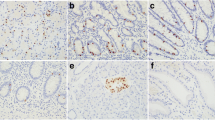
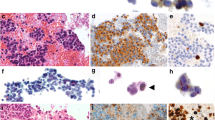
Abstract
Primary gastrointestinal neuroendocrine carcinoma (GI-NEC) cannot be distinguished morphologically from pulmonary neuroendocrine carcinoma (P-NEC). This can present a significant diagnostic challenge in cases where site of origin cannot be readily determined. To identify immunohistochemical (IHC) markers that can be used to reliably distinguish between GI-NECs and P-NECs, we constructed 3-mm tissue microarrays, one containing 13 GI-NECs and one containing 20 P-NECs. IHC was performed on both microarrays using 21 stains: AE1/AE3, CK7, CK20, synaptophysin, chromogranin, CD56, INSM1, SSTR2A, CDX2, SATB2, TTF1, Napsin A, PR, GATA3, PAX8, ISL1, beta-catenin, AFP, SMAD4, Rb, and p53. For GI-NEC, the most strongly expressed marker was synaptophysin (mean H-score 248), while AE1/AE3 was the most strongly expressed in P-NEC (mean H-score 230), which was stronger than in GI-NEC (p = 0.011). Other markers that were stronger overall in P-NEC than in GI-NEC included CK7 (p < 0.0001) and TTF1 (p < 0.0001). Markers that were stronger overall in GI-NEC than in P-NEC included SSTR2A (p = 0.0021), SATB2 (p = 0.018), CDX2 (p = 0.019), and beta-catenin (nuclear; p = 0.029). SMAD4, Rb, and p53 showed similar rates of abnormal protein expression. Based on these results, a stepwise algorithmic approach utilizing CK7, TTF1, beta-catenin, CDX2, and SSTR2A had a 91% overall accuracy in distinguishing these GI-NEC from P-NEC. This was tested on a second cohort of 10 metastatic GI-NEC and 10 metastatic P-NEC, with an accuracy in this cohort of 85% and an overall accuracy of 89% for the 53 cases tested. Our algorithm reasonably discriminates GI-NEC from P-NEC using currently available IHC stains.



Similar content being viewed by others
References
Dasari A, Shen C, Halperin D, Zhao B, Zhou S, Xu Y, Shih T, Yao JC (2017) Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol 3:1335–1342
Milione M, Maisonneuve P, Spada F, Pellegrinelli A, Spaggiari P, Albarello L, Pisa E, Barberis M, Vanoli A, Buzzoni R, Pusceddu S, Concas L, Sessa F, Solcia E, Capella C, Fazio N, la Rosa S (2017) The clinicopathologic heterogeneity of grade 3 gastroenteropancreatic neuroendocrine neoplasms: morphological differentiation and proliferation identify different prognostic categories. Neuroendocrinology 104:85–93
Heetfeld M, Chougnet CN, Olsen IH, Rinke A, Borbath I, Crespo G, Barriuso J, Pavel M, O'Toole D, Walter T, other Knowledge Network members (2015) Characteristics and treatment of patients with G3 gastroenteropancreatic neuroendocrine neoplasms. Endocr Relat Cancer 22:657–664
Basturk O, Saka B, Balci S et al (2015) Substaging of lymph node status in resected pancreatic ductal adenocarcinoma has strong prognostic correlations: proposal for a revised N classification for TNM staging. Ann Surg Oncol 22(Suppl 3):S1187–S1195
Walter T, Tougeron D, Baudin E, le Malicot K, Lecomte T, Malka D, Hentic O, Manfredi S, Bonnet I, Guimbaud R, Coriat R, Lepère C, Desauw C, Thirot-Bidault A, Dahan L, Roquin G, Aparicio T, Legoux JL, Lombard-Bohas C, Scoazec JY, Lepage C, Cadiot G, Stephanie L, Borbath I, Castex, Petorin C, Terrebonne E, Bouhier-Leporrier K, Suc E, Hautefeuille V, Bourgeois V, Cany L, Dewaele F, Niccoli P, Seitz JF, Lecaille C, Rebischung C, Rossi V, Baconnier M, Dubreuil O, Ferec M, Deplanque G, Geslin G, Wanicki Caron I, Lavau Denes S, Bedenne L, Ligeza C, Maringe E, Ran-Royo AL, Guigay J, Rougier P (2017) Poorly differentiated gastro-entero-pancreatic neuroendocrine carcinomas: are they really heterogeneous? Insights from the FFCD-GTE national cohort. Eur J Cancer 79:158–165
Sorbye H, Welin S, Langer SW, Vestermark LW, Holt N, Osterlund P, Dueland S, Hofsli E, Guren MG, Ohrling K, Birkemeyer E, Thiis-Evensen E, Biagini M, Gronbaek H, Soveri LM, Olsen IH, Federspiel B, Assmus J, Janson ET, Knigge U (2013) Predictive and prognostic factors for treatment and survival in 305 patients with advanced gastrointestinal neuroendocrine carcinoma (WHO G3): the NORDIC NEC study. Ann Oncol 24:152–160
Yamaguchi T, Machida N, Morizane C, Kasuga A, Takahashi H, Sudo K, Nishina T, Tobimatsu K, Ishido K, Furuse J, Boku N, Okusaka T (2014) Multicenter retrospective analysis of systemic chemotherapy for advanced neuroendocrine carcinoma of the digestive system. Cancer Sci 105:1176–1181
Howe HL, Wingo PA, Thun MJ, Ries LAG, Rosenberg HM, Feigal EG, Edwards BK (2001) Annual report to the nation on the status of cancer (1973 through 1998), featuring cancers with recent increasing trends. J Natl Cancer Inst 93:824–842
Mulshine JL, Treston AM, Brown PH, Birrer MJ, Shaw GL (1993) Initiators and promoters of lung cancer. Chest 103:4s–11s
Usuda K, Saito Y, Sagawa M, Sato M, Kanma K, Takahashi S, Endo C, Chen Y, Sakurada A, Fujimura S (1994) Tumor doubling time and prognostic assessment of patients with primary lung cancer. Cancer 74:2239–2244
Tamura T (2001) New state of the art in small-cell lung cancer. Oncology (Williston Park) 15:8–10
Zochbauer-Muller S, Pirker R, Huber H (1999) Treatment of small cell lung cancer patients. Ann Oncol 10:83–91
Adjei AA, Marks RS, Bonner JA (1999) Current guidelines for the management of small cell lung cancer. Mayo Clin Proc 74:809–816
Nicholson SA, Beasley MB, Brambilla E, Hasleton PS, Colby TV, Sheppard MN, Falk R, Travis WD (2002) Small cell lung carcinoma (SCLC): a clinicopathologic study of 100 cases with surgical specimens. Am J Surg Pathol 26:1184–1197
Fasano M, Della Corte CM, Papaccio F, Ciardiello F, Morgillo F (2015) Pulmonary large-cell neuroendocrine carcinoma: from epidemiology to therapy. J Thorac Oncol 10:1133–1141
Kinslow CJ, May MS, Saqi A, Shu CA, Chaudhary KR, Wang TJC, Cheng SK (2020) Large-Cell Neuroendocrine Carcinoma of the Lung: A Population-Based Study. Clin Lung Cancer 21:e99–e113
Cao L, Li ZW, Wang M, Zhang TT, Bao B, Liu YP (2019) Clinicopathological characteristics, treatment and survival of pulmonary large cell neuroendocrine carcinoma: a SEER population-based study. PeerJ 7:e6539
Dasari A, Mehta K, Byers LA, Sorbye H, Yao JC (2018) Comparative study of lung and extrapulmonary poorly differentiated neuroendocrine carcinomas: a SEER database analysis of 162,983 cases. Cancer 124:807–815
Bellizzi AM (2020) Immunohistochemistry in the diagnosis and classification of neuroendocrine neoplasms: what can brown do for you? Hum Pathol 96:8–33
Bellizzi AM (2013) Assigning site of origin in metastatic neuroendocrine neoplasms: a clinically significant application of diagnostic immunohistochemistry. Adv Anat Pathol 20:285–314
Bellizzi AM (2020) SATB2 in neuroendocrine neoplasms: strong expression is restricted to well-differentiated tumours of lower gastrointestinal tract origin and is most frequent in Merkel cell carcinoma among poorly differentiated carcinomas. Histopathology 76:251–264
Bellizzi AM (2020) An algorithmic immunohistochemical approach to define tumor type and assign site of origin. Adv Anat Pathol 27:114–163
Schatoff EM, Leach BI, Dow LE (2017) Wnt signaling and colorectal cancer. Curr Colorectal Cancer Rep 13:101–110
Hiroshima K, Iyoda A, Shida T, Shibuya K, Iizasa T, Kishi H, Tanizawa T, Fujisawa T, Nakatani Y (2006) Distinction of pulmonary large cell neuroendocrine carcinoma from small cell lung carcinoma: a morphological, immunohistochemical, and molecular analysis. Mod Pathol 19:1358–1368
Kargi A, Gurel D, Tuna B (2007) The diagnostic value of TTF-1, CK 5/6, and p63 immunostaining in classification of lung carcinomas. Appl Immunohistochem Mol Morphol 15:415–420
Hu J, Han B, Huang J (2020) Morphologic spectrum of neuroendocrine tumors of the prostate: an updated review. Arch Pathol Lab Med 144:320–325
McCluggage WG, Kennedy K, Busam KJ (2010) An immunohistochemical study of cervical neuroendocrine carcinomas: neoplasms that are commonly TTF1 positive and which may express CK20 and P63. Am J Surg Pathol 34:525–532
Sturm N, Rossi G, Lantuejoul S, Papotti M, Frachon S, Claraz C, Brichon PY, Brambilla C, Brambilla E (2002) Expression of thyroid transcription factor-1 in the spectrum of neuroendocrine cell lung proliferations with special interest in carcinoids. Hum Pathol 33:175–182
La Rosa S, Chiaravalli AM, Placidi C, Papanikolaou N, Cerati M, Capella C (2010) TTF1 expression in normal lung neuroendocrine cells and related tumors: immunohistochemical study comparing two different monoclonal antibodies. Virchows Arch 457:497–507
Masai K, Tsuta K, Kawago M, Tatsumori T, Kinno T, Taniyama T, Yoshida A, Asamura H, Tsuda H (2013) Expression of squamous cell carcinoma markers and adenocarcinoma markers in primary pulmonary neuroendocrine carcinomas. Appl Immunohistochem Mol Morphol 21:292–297
Rekhtman N, Pietanza CM, Sabari J, Montecalvo J, Wang H, Habeeb O, Kadota K, Adusumilli P, Rudin CM, Ladanyi M, Travis WD, Joubert P (2018) Pulmonary large cell neuroendocrine carcinoma with adenocarcinoma-like features: napsin A expression and genomic alterations. Mod Pathol 31:111–121
Zhang C, Schmidt LA, Hatanaka K, Thomas D, Lagstein A, Myers JL (2014) Evaluation of napsin A, TTF-1, p63, p40, and CK5/6 immunohistochemical stains in pulmonary neuroendocrine tumors. Am J Clin Pathol 142:320–324
Barbareschi M, Roldo C, Zamboni G, Capelli P, Cavazza A, Macri E, Cangi MG, Chilosi M, Doglioni C (2004) CDX-2 homeobox gene product expression in neuroendocrine tumors: its role as a marker of intestinal neuroendocrine tumors. Am J Surg Pathol 28:1169–1176
La Rosa S, Rigoli E, Uccella S, Chiaravalli AM, Capella C (2004) CDX2 as a marker of intestinal EC-cells and related well-differentiated endocrine tumors. Virchows Arch 445:248–254
Inzani F, Santoro A, Angelico G, Feraco A, Spadola S, Arciuolo D, Valente M, Carlino A, Piermattei A, Scaglione G, Scambia G, Rindi G, Zannoni GF (2020) Neuroendocrine carcinoma of the uterine cervix: a clinicopathologic and immunohistochemical study with focus on novel markers (Sst2-Sst5). Cancers 12:1211
Lee H, Fu Z, Koo BH, Sheehan CE, Young GQ, Lin J, Patil DT, Yang Z (2018) The expression of TTF1, CDX2 and ISL1 in 74 poorly differentiated neuroendocrine carcinomas. Ann Diagn Pathol 37:30–34
Cheuk W, Kwan MY, Suster S, Chan JK (2001) Immunostaining for thyroid transcription factor 1 and cytokeratin 20 aids the distinction of small cell carcinoma from Merkel cell carcinoma, but not pulmonary from extrapulmonary small cell carcinomas. Arch Pathol Lab Med 125:228–231
Magnusson K, de Wit M, Brennan DJ, Johnson LB, McGee SF, Lundberg E, Naicker K, Klinger R, Kampf C, Asplund A, Wester K, Gry M, Bjartell A, Gallagher WM, Rexhepaj E, Kilpinen S, Kallioniemi OP, Belt E, Goos J, Meijer G, Birgisson H, Glimelius B, Borrebaeck CAK, Navani S, Uhlén M, O'Connor DP, Jirström K, Pontén F (2011) SATB2 in combination with cytokeratin 20 identifies over 95% of all colorectal carcinomas. Am J Surg Pathol 35:937–948
Dragomir A, de Wit M, Johansson C, Uhlen M, Ponten F (2014) The role of SATB2 as a diagnostic marker for tumors of colorectal origin: results of a pathology-based clinical prospective study. Am J Clin Pathol 141:630–638
Li Z, Yuan J, Wei L, Zhou L, Mei K, Yue J, Gao H, Zhang M, Jia L, Kang Q, Huang X, Cao D (2015) SATB2 is a sensitive marker for lower gastrointestinal well-differentiated neuroendocrine tumors. Int J Clin Exp Pathol 8:7072–7082
Agaimy A, Erlenbach-Wunsch K, Konukiewitz B et al (2013) ISL1 expression is not restricted to pancreatic well-differentiated neuroendocrine neoplasms, but is also commonly found in well and poorly differentiated neuroendocrine neoplasms of extrapancreatic origin. Mod Pathol 26:995–1003
Nassar H, Albores-Saavedra J, Klimstra DS (2005) High-grade neuroendocrine carcinoma of the ampulla of vater: a clinicopathologic and immunohistochemical analysis of 14 cases. Am J Surg Pathol 29:588–594
Ordonez NG (2013) Broad-spectrum immunohistochemical epithelial markers: a review. Hum Pathol 44:1195–1215
Charoenpitakchai M, Liu E, Zhao Z, Koyama T, Huh WJ, Berlin J, Hande K, Walker R, Shi C (2017) In liver metastases from small intestinal neuroendocrine tumors, SSTR2A expression is heterogeneous. Virchows Arch 470:545–552
Konukiewitz B, Schlitter AM, Jesinghaus M, Pfister D, Steiger K, Segler A, Agaimy A, Sipos B, Zamboni G, Weichert W, Esposito I, Pfarr N, Klöppel G (2017) Somatostatin receptor expression related to TP53 and RB1 alterations in pancreatic and extrapancreatic neuroendocrine neoplasms with a Ki67-index above 20. Mod Pathol 30:587–598
Righi L, Volante M, Tavaglione V, Billè A, Daniele L, Angusti T, Inzani F, Pelosi G, Rindi G, Papotti M (2010) Somatostatin receptor tissue distribution in lung neuroendocrine tumours: a clinicopathologic and immunohistochemical study of 218 ‘clinically aggressive’ cases. Ann Oncol 21:548–555
Tsuta K, Wistuba II, Moran CA (2012) Differential expression of somatostatin receptors 1–5 in neuroendocrine carcinoma of the lung. Pathol Res Pract 208:470–474
White BD, Chien AJ, Dawson DW (2012) Dysregulation of Wnt/β-catenin signaling in gastrointestinal cancers. Gastroenterology 142:219–232
Vijayvergia N, Boland PM, Handorf E, Gustafson KS, Gong Y, Cooper HS, Sheriff F, Astsaturov I, Cohen SJ, Engstrom PF (2016) Molecular profiling of neuroendocrine malignancies to identify prognostic and therapeutic markers: a Fox Chase Cancer Center Pilot Study. Br J Cancer 115:564–570
Rekhtman N, Pietanza MC, Hellmann MD, Naidoo J, Arora A, Won H, Halpenny DF, Wang H, Tian SK, Litvak AM, Paik PK, Drilon AE, Socci N, Poirier JT, Shen R, Berger MF, Moreira AL, Travis WD, Rudin CM, Ladanyi M (2016) Next-Generation sequencing of pulmonary large cell neuroendocrine carcinoma reveals small cell carcinoma-like and non-small cell carcinoma-like subsets. Clin Cancer Res 22:3618–3629
Derks JL, Leblay N, Thunnissen E, van Suylen R, den Bakker M, Groen HJM, Smit EF, Damhuis R, van den Broek E, Charbrier A, Foll M, McKay J, Fernandez-Cuesta L, Speel EM, Dingemans AC, PALGA-Group (2018) Molecular subtypes of pulmonary large-cell neuroendocrine carcinoma predict chemotherapy treatment outcome. Clin Cancer Res 24:33–42
Liu H, Zhang Y, Chang J, Liu Z, Tang N (2018) Differential expression of neuroendocrine markers, TTF-1, p53, and Ki-67 in cervical and pulmonary small cell carcinoma. Medicine (Baltimore) 97:e11604
Lloyd RV, Osamura RY, Klöppel G, Rosai J (eds) (2017) WHO classification of tumours of endocrine organs. 4th ed. IARC Press, Lyon
Bosman FT, Carneiro F, Hruban RH, Theise ND (eds) (2010) WHO classification of tumours of the digestive system. 4th ed. IARC Press, Lyon
National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: small cell lung cancer. https://www.nccn.org/professionals/physician_gls/pdf/sclc.pdf (Accessed 3/9/2021).
Kaufman HL, Russell J, Hamid O, Bhatia S, Terheyden P, D'Angelo SP, Shih KC, Lebbé C, Linette GP, Milella M, Brownell I, Lewis KD, Lorch JH, Chin K, Mahnke L, von Heydebreck A, Cuillerot JM, Nghiem P (2016) Avelumab in patients with chemotherapy-refractory metastatic Merkel cell carcinoma: a multicentre, single-group, open-label, phase 2 trial. Lancet Oncol 17:1374–1385
National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: merkel cell carcinoma. https://www.nccn.org/professionals/physician_gls/pdf/mcc.pdf (Accessed 3/9/2021).
National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: neuroendocrine and adrenal tumors. https://www.nccn.org/professionals/physician_gls/pdf/neuroendocrine.pdf (Accessed 3/9/2021).
Garcia-Carbonero R, Sorbye H, Baudin E, Raymond E, Wiedenmann B, Niederle B, Sedlackova E, Toumpanakis C, Anlauf M, Cwikla JB, Caplin M, O''Toole D, Perren A, all other Vienna Consensus Conference participants (2016) ENETS consensus guidelines for high-grade gastroenteropancreatic neuroendocrine tumors and neuroendocrine carcinomas. Neuroendocrinology 103:186–194
Uccella S, La Rosa S, Metovic J et al (2021) Genomics of high-grade neuroendocrine neoplasms: well-differentiated neuroendocrine tumor with high-grade features (G3 NET) and neuroendocrine carcinomas (NEC) of various anatomic sites. Endocr Pathol 32:192–210
Pierceall WE, Wolfe M, Suschak J, Chang H, Chen Y, Sprott KM, Kutok JL, Quan S, Weaver DT, Ward BE (2011) Strategies for H-score normalization of preanalytical technical variables with potential utility to immunohistochemical-based biomarker quantitation in therapeutic response diagnostics. Anal Cell Pathol (Amst) 34:159–168
Acknowledgements
Teri Bowman, HT, ASCP assisted with immunohistochemical staining for this project.
Author information
Authors and Affiliations
Contributions
SY analyzed stains and data, devised the algorithm, and wrote the manuscript. JLH assisted in case selection, performed some tissue staining, and offered expert advice. RSG conceived the study, assisted in case selection, arranged tissue staining, and edited the manuscript.
Corresponding author
Ethics declarations
Compliance with ethical standards
The authors adhered to institutional ethical standards.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yu, S., Hornick, J.L. & Gonzalez, R.S. An algorithmic approach utilizing CK7, TTF1, beta-catenin, CDX2, and SSTR2A can help differentiate between gastrointestinal and pulmonary neuroendocrine carcinomas. Virchows Arch 479, 481–491 (2021). https://doi.org/10.1007/s00428-021-03085-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-021-03085-7