peroxidation and alters the activities of antioxidant enzymes in growing rice plants. Plant Sci 164:645–655. https://doi.org/10.1016/S0168-9452(03)00022-0
Yruela I (2009) Copper in plants: acquisition, transport and interactions. Funct Plant Biol 36:409–430. https://doi.org/10.1071/FP08288
Yruela I (2013) Transition metals in plant photosynthesis. Metallomics 5:1090–1109. https://doi.org/10.1039/c3mt00086a
Zhou X, Wang S, Liu Y, Huang G, Yao S, Hu H (2020) Coupling phytoremediation efficiency and detoxification to assess the role of P in the Cu tolerant Ricinus communis L. Chemosphere 247:125965. https://doi.org/10.1016/j.chemosphere.2020.125965
Acknowledgements
This study was financed by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001 (DGG, PJL, TVD) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (HCO, fellowship 306583/2017-8). The authors also thank the Laboratory of Soils (João Machado dos Santos, Marcio Alves de Lima Praxedes and Dr. Marla Alessandra de Araújo) and Laboratory of Animal Ecophysiology (Dr. Claudia Bueno dos Reis Martinez and Dr. Wagner Risso) from Universidade Estadual de Londrina for access to equipment and assistance in nutritional and biochemical analyses.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gomes, D.G., Lopes-Oliveira, P.J., Debiasi, T.V. et al. Regression models to stratify the copper toxicity responses and tolerance mechanisms of Glycine max (L.) Merr. plants. Planta 253, 43 (2021). https://doi.org/10.1007/s00425-021-03573-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-021-03573-9