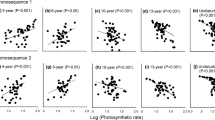
Abstract
Plant phenology differs largely among coexisting species within communities that share similar habitat conditions. However, the factors explaining such phenological diversity of plants have not been fully investigated. We hypothesize that species traits, including leaf mass per area (LMA), seed mass, stem tissue mass density (STD), maximum plant height (Hmax), and relative growth rate in height (RGRH), explain variation in plant phenology, and tested this hypothesis in an alpine meadow. Results showed that both LMA and STD were positively correlated with the onset (i.e., beginning) and offset (i.e., ending) times of the four life history events including two reproductive events (flowering and fruiting) and two vegetative events (leafing and senescing). In contrast, RGRH was negatively correlated with the four life phenological events. Moreover, Hmax was positively correlated with reproductive events but not with vegetative events. However, none of the eight phenological events was associated with seed size. In addition, the combination of LMA and STD accounted for 50% of the variation in plant phenologies. Phylogenetic generalized least squares analysis showed plant phylogeny weakened the relationships between species traits vs. phenologies. Phylogeny significantly regulated the variation in the ending but not the beginning of phenologies. Our results indicate that species traits are robust indicators for plant phenologies and can be used to explain the diversity of plant phenologies among co-occurring herbaceous species in grasslands. The findings highlight the important role of the combination of and trade-offs between functional traits in determing plant phenology diversity in the alpine meadow.




Similar content being viewed by others
References
Armstrong JB, Takimoto G, Schindler DE, Hayes MM, Kauffman MJ (2016) Resource waves: phenological diversity enhances foraging opportunities for mobile consumers. Ecology 97:1099–1112. https://doi.org/10.1890/15-0554.1
Bolmgren K, Cowan DP (2008) Time-size tradeoffs: a phylogenetic comparative study of flowering time, plant height and seed mass in a north-temperate flora. Oikos 117:424–429. https://doi.org/10.1111/j.2007.0030-1299.16142.x
Brody AK (1997) Effects of pollinators, herbivores, and seed predators on flowering phenology. Ecology 78:1624–1631. https://doi.org/10.1890/0012-9658(1997)078[1624:EOPHAS]2.0.CO;2
Castro-Díez P, Milla R, Sanz V (2005) Phenological comparison between two co-occurring Mediterranean woody species differing in growth form. Flora 200:88–95. https://doi.org/10.1016/j.flora.2004.07.002
Catorci A, Cesaretti S, Gatti R, Tardella FM (2012) Trait-related flowering patterns in submediterranean mountain meadows. Plant Ecol 213:1315–1328. https://doi.org/10.1007/s11258-012-0090-9
Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE (2009) Towards a worldwide wood economics spectrum. Ecol Lett 12:351–366. https://doi.org/10.1111/j.1461-0248.2009.01285.x
Cleland EE, Chuine I, Menzel A, Mooney HA, Schwartz MD (2007) Shifting plant phenology in response to global change. Trends Ecol Evol 22:357–365. https://doi.org/10.1016/j.tree.2007.04.003
Cornelissen JHC, Lavorel S, Garnier E, Diaz S, Buchmann N, Gurvich DE, Reich PB, ter Steege H, Morgan HD, van der Heijden MGA (2003) A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust J Bot 51:335–380. https://doi.org/10.1071/BT02124
Craine JM, Wolkovich EM, Towne EG, Kembel SW (2012) Flowering phenology as a functional trait in a tallgrass prairie. New Phytol. 193:673–682. https://doi.org/10.1111/j.1469-8137.2011.03953.x
Davis CC, Willis CG, Primack RB, Miller-Rushing AJ (2012) The importance of phylogeny to the study of phenological response to global climate change. Philos Trans R Soc B 365:3201–3213. https://doi.org/10.1098/rstb.2010.0130
Du G, Qi W (2010) Trade-offs between flowering time, plant height, and seed size within and across 11 communities of a QingHai-Tibetan flora. Plant Ecol 209:321–333. https://doi.org/10.1007/s11258-010-9763-4
Du YJ, Mao LF, Queenborough SA, Freckleton RP, Chen B, Ma KP (2015) Phylogenetic constraints and trait correlates of flowering phenology in the angiosperm flora of China. Glob Ecol Biogeogr 24:928–938. https://doi.org/10.1111/geb.12303
ECCAS (1998) Editor Committee of the Chinese Academy of Sciences for Flora of China. Flora of China Science Press, Beijing
Fantinato E, Del Vecchio S, Giovanetti M, Acosta ATR, Buffa G (2018) New insights into plants co-existence in species-rich communities: the pollination interaction perspective. J Veg Sci 29:6–14. https://doi.org/10.1111/jvs.12592
Felsenstein J (1985) Confidence limits phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Fotis AT, Curtis PS (2017) Effects of structural complexity on within-canopy light environments and leaf traits in a northern mixed deciduous forest. Tree Physiol 37:1426–1435. https://doi.org/10.1093/treephys/tpw124
Freckleton RP, Harvey PH, Pagel M (2002) Phylogenetic analysis and comparative data: a test and review of evidence. Am Nat 160:712–726. https://doi.org/10.1086/343873
Gaudinier A, Blackman BK (2020) Evolutionary processes from the perspective of flowering time diversity. New Phytol 225:1883–1898. https://doi.org/10.1111/nph.16205
Hara T, Kimura M, Kikuzawa K (1991) Growth patterns of tree height and stem diameter in populations of Abiesveitchii, A. mariesii and Betulaermanii. J Ecol 79:1085–1098. https://doi.org/10.2307/2261100
Harvey PH, Pagel MD (1991) The comparative method in evolutionary biology. Oxford University Press, Oxford
Hunt R, Cornelissen JHC (1997) Components of relative growth rate and their interrelations in 59 temperate plant species. New Phytol 135:395–417. https://doi.org/10.1046/j.1469-8137.1997.00671.x
Jia P, Bayaerta T, Li XQ, Du GZ (2011) Relationships between flowering phenology and functional traits in eastern Tibet alpine meadow. Arct Ant Alp Res 43:585–592. https://doi.org/10.1657/1938-4246-43.4.585
Johnson SD (1993) Climatic and phylogenetic determinants of flowering seasonality in the Cape flora. J Ecol 81:567–572. https://doi.org/10.2307/2261535
Kaufmann KW (1981) Fitting and using growth curves. Oecologia 49:293–299. https://doi.org/10.1007/BF00347588
King DA, Davies SJ, Nur Supardi MN, Tan S (2005) Tree growth is related to light interception and wood density in two mixed dipterocarp forests of Malaysia. Funct Ecol 19:445–453. https://doi.org/10.1111/j.1365-2435.2005.00982.x
King DA, Davies SJ, Tan S, Noor NSM (2006a) The role of wood density and stem support costs in the growth and mortality of tropical trees. J Ecol 94:670–680. https://doi.org/10.1111/j.1365-2745.2006.01112.x
King DA, Davies SJ, Noor NSM (2006b) Growth and mortality are related to adult tree size in a Malaysian mixed dipterocarp forest. For Ecol Manag 223:152–158. https://doi.org/10.1016/j.foreco.2005.10.066
Kochmer JP, Handel SN (1986) Constraints and competition in the evolution of flowering phenology. Ecol Monogr 56:303–325. https://doi.org/10.2307/1942549
Lima ALA, Rodal MJN (2010) Phenology and wood density of plants growing in the semi-arid region of northeastern Brazil. J Arid Environ 74:1363–1373. https://doi.org/10.1016/j.jaridenv.2010.05.009
Liu Y, Reich PB, Li G, Sun S (2011) Shifting phenology and abundance under experimental warming alters trophic relationships and plant reproductive capacity. Ecology 92:1201–1207. https://doi.org/10.1890/10-2060.1
Liu Y, Miao R, Chen A, Miao Y, Liu Y, Wu X (2017) Effects of nitrogen addition and mowing on reproductive phenology of three early-flowering forb species in a Tibetan alpine meadow. Ecol Eng 99:119–125. https://doi.org/10.1016/j.ecoleng.2016.11.033
Mason NWH, Pipenbaher N, Škornik S, Kaligarič M (2013) Does complementarity in leaf phenology and inclination promote co-existence in a species-rich meadow? Evidence from functional groups. J Veg Sci 24:94–100. https://doi.org/10.1111/j.1654-1103.2012.01451.x
Miller-Rushing AJ, Forrest J (2010) Toward a synthetic understanding of the role of phenology in ecology and evolution. Philos Trans R Soc B 365:3101–3260. https://doi.org/10.1098/rstb.2010.0145
Mo L, Luo P, Mou CX, Yang H, Wang J, Wang ZY, Li YJ, Luo C, Li T, Zuo DD (2018) Winter plant phenology in the alpine meadow on the eastern Qinghai-Tibetan Plateau. Ann Bot Lond 1226:1033–1045. https://doi.org/10.1093/aob/mcy112
Nakadai R, Murakami M, Hirao T (2014) Effects of phylogeny, leaf traits, and the altitudinal distribution of host plants on herbivore assemblages on congeneric Acer species. Oecologia 175:1237–1245. https://doi.org/10.1007/s00442-014-2964-0
Nord EA, Lynch JP (2009) Plant phenology: a critical controller of soil resource acquisition. J Exp Bot 60:1927–1937. https://doi.org/10.1093/jxb/erp018
Ollerton J, Lack AJ (1992) Flowering phenology: an example of relaxation of natural selection? Trends Ecol Evol 7:274–276. https://doi.org/10.1016/0169-5347(92)90175-B
Pagel M (1999) Inferring the historical patterns of biological evolution. Nature 401:877–884. https://doi.org/10.1038/44766
Paradis E, Claude J, Strimmer K (2004) APE: analyses of phylogentics and evolution in R language. Bioinformatics 20:289–290. https://doi.org/10.1093/bioinformatics/btg412
Polgar CA, Primack RB (2011) Leaf-out phenology of temperate woody plants: from trees to ecosystems. New Phytol 191:926–941. https://doi.org/10.1111/j.1469-8137.2011.03803.x
Poorter L, Bongers F, Sterck FJ, Woll H (2003) Architecture of 53 rain forest tree species differing in adult stature and shade tolerance. Ecology 84:602–608. https://doi.org/10.1890/0012-9658(2003)084[0602:AORFTS]2.0.CO;2
Poorter L, Bongers L, Bongers F (2006) Architecture of 54 moist-forest tree species: traits, trade-offs, and functional groups. Ecology 87:1289–1301. https://doi.org/10.1890/0012-9658(2006)87[1289:AOMTST]2.0.CO;2
Poorter L, Wright SJ, Paz H, Ackerly DD, Condit R, Ibarra-Manríquez G, Harms KE, Licona JC, Martínez-Ramos M, Mazer SJ, Muller-Landau HC, Peña-Claros M, Webb CO, Wright IJ (2008) Are functional traits good predictors of demographic rates? Evidence from five neotropical forests. Ecology 89:1908–1920. https://doi.org/10.1890/07-0207.1
R Development Core Team (2019) R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing
Rathcke B, Lacey EP (1985) Phenological patterns of terrestrial plants. Annu Rev Ecol Evol Syst 16:179–214. https://doi.org/10.1146/annurev.es.16.110185.001143
Singh KP, Kushwaha CP (2006) Diversity of flowering and fruiting phenology of trees in a tropical deciduous forest in India. Ann Bot 97:265–276. https://doi.org/10.1093/aob/mcj028
Staggemeier VG et al (2010) The shared influence of phylogeny and ecology on the reproductive patterns of Myrteae (Myrtaceae). J Ecol 98:1409–1421. https://doi.org/10.1111/j.1365-2745.2010.01717.x
Statsoft Inc. (2001) Statistica 6.0 Tulsa, Oklahoma, USA
Sun S, Jin D, Li R (2006) Timing of leaf emergence in relation to leaf traits of woody species in eastern Chinese Quercus fabri forests. Act Oecol 30:212–222. https://doi.org/10.1016/j.actao.2006.04.001
Sun S, Frelich LE (2011) Flowering phenology and height growth pattern are associated with maximum plant height, relative growth rate and stem tissue mass density in herbaceous grassland species. J Ecol 99:991–1000. https://doi.org/10.1111/j.1365-2745.2011.01830.x
Vile D, Shipley B, Garnier E (2006) A structural equation model to integrate changes in functional strategies during old-field succession. Ecology 87:504–517. https://doi.org/10.1890/05-0822
Wang J, Ives NE, Echowicz MJL (1992) The relation of foliar phenology to xylem embolism in trees. Funct Ecol 6:469–475. https://doi.org/10.2307/2389285
Wang GC, Huang Y, Wei YR, Zhang W, Li TT, Zhang Q (2019) Inner Mongolian grassland plant phenological changes and thei r climatic drivers. Sci Tot Environ 683:1–8. https://doi.org/10.1016/j.scitotenv.2019.05.125
Webb CO, Ackerly DD, Kembel SW (2008) Phylocom: software for the analysis of phylogenetic community structure and trait evolution. Bioinformatics 24:2098–2100. https://doi.org/10.1093/bioinformatics/btn358
Westoby M, Falster DS, Moles AT, Vesk PA, Wright IJ (2002) Plant ecology strategies: some leading dimensions of variation between species. Annu Rev Eco Evol Syst 33:125–159. https://doi.org/10.1146/annurev.ecolsys.33.010802.150452
Wu X, Duffy JE, Reich PB, Sun SC (2011) A brown-world cascade in the dung decomposer food web of an alpine meadow: effects of predator interactions and warming. Ecol Monogr 81:313–328. https://doi.org/10.1890/10-0808.1
Xia J, Wan S (2013) Independent effects of warming and nitrogen addition on plant phenology in the Inner Mongolian steppe. Ann Bot 111:1207–1217. https://doi.org/10.1093/aob/mct079
Xu J, Chen Y, Zhang L, Chai Y, Wang M, Guo Y, Li T, Yue M (2017) Using phylogeny and functional traits for assessing community assembly along environmental gradients: a deterministic process driven by elevation. Ecol Evol 7:5056–5069. https://doi.org/10.1002/ece3.3068
Acknowledgements
We thank Jian Feng, Yibin Yuan, and Ang Zhang for the help with data collection; Xinqiang Xi and Xiaoli Hu for the help on phylogenetic regression analyses.
Funding
This study was funded by National Natural Science Foundation of China (Grant number 31530007, 31670477, and 31770522).
Author information
Authors and Affiliations
Contributions
YL, GL, and SS collected the data and wrote the first draft of the manuscript. YL and XW analyzed the data. YL, GL, KJN, ZY, and SS contributed substantially to technical support and English revisions. SS design the experiment.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Data accessibility
All basic data deposited in the Appendix as support information Table S1 and Table S2.
Additional information
Communicated by Jonathan A. Myers.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Y., Li, G., Wu, X. et al. Linkage between species traits and plant phenology in an alpine meadow. Oecologia 195, 409–419 (2021). https://doi.org/10.1007/s00442-020-04846-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04846-y