Abstract
Background
Rectal carcinoids are increasing in incidence worldwide. Frequently thought of as a relatively benign condition, there are limited data regarding optimal treatment strategies for both localized and more advanced disease. The aim of this study was to summarize published experiences with rectal carcinoids and to present the most current data.
Methods
Following PRISMA guidelines, an electronic literature search performed of PubMed, Medline, Embase, and the Cochrane Library using the terms “rectum” or “rectal” AND “carcinoid” over a 20-year study period from January 1993 to May 2013. Non-English-language studies, animal studies, and studies of fewer than 100 patients were excluded. Study end points included demographic information, tumor features, intervention and outcomes. All included articles were quality assessed.
Results
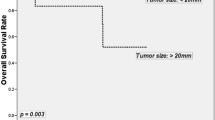
Using the search parameters and exclusions as outlined above, a total of 14 articles were identified for detailed analysis. The quality of articles was low/moderate for all included scoring 9 to 17 of 27. The articles included 4,575 patients diagnosed with a rectal carcinoid. Approximately 80 % of tumors were <10 mm, 15 % 11–20 mm, and 5 % >20 mm. Eight percent of patients presented with regional lymph node metastases, and 4 % presented with distant metastases. Tumor size >10 mm, and muscular and lymphovascular invasion are independently associated with an increased risk of metastases. The 5-year survival was 93 % in patients presenting with localized disease and 86 % overall.
Conclusions
Small tumors up to 10 mm without any adverse features can be treated with endoscopic or local excision. The treatment of carcinoids between 10 and 20 mm is still contentious, but those up to 16 mm without adverse feature are suitable for local/endoscopic excision followed by careful histopathological assessment. Those >20 mm or with adverse features require radical surgery with mesorectal clearance in suitable patients.

Similar content being viewed by others
References
Langhans T (1867) Ueber einen drusenpolyp im ileum. Virchows Arch Pathol Anat Physiol Klin Med 38:559–560
Lubarsch O (1888) Uber den primaren Krebs des ileum nebst Bemerkungen uber das gleichzeitige Vorkommen von Krebs and Tuberkulose. Virchows Arch 3:280–317
Oberndorfer S (1907) Karzinoide tumoren des dunndarms. Frankf Z Pathol 1:426–432
Anthony LB, Strosberg JR, Klimstra DS et al (2010) The NANETS consensus guidelines for the diagnosis and management of gastrointestinal neuroendocrine tumors (NETS): well-differentiated nets of the distal colon and rectum. Pancreas 39:767–774
Soga J (1997) Carcinoids of the rectum: an evaluation of 1271 reported cases. Surg Today 27:112–119
Scherubl H (2009) Rectal carcinoids are on the rise: early detection by screening endoscopy. Endoscopy 41:162–165
Modlin IM, Lye KD, Kidd M (2003) A 5-decade analysis of 13,715 carcinoid tumors. Cancer 97:934–959
Edge SB, Compton CC (2010) The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 17:1471–1474
Downs SH, Black N (1998) The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Commun Health 52:377–384
Moher D, Liberati A, Tetzlaff J et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535
Maggard MA, O’Connell JB, Ko CY (2004) Updated population-based review of carcinoid tumors. Ann Surg 240:117–122
Kim MS, Hur H, Min BS et al (2013) Clinical outcomes for rectal carcinoid tumors according to a new (AJCC 7th edition) TNM staging system: a single institutional analysis of 122 patients. J Surg Oncol 107:835–841
Zhong DD, Shao LM, Cai JT (2013) Endoscopic mucosal resection vs endoscopic submucosal dissection for rectal carcinoid tumours: a systematic review and meta-analysis. Colorectal Dis 15:283–291
Lee DS, Jeon SW, Park SY et al (2010) The feasibility of endoscopic submucosal dissection for rectal carcinoid tumors: comparison with endoscopic mucosal resection. Endoscopy 42:647–651
Onozato Y, Kakizaki S, Iizuka H et al (2010) Endoscopic treatment of rectal carcinoid tumors. Dis Colon Rectum 53:169–176
Park HW, Byeon JS, Park YS et al (2010) Endoscopic submucosal dissection for treatment of rectal carcinoid tumors. Gastrointest Endosc 72:143–149
Zhou PH, Yao LQ, Qin XY et al (2010) Advantages of endoscopic submucosal dissection with needle-knife over endoscopic mucosal resection for small rectal carcinoid tumors: a retrospective study. Surg Endosc 24:2607–2612
Son HJ, Sohn DK, Hong CW et al (2013) Factors associated with complete local excision of small rectal carcinoid tumor. Int J Colorectal Dis 28:57–61
Kim HH, Park SJ, Lee SH et al (2012) Efficacy of endoscopic submucosal resection with a ligation device for removing small rectal carcinoid tumor compared with endoscopic mucosal resection: analysis of 100 cases. Dig Endosc 24:159–163
Kim KM, Eo SJ, Shim SG et al (2013) Treatment outcomes according to endoscopic treatment modalities for rectal carcinoid tumors. Clin Res Hepatol Gastroenterol. doi:10.1016/j.clinre.2012.07.007
Kasuga A, Chino A, Uragami N et al (2012) Treatment strategy for rectal carcinoids: a clinicopathological analysis of 229 cases at a single cancer institution. J Gastroenterol Hepat 27:1801–1807
Colonoscopy Study Group of Korean Society of C (2011) Clinical characteristics of colorectal carcinoid tumors. J Korean Soc Coloproctol 27:17–20
Shields CJ, Tiret E, Winter DC et al (2010) Carcinoid tumors of the rectum: a multi-institutional international collaboration. Ann Surg 252:750–755
Yoon SN, Yu CS, Shin US et al (2010) Clinicopathological characteristics of rectal carcinoids. Int J Colorectal Dis 25:1087–1092
Wang M, Peng J, Yang W et al (2011) Prognostic analysis for carcinoid tumours of the rectum: a single institutional analysis of 106 patients. Colorectal Dis 13:150–153
Kim BN, Sohn DK, Hong CW et al (2008) Atypical endoscopic features can be associated with metastasis in rectal carcinoid tumors. Surg Endosc 22:1992–1996
Li AF, Hsu CY, Li A et al (2008) A 35-year retrospective study of carcinoid tumors in Taiwan: differences in distribution with a high probability of associated second primary malignancies. Cancer 112:274–283
Soga J (2005) Early-stage carcinoids of the gastrointestinal tract: an analysis of 1914 reported cases. Cancer 103:1587–1595
Modlin IM, Oberg K, Chung DC et al (2008) Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol 9:61–72
Kobayashi K, Katsumata T, Yoshizawa S et al (2005) Indications of endoscopic polypectomy for rectal carcinoid tumors and clinical usefulness of endoscopic ultrasonography. Dis Colon Rectum 48:285–291
Ramage JK, Davies AH, Ardill J et al (2005) UKNETwork for Neuroendocrine Tumours Guidelines for the management of gastroenteropancreatic neuroendocrine (including carcinoid) tumours. Gut 54(suppl 4):1–16
Rindi G, Arnold R, Bosman FT, Capella C, Klimstra DS, Kloppel G, Komminoth P, Solcia E (2010) Nomenclature and classification of neuroendocrine neoplasms of the digestive system. In: Bosman FT, Carneiro F, Hruban RH, Theise ND (eds) Who classification of tumours of the digestive system, 4th edn. IARC, Lyon, pp 13–14
Modlin IM, Kidd M, Latich I et al (2005) Current status of gastrointestinal carcinoids. Gastroenterology 128:1717–1751
Disclosures
Mr. McDermott, Miss Heeney, Dr. Courtney, Miss Mohan, and Professor Winter have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McDermott, F.D., Heeney, A., Courtney, D. et al. Rectal carcinoids: a systematic review. Surg Endosc 28, 2020–2026 (2014). https://doi.org/10.1007/s00464-014-3430-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-014-3430-0