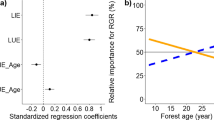
Abstract
Predictions of plant responses to climate change are frequently based on organisms’ presence in warmer locations, which are then assumed to reflect future performance in cooler areas. However, as plant life stages may be affected differently by environmental changes, there is little empirical evidence that this approach provides reliable estimates of short-term responses to global warming. Under this premise, we analyzed 8 years of early recruitment data, seed production and seedling establishment and survival, collected for two tree species at two latitudes. We quantified recruitment to a wide range of environmental conditions, temperature, soil moisture and light, and simulated recruitment under two forecasted climatic scenarios. Annual demographic transitions were affected by the particular conditions taking place during their onset, but the effects of similar environmental shifts differed among the recruitment stages; seed production was higher in warmer years, while seedling establishment and survival peaked during cold years. Within a species, these effects also varied between latitudes; increasing temperatures at the southern location will have stronger detrimental effects on recruitment than similar changes at the northern locations. Our simulations illustrate that warmer temperatures may increase seed production, but they will have a negative effect on establishment and survival. When the three early recruitment processes were simultaneously considered, simulations showed little change in recruitment dynamics at the northern site and a slight decrease at the southern site. It is only when we considered these three stages that we were able to assess likely changes in early recruitment under the predicted conditions.





Similar content being viewed by others
References
Handler S et al (2014) Michigan forest ecosystem vulnerability assessment and synthesis: a report from the Northwoods Climate Change Response Framework. US Department of Agriculture, Forest Service, Northern Research Station, Gen. Tech. Rep. NRS-129, Newtown Square, PA, p 242
Albrecht MA, McCarthy BC (2009) Seedling establishment shapes the distribution of shade-adapted forest herbs across a topographical moisture gradient. J Ecol 97:1037–1049
Anderson-Teixeira KJ, Miller AD, Mohan JE, Hudiburg TW, Duval BD, DeLucia EH (2013) Altered dynamics of forest recovery under a changing climate. Glob Change Biol 19:2001–2021
Barnes BV, Wagner WH (2004) Michigan trees. The University of Michigan Press, Ann Arbor
Bykova O, Chuine I, Morin X, Higgins SI (2012) Temperature dependence of the reproduction niche and its relevance for plant species distributions. J Biogeogr 39:2191–2200
Caldeira MC et al (2014) Direct and indirect effects of tree canopy facilitation in the recruitment of Mediterranean oaks. J App Ecol 51:349–353
Canham CD, Thomas RQ (2010) Frequency, not relative abundance, of temperate tree species varies along climate gradients in eastern North America. Ecology 91:3433–3440
Cavender-Bares J, Bazzaz FA (2000) Changes in drought response strategies with ontogeny in Quercus rubra: implications for scaling from seedlings to mature trees. Oecologia 124:8–18
Clark JS (2005) Why environmental scientists are becoming Bayesians. Ecol Lett 8:2–14
De Steven D (1991) Experiments on mechanisms of tree establishment in old-field succession: seedling emergence. Ecology 72:1066–1075
Diez JM et al (2014) Beyond seasonal climate: statistical estimation of phenological responses to weather. Ecol Appl 24:1793–1802
Flexas J, Medrano H (2002) Drought-inhibition of photosynthesis in C3 plants: stomatal and non-stomatal limitations revisited. Ann Bot 89:183–189
Gaillard J-M et al (2013) How does climate change influence demographic processes of widespread species? Lessons from the comparative analysis of contrasted populations of roe deer. Ecol Lett 16:48–57
Galvez DA, Landhausser SM, Tyree MT (2013) Low root reserve accumulation during drought may lead to winter mortality in poplar seedlings. New Phytol 198:139–148
Godman RM, Mattson GA (1976) Seed crops and regeneration problems of 19 species in northeastern Wisconsin. Res. Pap. NC-123. Department of Agriculture, Forest Service, North Central Forest Experiment Station, St. Paul, p 5
Godman RM, Yawney HW, Tubbs CH (1990) Acer saccharum Marsh. sugar maple. In: Burns RM, Honkala BH, Technical coordinators (eds) Silvics of North America. Vol. 2. Hardwoods, vol Agric. Handb. 654. Department of Agriculture, Forest Service, Washington, DC, pp 78–91
Green PT, Harms KE, Connell JH (2014) Nonrandom, diversifying processes are disproportionately strong in the smallest size classes of a tropical forest. PNAS 111:18649–18654
Grubb PJ (1977) The maintenance of species-richness in plant communities: the importance of the regeneration niche. Biol Rev Camb Philos Soc 52:102–145
Harper JL (1977) Polulation biology of plants. Academic Press, London
Hoch G, Siegwolf RTW, Keel SG, Körner C, Han Q (2013) Fruit production in three masting tree species does not rely on stored carbon reserves. Oecologia 171:653–662
Hoffmann AA, Camac JS, Williams RJ, Papst W, Jarrad FC, Wahren CH (2010) Phenological changes in six Australian subalpine plants in response to experimental warming and year-to-year variation. J Ecol 98:927–937. doi:10.1111/j.1365-2745.2010.01667.x
Horvath D (2009) Common mechanisms regulate flowering and dormancy. Plant Sci 177:523–531
Ibáñez I, McCarthy-Neumann S (2014) Integrated assessment of the direct and indirect effects of resource gradients on tree species recruitment. Ecology 95:364–375
Ibáñez I, Schupp EW (2001) Positive and negative interactions between environmental conditions affecting Cercocarpus ledifolius seedling survival. Oecologia 129:543–550
Ibáñez I et al (2006) Predicting biodiversity change: outside the climate envelope, beyond the species-area curve. Ecology 87:1896–1906
Ibáñez I, Clark JS, LaDeau SL, Hille Ris Lambers J (2007) Exploiting temporal variability to understand tree recruitment response to climate change. Ecol Monogr 77:163–177
Ibáñez I et al (2010) Forecasting phenology under global warming. Philos Trans R Soc B 365:3247–3260
Ibáñez I et al (2013) Moving forward in global-change ecology: capitalizing on natural variability. Ecol Evol 3:170–181
IPCC (2013) Climate change 2013: the physical science basis. In: Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge, United Kingdom and New York, NY, USA
Kaelke CM, Kruger EL, Reich PB (2001) Trade-offs in seedling survival, growth, and physiology among hardwood species of contrasting successional status along a light-availability gradient. Can J For Res 31:1602–1616
Kelly D (1994) The evolutionary ecology of mast seeding. Trends Ecol Evol 9:465–470
Kroiss SJ, HilleRisLambers J (2014) Recruitment limitation of long-lived conifers: implications for climate change responses. Ecology 96:1286–1297
Kumar S, Nayyar H, Bhanwara RK, Upadhyaya HD (2010) Chilling stress effects on reproductive biology of chickpea. SAT Agric Res 8:1–14
Kwit MC, Rigg LS, Goldblum D (2010) Sugar maple seedling carbon assimilation at the northern limit of its range: the importance of seasonal light. Can J For Res 40:385–393
Lambers H, Chapin FS, Pons TL (2008) Plant physiological ecology, 2nd edn. Springer, New York
Lavine M, Beckage B, Clark JS (2002) Statistical modeling of seedling mortality. J Agric Biol Environ Stat 7:21–41
Little EL (1979) Checklist of United States trees (native and naturalized). Department of Agriculture, Forest Service, Washington, DC
McKenney DW, Pedlar JH, Rood RB, Price D (2011) Revisiting projected shifts in the climate envelopes of North American trees using updated general circulation models. Glob Change Biol 17:2720–2730
Medlyn BE et al (2002) Temperature response of parameters of a biochemically based model of photosynthesis. II. A review of experimental data. Plant Cell Environ 25:1167–1179
Montesinos D, García-Fayos P, Verdú M (2012) Masting uncoupling: mast seeding does not follow all mast flowering episodes in a dioecious juniper tree. Oikos 121:1725–1736
Niinemets U (2010) Responses of forest trees to single and multiple environmental stresses from seedlings to mature plants: past stress history, stress interactions, tolerance and acclimation. For Ecol Manag 260:1623–1639
Nopp-Mayr U, Kempter I, Muralt G, Gratzer G (2012) Seed survival on experimental dishes in a central European old-growth mixed-species forest—effects of predator guilds, tree masting and small mammal population dynamics. Oikos 121:337–346
Ogle K et al (2015) Quantifying ecological memory in plant and ecosystem processes. Ecol Lett 18:221–235
Parks CG, Bernier P (2010) Adaptation of forests and forest management to changing climate with emphasis on forest health: a review of science, policies and practices. For Ecol Manag 259:657–659
Pearson RG, Dawson TP (2003) Predicting the impacts of climate change on the distribution of species: are bioclimate envelope models useful? Glob Ecol Biogeogr 12:361–371
Peltier D, Ibáñez I (2015) Patterns and variability in seedling carbon assimilation: implications for tree seedlings recruitment under climate change. Tree Physiol 35:71–85
Perez-Ramos IM, Rodriguez-Calcerrada J, Ourcival JM, Rambal S (2013) Quercus ilex recruitment in a drier world: a multi-stage demographic approach. Perspect Plant Ecol 15:106–117
Prasad AM, Iverson LR, Matthews S, Peters M (2007–ongoing) A Climate Change Atlas for 134 Forest Tree Species of the Eastern United States [database], Northern Research Station, USDA Forest Service, Delaware, Ohio. http://www.nrs.fs.fed.us/atlas/tree. Accessed 30 May 2017
Rennenberg H et al (2006) Physiological responses of forest trees to heat and drought. Plant Biol 8:556–571
Roberts AMI, Tansey C, Smithers RJ, Phillimore AB (2015) Predicting a change in the order of spring phenology in temperate forests. Glob Change Biol 21:2603–2611
Rollinson CR, Kaye MW, Canham CD (2016) Interspecific variation in growth responses to climate and competition of five eastern tree species. Ecology 97:1003–1011
Rother DC, Jordano P, Rodrigues RR, Pizo MA (2013) Demographic bottlenecks in tropical plant regeneration: a comparative analysis of causal influences. Perspect Plant Ecol 15:86–96
Schaetzl RJ, Krist FJ, Miller BA (2012) A taxonomically based ordinal estimate of soil productivity for landscape. Soil Sci 177:288–299
Schnurr JL, Canham CD, Ostefeld RS, Inouye RS (2004) Neighborhood analyses of small-mammal dynamics: impacts on seed predation and seedling establishment. Ecology 85:741–755
Shevtsova A et al (2009) Critical periods for impact of climate warming on early seedling establishment in subarctic tundra. Glob Change Biol 15:2662–2680
Sipe TW, Bazzaz FA (1994) Gap partitioning among maples (Acer) in central New England, shoot architecture and photosynthesis. Ecology 75:2318–2332
Smaill SJ, Clinton PW, Allen RB, Davis MR (2011) Climate cues and resources interact to determine seed production by a masting species. J Ecol 99:870–877
Springate DA, Kover PX (2014) Plant responses to elevated temperatures: a field study on phenological sensitivity and fitness responses to simulated climate warming. Glob Change Biol 20:456–465
Thomas A, O’Hara R, Ligges U, Sturts S (2006) Making BUGS open. R News 6:12–17
Thuiller W, Richardson DM, Pysek P, Midgley GF, Hughes GO, Rouget M (2005) Niche-based modelling as a tool for predicting the risk of alien plant invasions at a global scale. Glob Change Biol 11:2234–2250
Walters RS, Yawney HW (1990) Acer rubrum L. red maple. In: Burns RM, Honkala BH, Technical Coordinators (eds) Silvics of North America. Vol. 2. Hardwoods, vol Agric. Handb. 654. Department of Agriculture, Forest Service, Washington, DC, pp 60–69
Weltzin JF, Snyder KA, Williams DG (2001) Experimental manipulations of precipitation seasonality: effects on oak (Quercus) seedling demography and physiology. West N Am Nat 61:463–472
Woodall CW, Oswalt CM, Westfall JA, Perry CH, Nelson MD, Finley AO (2009) An indicator of tree migration in forests of the eastern United States. For Ecol Manag 257:1434–1444
Zang C, Hartl-Meier C, Dittmar C, Rothe A, Menzel A (2014) Patterns of drought tolerance in major European temperate forest trees: climatic drivers and levels of variability. Glob Change Biol 20:3767–3779
Zhu K, Woodall CW, Clark JS (2011) Failure to migrate: lack of tree range expansion in response to climate change. Glob Change Biol 18:1042–1052
Ziello C, Böck A, Estrella N, Ankerst D, Menzel A (2012) First flowering of wind-pollinated species with the greatest phenological advances in Europe. Ecography 35:1017–1023
Acknowledgements
Funding was provided by the National Science Foundation (NSF) Grant (DEB 1252664) to I. Ibáñez, and by NSF-GRF and NSF-DDIG (DEB 11309805) to D. Katz.
Author information
Authors and Affiliations
Contributions
Author contribution statement
II conceived and designed the experiment. II, DK and BL collected data. II analyzed the data. II, DK and BL wrote the manuscript.
Corresponding author
Additional information
Communicated by John Dwyer.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ibáñez, I., Katz, D.S.W. & Lee, B.R. The contrasting effects of short-term climate change on the early recruitment of tree species. Oecologia 184, 701–713 (2017). https://doi.org/10.1007/s00442-017-3889-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-017-3889-1