Abstract
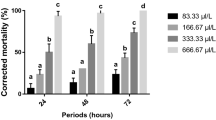
The indiscriminate use of synthetic insecticides to control Aedes aegypti has led to emergence of resistant populations. Moringa oleifera seeds contain the lectins WSMoL and cMoL. WSMoL has larvicidal activity on fourth-stage of A. aegypti organophosphate-susceptible larvae (Rockefeller L4). This study reports on the effects of cMoL on the survival of Rockefeller L4 as well as of WSMoL and cMoL on L4 from an organophosphate-resistant population (Rec-R). The effects of lectins on digestive (amylase, trypsin, and protease) and detoxifying (superoxide dismutase (SOD), α- and β-esterases) enzymes from larvae were also determined. cMoL (0.1–0.8 mg/ml) did not kill Rockefeller L4 as well as WSMoL and cMoL (0.1–0.8 mg/ml) were not larvicidal for Rec-R L4. WSMoL stimulated protease, trypsin-like, and α-amylase from Rockefeller L4 while cMoL inhibited these enzymes. WSMoL had no effect on trypsin-like activity from Rec-R L4 but inhibited protease and α-amylase. Among digestive enzymes of Rec-R L4, cMoL inhibited only trypsin-like activity. cMoL inhibited SOD activities from Rockefeller and Rec-R L4 in a higher level than WSMoL while β-esterase from Rockefeller L4 was more inhibited by WSMoL. The lectins promoted low stimulation or inhibition of α-esterase activities from both populations. In conclusion, Rockefeller and Rec-R larvae were distinctly affected by M. oleifera lectins, and larvicidal mechanism of WSMoL on Rockefeller L4 may involve deregulation of digestive enzymes. cMoL interfered mainly on SOD activity and thus it can be investigated as a synergistic agent for controlling populations whose resistance is linked to an increased detoxifying process mediated by this enzyme.



Similar content being viewed by others
References
Albuquerque LP, Santana GMS, Pontual EV, Napoleão TH, Coelho LCBB, Paiva PMG (2012) Effect of Microgramma vaccinifolia rhizome lectin on survival and digestive enzymes of Nasutitermes corniger (Isoptera, Termitidae). Int Biodeter Biodegr 75:158–166
Azeez A, Sane AP, Bhatnagar D, Nath P (2007) Enhanced expression of serine proteases during floral senescence in Gladiolus. Phytochemistry 68:1352–1357
Babu SR, Subrahmanyam B (2010) Bio-potency of serine proteinase inhibitors from Acacia senegal seeds on digestive proteinases, larval growth, and development of Helicoverpa armigera (Hübner). Pest Biochem Physiol 98:349–358
Bernfeld P (1955) Amylases, α and β. Method Enzymol 1:149–158
Bing DH, Weyand JG, Stavinsky AB (1967) Hemagglutination with aldehyde-fixed erythrocytes for assay of antigens and antibodies. Proc Soc Exp Biol Med 124:1166–1170
Braga IA, Valle D (2007) Aedes aegypti: surveillance, resistance monitoring, and control alternatives in Brazil. Epidemiol Serv Saúde 16:295–302
Coelho JS, Santos NDL, Napoleão TH, Gomes FS, Ferreira RS, Zingali RB, Coelho LCBB, Leite SP, Navarro DMAF, Paiva PMG (2009) Effect of Moringa oleifera lectin on development and mortality of Aedes aegypti. Chemosphere 77:934–938
Fitches E, Woodhouse SD, Edwards JP, Gatehouse JA (2001) In vitro and in vivo binding of snowdrop (Galanthus nivalis agglutinin; GNA) and jackbean (Canavalia ensiformis; ConA) lectins within tomato moth (Lacanobia oleracea) larvae: mechanisms of insecticidal action. J Insect Physiol 47:777–787
Flores AE, Albeldaño-Vázquez W, Salas IF, Badii MH, Becerra HL, Garcia GP, Fuentes SL, Brogdon WG, Black WC, Beaty B (2005) Elevated α-esterase levels associated with permethrin tolerance in Aedes aegypti (L.) from Baja California. Pest Biochem Physiol 82:66–78
Green AA, Hughes L (1955) Protein fractionation on the basis of solubility in aqueous solution of salts and organic solvents. In: Colowick S, Kaplan N (eds) Methods in Enzymology. Academic, New York, pp 67–90
Guirado MM, Bicudo HEMC (2009) Alguns aspectos do controle populacional e da resistência a inseticidas em Aedes aegypti (Diptera, Culicidae). BEPA 6:64
Kakade ML, Simons N, Liener IE (1969) An evaluation of natural versus synthetic substrates for measuring the antitryptic activity of soybean samples. Cereal Chem 46:518–526
Jagadeshwaran U, Vijayan VA (2009) Biochemical characterization of deltamethrin resistance in a laboratory-selected strain of Aedes aegypti. Parasitol Res 104:1431–1438
Larson RT, Lorch JM, Pridgeon JW, Becnel JJ, Clarck GG, Lan Q (2010) The biological activity of α-mangostin, a larvicidal botanic mosquito sterol carrier protein-2 inhibitor. J Med Entomol 47:249–257
Lima EP, Paiva MHS, Araújo AP, Silva EVG, Silva UM, Oliveira LN, Santana AEG, Barbosa CN, Paiva Neto CC, Goulart MOF, Wilding CS, Ayres CF, Melo-Santos MAV (2011) Insecticide resistance in Aedes aegypti populations from Ceará, Brazil. Parasit Vectors 4:5
Lima JBP, Cunha MP, da Silva Jr RC, Galardo AKR, Soares SS, Braga IA, Ramos RP, Valle D (2003) Resistance of Aedes aegypti to organophosphates in several municipalities in the state of Rio de Janeiro and Espírito Santo, Brazil. Am J Trop Med Hyg 68:329–333
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lumjuan N, Rajatileka S, Changsom D, Wicheer J, Leelapat P, Prapanthadara L, Somboon P, Lycett G, Ranson H (2011) The role of the Aedes aegypti Epsilon glutathione transferases in conferring. Insect Biochem Mol Biol 41:203–209
Luz LA, Silva MCC, Ferreira RS, Santana LA, Silva-Lucca RA, Mentele R, Oliva MLV, Paiva PMG, Coelho LCBB (2013) Structural characterization of coagulant Moringa oleifera lectin and its effect on hemostatic parameters. Int J Biol Macromol 58:31–36
Macedo MLR, Freire MGM, Silva MBR, Coelho LCBB (2007) Insecticidal action of Bauhinia monandra leaf lectin (BmoLL) against Anagasta kuehniella (Lepidoptera: Pyralidae), Zabrotes subfasciatus, and Callosobruchus maculatus (Coleoptera: Bruchidae). Comp Biochem Physiol A 146:486–498
Maheswaran R, Ignacimuthu S (2012) A novel herbal formulation against dengue vector mosquitoes Aedes aegypti and Aedes albopictus. Parasitol Res 110:1801–1813
Melo-Santos MAV, Varjal-Melo JJM, Araújo AP, Gomes TCS, Paiva MHS, Regis LN, Furtado AF, Magalhães T, Macoris MLG, Andrighetti MTM, Ayres CFJ (2010) Resistance to the organophosphate temephos: mechanisms, evolution, and reversion in an Aedes aegypti laboratory strain from Brazil. Acta Trop 113:180–189
Müller GJ, Lassmann H, Johansen FF (2007) Antiapoptotic signaling and failure of apoptosis in the ischemic rat hippocampus. Neurobiol Dis 25:582–593
Napoleão TH, Pontual EV, Lima TA, Santos NDL, Sá RA, Coelho LCBB, Navarro DMAF, Paiva PMG (2012) Effect of Myracrodruon urundeuva leaf lectin on survival and digestive enzymes of Aedes aegypti larvae. Parasitol Res 110:609–616
Nivsarkar M, Kumar GP, Laloraya M, Laloraya MM (1991) Superoxide dismutase in the anal gills of the mosquito larvae of Aedes aegypti: its inhibition by alpha-terthienyl. Arch Insect Biochem Physiol 16:249–255
Oliveira CFR, Luz LA, Paiva PMG, Coelho LCBB, Marangoni S, Macedo MLR (2011) Evaluation of seed coagulant Moringa oleifera lectin (cMoL) as a bio-insecticidal tool with potential for the control of insects. Process Biochem 46:498–504
Polson KA, Brogdon WG, Rawlins SC, Chadee DD (2011) Characterization of insecticide resistance in Trinidadian strains of Aedes aegypti mosquitoes. Acta Trop 117:31–38
Paiva PMG, Coelho LCBB (1992) Purification and partial characterization of two lectin isoforms from Cratylia mollis Mart. (camaratu bean). Appl Biochem Biotechnol 36:113–118
Paiva PMG, Santana GMS, Souza IFAC, Albuquerque LP, Agra-Neto AC, Albuquerque AC, Luz LA, Napoleão TH, Coelho LCBB (2011) Effect of lectins from Opuntia ficus indica cladodes and Moringa oleifera seeds on survival of Nasutitermes corniger. Int Biodegr Biodeter 65:982–989
Paiva PMG, Pontual EV, Napoleão TH, Coelho LCBB (2013) Lectins and trypsin inhibitors from plants: biochemical characteristics and adverse effects on insect larvae. Nova Science Publishers, Inc., New York, 52 p
Rolim LADMM, Macêdo MFS, Sisenando HA, Napoleão TH, Felzenszwalb I, Aiub CAF, Coelho LCBB, Medeiros SRB, Paiva PMG (2011) Genotoxicity evaluation of Moringa oleifera seed extract and lectin. J Food Sci 76:T53–T58
Sá RA, Santos NDL, da Silva CSB, Napoleão TH, Gomes FS, Cavada BS, Coelho LCBB, Navarro DMAF, Bieber LW, Paiva PMG (2009) Larvicidal activity of Myracrodruon urundeuva lectins on Aedes aegypti. Comp Biochem Physiol C 149:300–306
Santos AFS, Luz LA, Argolo ACC, Teixeira JA, Paiva PMG, Coelho LCBB (2009) Isolation of a seed coagulant Moringa oleifera lectin. Process Biochem 44:504–508
Santos NDL, Moura KS, Napoleão TH, Santos GKN, Coelho LCBB, Navarro DMAF, Paiva PMG (2012) Oviposition-stimulant and ovicidal activities of Moringa oleifera lectin on Aedes aegypti. PLoS ONE 7:e44840
Simas NK, Lima EC, Kuster RM, Lage CLS, Filho AMO (2007) Potential use of Piper nigrum ethanol extract against pyrethroid-resistant Aedes aegypti larvae. Rev Soc Bras Med Trop 40:405–407
Somwang P, Yanola J, Suwan W, Walton C, Lumjuan N, Prapanthadara L, Somboon P (2011) Enzymes-based resistant mechanism in pyrethroid resistant and susceptible Aedes aegypti strains from northern Thailand. Parasitol Res 109:531–537
World Health Organization (1981) Instructions for Determining the Susceptibility or Resistance of Mosquito Larvae to Insecticides, WHO/VBC/81.807, pp. 1–6
World Health Organization (2012) Dengue and severe dengue. Fact Sheet 117
Acknowledgments
The authors express their gratitude to the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for research grants and fellowships (CMFO, LCBBC, and PMGP). We are also grateful to the Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (FACEPE), the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and the Brazilian Ministry of Science, Technology and Innovation (MCTI) for financial support. A.C. Agra-Neto would like to thank CAPES for graduate scholarship. E.V. Pontual would like to thank FACEPE and CAPES for post-doctoral scholarship. N.D.L. Santos and L.A. Luz would like to thank FACEPE for graduate scholarships.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agra-Neto, A.C., Napoleão, T.H., Pontual, E.V. et al. Effect of Moringa oleifera lectins on survival and enzyme activities of Aedes aegypti larvae susceptible and resistant to organophosphate. Parasitol Res 113, 175–184 (2014). https://doi.org/10.1007/s00436-013-3640-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-013-3640-8