Abstract
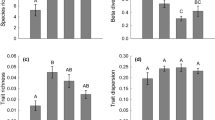
A major goal of metacommunity ecology is to infer the local- and regional-scale processes that underlie community assembly. In dendritic ecological networks, branching patterns and directional flow can alter the balance between local and regional factors during assembly. Vertical habitat structure may further affect community assembly in dendritic metacommunities. In this study, we analyzed the bacterial metacommunity of a fifth-order mountain stream network to assess differences in community assembly (1) between planktonic and benthic habitats, (2) across spatial scales, and (3) between headwater and downstream regions of the network. Using taxonomic and phylogenetic null modeling, we found habitat-specific spatial patterns of community assembly across the dendritic network. Compositional differences between planktonic and benthic communities were maintained by variable selection, but we also found evidence of local dispersal limitation between the two habitats. Planktonic community assembly was scale dependent, transitioning from homogeneous selection at local scales to variable selection at regional scales, while benthic community assembly was less scale dependent. Variable selection structured headwaters in both habitat types, but downstream communities were primarily structured by homogeneous selection, especially in sediments. Taken together, our results show that vertical habitat structure contributes to the scale-dependent processes of community assembly across the dendritic metacommunity.





Similar content being viewed by others
References
Altermatt F (2013) Diversity in riverine metacommunities: a network perspective. Aquat Ecol 47:365–377. https://doi.org/10.1007/s10452-013-9450-3
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46. https://doi.org/10.1111/j.1442-9993.2001.01070.pp.x
Bachmann RW, Canfield DE (1996) Use of an alternative method for monitoring total nitrogen concentrations in Florida lakes. Hydrobiologia 323:1–8. https://doi.org/10.1007/BF00020542
Battin TJ, Besemer K, Bengtsson MM et al (2016) The ecology and biogeochemistry of stream biofilms. Nat Rev Microbiol 14:251–263. https://doi.org/10.1038/nrmicro.2016.15
Besemer K, Peter H, Logue JB et al (2012) Unraveling assembly of stream biofilm communities. ISME J 6:1459–1468. https://doi.org/10.1038/ismej.2011.205
Brown BL, Swan CM (2010) Dendritic network structure constrains metacommunity properties in riverine ecosystems. J Anim Ecol 79:571–580. https://doi.org/10.1111/j.1365-2656.2010.01668.x
Brown BL, Swan CM, Auerbach DA et al (2011) Metacommunity theory as a multispecies, multiscale framework for studying the influence of river network structure on riverine communities and ecosystems. J North Am Benthol Soc 30:310–327. https://doi.org/10.1899/10-129.1
Cadotte MW, Davies TJ (2016) Phylogenies in ecology: a guide to concepts and methods. Princeton University Press, Princeton
Caporaso JG, Lauber CL, Walters WA et al (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J 6:1621–1624. https://doi.org/10.1038/ismej.2012.8
Carrara F, Altermatt F, Rodriguez-Iturbe I, Rinaldo A (2012) Dendritic connectivity controls biodiversity patterns in experimental metacommunities. Proc Natl Acad Sci 109:5761–5766. https://doi.org/10.1073/pnas.1119651109
Chase JM, Myers JA (2011) Disentangling the importance of ecological niches from stochastic processes across scales. Philos Trans R Soc B Biol Sci 366:2351–2363. https://doi.org/10.1098/rstb.2011.0063
Chase JM, Kraft NJB, Smith KG et al (2011) Using null models to disentangle variation in community dissimilarity from variation in α-diversity. Ecosphere 2:art24. https://doi.org/10.1890/ES10-00117.1
Daly C, Conklin DR, Unsworth MH (2010) Local atmospheric decoupling in complex topography alters climate change impacts. Int J Climatol 30:1857–1864. https://doi.org/10.1002/joc.2007
Fierer N, Morse JL, Berthrong ST et al (2007) Environmental controls on the landscape-scale biogeography of stream bacterial communities. Ecology 88:2162–2173. https://doi.org/10.1890/06-1746.1
Freimann R, Bürgmann H, Findlay SEG, Robinson CT (2015) Hydrologic linkages drive spatial structuring of bacterial assemblages and functioning in alpine floodplains. Front Microbiol 6:1–15. https://doi.org/10.3389/fmicb.2015.01221
Grant EHC, Lowe WH, Fagan WF (2007) Living in the branches: population dynamics and ecological processes in dendritic networks. Ecol Lett 10:165–175. https://doi.org/10.1111/j.1461-0248.2006.01007.x
Harr RD (1977) Water flux in soil and subsoil on a steep forested slope. J Hydrol 33:37–58. https://doi.org/10.1016/0022-1694(77)90097-X
Hart DD, Finelli CM (1999) Physical-biological coupling in streams: the pervasive effects of flow on benthic organisms. Annu Rev Ecol Syst 30:363–395. https://doi.org/10.1146/annurev.ecolsys.30.1.363
Hassell N, Tinker KA, Moore T, Ottesen EA (2018) Temporal and spatial dynamics in microbial community composition within a temperate stream network. Environ Microbiol 20:3560–3572. https://doi.org/10.1111/1462-2920.14311
Hermans SM, Buckley HL, Lear G (2018) Optimal extraction methods for the simultaneous analysis of DNA from diverse organisms and sample types. Mol Ecol Resour 18:557–569. https://doi.org/10.1111/1755-0998.12762
Hermans SM, Buckley HL, Case BS, Lear G (2020) Connecting through space and time: catchment-scale distributions of bacteria in soil, stream water and sediment. Environ Microbiol 22:1000–1010. https://doi.org/10.1111/1462-2920.14792
Jost L (2007) Partitioning diversity into independent alpha and beta components. Ecology 88:2427–2439. https://doi.org/10.1890/06-1736.1
Kaestli M, Munksgaard N, Gibb K, Davis J (2019) Microbial diversity and distribution differ between water column and biofilm assemblages in arid-land waterbodies. Freshw Sci 38:869–882. https://doi.org/10.1086/706106
Kembel SW, Cowan PD, Helmus MR et al (2010) Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26:1463–1464. https://doi.org/10.1093/bioinformatics/btq166
Leff LG, McArthur JV, Shimkets LG (1992) Information spiraling: movement of bacteria and their genes in streams. Microb Ecol 24:11–24. https://doi.org/10.1007/BF00171967
Legendre P, Legendre L (2012) Numerical ecology, 3rd edn. Elsevier, Amsterdam
Leibold MA, Chase JM (2018) Metacommunity ecology. Princeton University Press, Princeton
McKee A, Bierlmaier F (1987) H. J. Andrews Experimental Forest, Oregon. In: Greenland D (ed) The climates of the long-term ecological research sites. Institute of Arctic and Alpine Research. University of Colorado, Boulder, pp 11–17
Mouquet N, Loreau M (2003) Community patterns in source-sink metacommunities. Am Nat 162:544–557. https://doi.org/10.1086/378857
Niño-García JP, Ruiz-González C, del Giorgio PA (2016) Interactions between hydrology and water chemistry shape bacterioplankton biogeography across boreal freshwater networks. ISME J 10:1755–1766. https://doi.org/10.1038/ismej.2015.226
Oksanen J, Blanchet FG, Friendly M, et al (2019) vegan: community ecology package. R package version 2.5–4. Version 2.5–4
Prepas EE, Rigler FH (1982) Improvements in quantifying the phosphorus concentration in lake water. Can J Fish Aquat Sci 39:822–829. https://doi.org/10.1139/f82-112
Price MN, Dehal PS, Arkin AP (2010) FastTree 2—approximately maximum-likelihood trees for large alignments. PLoS ONE 5:e9490–e9490. https://doi.org/10.1371/journal.pone.0009490
Quast C, Pruesse E, Yilmaz P et al (2013) The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41:590–596. https://doi.org/10.1093/nar/gks1219
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/.
Read DS, Gweon HS, Bowes MJ et al (2015) Catchment-scale biogeography of riverine bacterioplankton. ISME J 9:516–526. https://doi.org/10.1038/ismej.2014.166
Rognes T, Flouri T, Nichols B et al (2016) VSEARCH: a versatile open source tool for metagenomics. PeerJ 4:e2584–e2584. https://doi.org/10.7717/peerj.2584
Ruiz-González C, Niño-García JP, del Giorgio PA (2015) Terrestrial origin of bacterial communities in complex boreal freshwater networks. Ecol Lett 18:1198–1206. https://doi.org/10.1111/ele.12499
Savio D, Sinclair L, Ijaz UZ et al (2015) Bacterial diversity along a 2600 km river continuum. Environ Microbiol 17:4994–5007. https://doi.org/10.1111/1462-2920.12886
Schloss PD, Westcott SL, Ryabin T et al (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541. https://doi.org/10.1128/aem.01541-09
Schmera D, Árva D, Boda P et al (2018) Does isolation influence the relative role of environmental and dispersal-related processes in stream networks? An empirical test of the network position hypothesis using multiple taxa. Freshw Biol 63:74–85. https://doi.org/10.1111/fwb.12973
Stegen JC, Lin X, Fredrickson JK et al (2013) Quantifying community assembly processes and identifying features that impose them. ISME J 7:2069–2079. https://doi.org/10.1038/ismej.2013.93
Stegen JC, Lin X, Fredrickson JK, Konopka AE (2015) Estimating and mapping ecological processes influencing microbial community assembly. Front Microbiol 6:370–370. https://doi.org/10.3389/fmicb.2015.00370
Swanson FJ, James ME (1975) Geology and geomorphology of the H.J. Andrews Experimental Forest, Western Cascades, Oregon. U.S. Department of Agriculture, Forest Service, Pacific Northwest Forest and Range Experiment Station, Portland, OR
Swanson FJ, Jones JA (2002) Geomorphology and hydrology of the H.J. andrews experimental forest, blue river, oregon. Field Guide Geol Process Cascadia Dep Geol Miner Ind Spec Pap 36:289–314
Tonkin JD, Altermatt F, Finn DS et al (2018) The role of dispersal in river network metacommunities: patterns, processes, and pathways. Freshw Biol 63:141–163. https://doi.org/10.1111/fwb.13037
Tornwall BM, Swan CM, Brown BL (2017) Manipulation of local environment produces different diversity outcomes depending on location within a river network. Oecologia. https://doi.org/10.1007/s00442-017-3891-7
Vannote RL, Minshall GW, Cummins KW et al (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137. https://doi.org/10.1139/f80-017
Vellend M (2016) The theory of ecological communities. Princeton University Press, Princeton
Ward AS, Wondzell SM, Schmadel NM et al (2019) Spatial and temporal variation in river corridor exchange across a 5th-order mountain stream network. Hydrol Earth Syst Sci 23:5199–5225. https://doi.org/10.5194/hess-23-5199-2019
Westcott SL, Schloss PD (2017) OptiClust, an improved method for assigning amplicon-based sequence data to operational taxonomic units. mSphere 2:e00073–e117. https://doi.org/10.1128/mSphereDirect.00073-17
Wilhelm L, Singer GA, Fasching C et al (2013) Microbial biodiversity in glacier-fed streams. ISME J 7:1651–1660. https://doi.org/10.1038/ismej.2013.44
Wisnoski NI, Muscarella ME, Larsen ML et al (2020) Metabolic insight into bacterial community assembly across ecosystem boundaries. Ecology 101:e02968. https://doi.org/10.1002/ecy.2968
Zhou J, Ning D (2017) Stochastic community assembly: does it matter in microbial ecology? Microbiol Mol Biol Rev 81:e00002–17. https://doi.org/10.1128/MMBR.00002-17
Acknowledgements
We thank Adam Ward for logistical and financial support during sample collection, and Annie Bowling for technical assistance. This work was supported by the National Science Foundation (DEB-1442246, JTL), US Army Research Office Grant (W911NF-14-1-0411, JTL), and the Department of Biology at Indiana University (George W. Brackenridge Fellowship, Louise Constable Hoover Fellowship to NIW). Data and code for the project is archived at the NCBI Sequence Read Archive (BioProject PRJNA606285) and in a Zenodo archive (https://doi.org/10.5281/zenodo.4052168) of the GitHub repository (https://github.com/LennonLab/HJA-streams). LIDAR Data and facilities were provided by the HJ Andrews Experimental Forest and Long-Term Ecological Research program, administered cooperatively by the USDA Forest Service Pacific Northwest Research Station, Oregon State University, and the Willamette National Forest. This material is based upon work supported by the National Science Foundation under Grant No. DEB-1440409.
Author information
Authors and Affiliations
Contributions
NIW and JTL conceived the study. NIW collected and analyzed the data. NIW and JTL wrote the manuscript.
Corresponding author
Additional information
Communicated by Jamie M. Kneitel.
In dendritic ecological networks, such as streams, branching patterns and directional flow can alter the balance between local and regional processes underlying community assembly. Streams also contain vertical habitat structure, encompassing planktonic and benthic habitats that impose contrasting sets of environmental filters and differ in spatial connectivity within the metacommunity. In this study, we demonstrated that the relative importance of community assembly processes in a stream bacterial metacommunity was not only spatially variable across the network, but also dependent on spatial scale. Furthermore, the strength and pattern of scale-dependence differed within and between vertical habitats in the watershed, suggesting the metacommunity resembles a multi-layer dendritic network.
Rights and permissions
About this article
Cite this article
Wisnoski, N.I., Lennon, J.T. Microbial community assembly in a multi-layer dendritic metacommunity . Oecologia 195, 13–24 (2021). https://doi.org/10.1007/s00442-020-04767-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04767-w