Abstract
Purpose
Cancer survivors are often sedentary. Self-monitoring may promote physical activity through self-activation. We conducted a pilot trial to evaluate whether wearable activity tracker with personalized text message feedback would increase physical activity.
Methods
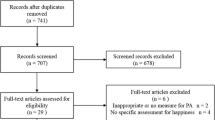
We enrolled 30 patients with solid tumor cancers into a non-randomized prospective intervention trial (NCT02627079): 15 had completed treatment in the past year and 15 under active treatment. Each participant received an activity tracker and daily text messages personalized to their activity level. We assessed patient-reported outcomes and 6-min walk (6 MW) at baseline and 3 months.
Results
Twenty-six participants completed the study. There was substantial variation in baseline activity. Overall, 39% of participants increased their steps taken by at least 20%, and 23% increased their 6 MW distance by 20% or more. More participants who had completed treatment strongly agreed (73%) that the intervention increased their exercise levels than those receiving active treatment (47%). At 3 months, there was a significant improvement in median Beck Depression Inventory-II and Godin Leisure Index composite scores. At 6 months, 72% still wore their activity tracker at least 4 days per week.
Conclusion
We found that the intervention was well-accepted with a high completion rate at 3 months and continued self-use at 6 months. In this pilot study of combined activity tracker and motivational messaging, we found a signal for increased physical activity over a 3-month period. Future research is needed to study this technique for its impact on activity and other physical and psychological measures of well-being.
Implication for cancer survivors
Activity tracker with personalized motivational messaging may be useful in promoting physical activity in cancer survivors.


Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Society AC (2019) Cancer treatment and survivorship facts & figures 2019. American Cancer Society, Atlanta
Lakoski SG, Eves ND, Douglas PS, Jones LW (2012) Exercise rehabilitation in patients with cancer. Nat Rev Clin Oncol 9(5):288–296. https://doi.org/10.1038/nrclinonc.2012.27
Zhang X, Pawlikowski M, Olivo-Marston S, Williams KP, Bower JK, Felix AS (2021) Ten-year cardiovascular risk among cancer survivors: the National Health and Nutrition Examination Survey. PLoS ONE 16(3):e0247919. https://doi.org/10.1371/journal.pone.0247919
Sweegers MG, Boyle T, Vallance JK, Chinapaw MJ, Brug J, Aaronson NK et al (2019) Which cancer survivors are at risk for a physically inactive and sedentary lifestyle? Results from pooled accelerometer data of 1447 cancer survivors. Int J Behav Nutr Phys Act 16(1):66. https://doi.org/10.1186/s12966-019-0820-7
Capozzi LC, Boldt KR, Lau H, Shirt L, Bultz B, Culos-Reed SN (2014) A clinic-supported group exercise program for head and neck cancer survivors: managing cancer and treatment side effects to improve quality of life. Support Care Cancer. https://doi.org/10.1007/s00520-014-2436-4
Courneya KS, Mackey JR, Bell GJ, Jones LW, Field CJ, Fairey AS (2003) Randomized controlled trial of exercise training in postmenopausal breast cancer survivors: cardiopulmonary and quality of life outcomes. J Clin Oncol 21(9):1660–1668. https://doi.org/10.1200/JCO.2003.04.093
Lewis C, Xun P, He K (2014) Physical activity in relation to quality of life in newly diagnosed colon cancer patients: a 24-month follow-up. Qual Life Res 23(8):2235–2246. https://doi.org/10.1007/s11136-014-0679-7
Kenfield SA, Stampfer MJ, Giovannucci E, Chan JM (2011) Physical activity and survival after prostate cancer diagnosis in the health professionals follow-up study. J Clin Oncol 29(6):726–732. https://doi.org/10.1200/JCO.2010.31.5226
Schmid D, Leitzmann MF (2014) Association between physical activity and mortality among breast cancer and colorectal cancer survivors: a systematic review and meta-analysis. Ann Oncol 25(7):1293–1311. https://doi.org/10.1093/annonc/mdu012
Blanchard CM, Courneya KS, Stein K, American Cancer Society’s SCS, II (2008) Cancer survivors’ adherence to lifestyle behavior recommendations and associations with health-related quality of life: results from the American Cancer Society’s SCS-II. J Clin 26(13):2198–204. https://doi.org/10.1200/JCO.2007.14.6217
Sweegers MG, Altenburg TM, Chinapaw MJ, Kalter J, Verdonck-de Leeuw IM, Courneya KS et al (2018) Which exercise prescriptions improve quality of life and physical function in patients with cancer during and following treatment? A systematic review and meta-analysis of randomised controlled trials. Br J Sports Med 52(8):505–513. https://doi.org/10.1136/bjsports-2017-097891
Xu A, Wang Y, Wu X (2019) Effectiveness of e-health based self-management to improve cancer-related fatigue, self-efficacy and quality of life in cancer patients: systematic review and meta-analysis. J Adv Nurs 75(12):3434–3447. https://doi.org/10.1111/jan.14197
Cox M, Basen-Engquist K, Carmack CL, Blalock J, Li Y, Murray J et al (2017) Comparison of Internet and telephone interventions for weight loss among cancer survivors: randomized controlled trial and feasibility study. JMIR Cancer 3(2):e16. https://doi.org/10.2196/cancer.7166
Petersen JM, Prichard I, Kemps E (2019) A comparison of physical activity mobile apps with and without existing web-based social networking platforms: systematic review. J Med Internet Res 21(8):e12687. https://doi.org/10.2196/12687
Winger JG, Mosher CE, Rand KL, Morey MC, Snyder DC, Demark-Wahnefried W (2014) Diet and exercise intervention adherence and health-related outcomes among older long-term breast, prostate, and colorectal cancer survivors. Ann Behav Med 48(2):235–245. https://doi.org/10.1007/s12160-014-9598-7
Loncar-Turukalo T, Zdravevski E, Machado da Silva J, Chouvarda I, Trajkovik V (2019) Literature on wearable technology for connected health: scoping review of research trends, advances, and barriers. J Med Internet Res 21(9):e14017. https://doi.org/10.2196/14017
Gresham G, Schrack J, Gresham LM, Shinde AM, Hendifar AE, Tuli R et al (2018) Wearable activity monitors in oncology trials: current use of an emerging technology. Contemp Clin Trials 64:13–21. https://doi.org/10.1016/j.cct.2017.11.002
Bennett GG, Foley P, Levine E, Whiteley J, Askew S, Steinberg DM et al (2013) Behavioral treatment for weight gain prevention among black women in primary care practice: a randomized clinical trial. JAMA Intern Med 173(19):1770–1777. https://doi.org/10.1001/jamainternmed.2013.9263
Bennett GG, Herring SJ, Puleo E, Stein EK, Emmons KM, Gillman MW (2010) Web-based weight loss in primary care: a randomized controlled trial. Obesity 18(2):308–313. https://doi.org/10.1038/oby.2009.242
Bennett GG, Warner ET, Glasgow RE, Askew S, Goldman J, Ritzwoller DP et al (2012) Obesity treatment for socioeconomically disadvantaged patients in primary care practice. Arch Intern Med 172(7):565–574. https://doi.org/10.1001/archinternmed.2012.1
Lee H, Uhm KE, Cheong IY, Yoo JS, Chung SH, Park YH et al (2018) Patient satisfaction with mobile health (mHealth) application for exercise intervention in breast cancer survivors. J Med Syst 42(12):254. https://doi.org/10.1007/s10916-018-1096-1
Lin PH, Wang Y, Levine E, Askew S, Lin S, Chang C et al (2014) A text messaging-assisted randomized lifestyle weight loss clinical trial among overweight adults in Beijing. Obesity 22(5):E29-37. https://doi.org/10.1002/oby.20686
Pollak KI, Alexander SC, Bennett G, Lyna P, Coffman CJ, Bilheimer A et al. (2014) Weight-related SMS texts promoting appropriate pregnancy weight gain: a pilot study. Patient education and counseling. https://doi.org/10.1016/j.pec.2014.07.030
Pollak KI, Lyna P, Bilheimer A, Farrell D, Gao X, Swamy GK et al (2013) A pilot study testing SMS text delivered scheduled gradual reduction to pregnant smokers. Nicotine Tob Res 15(10):1773–1776. https://doi.org/10.1093/ntr/ntt045
Steinberg DM, Levine EL, Askew S, Foley P, Bennett GG (2013) Daily text messaging for weight control among racial and ethnic minority women: randomized controlled pilot study. J Med Internet Res 15(11):e244. https://doi.org/10.2196/jmir.2844
Laboratories ACoPSfCPF (2002) ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med 166(1):111–7.
Powell C, Carson BP, Dowd KP, Donnelly AE (2017) Simultaneous validation of five activity monitors for use in adult populations. Scand J Med Sci Sports 27(12):1881–1892. https://doi.org/10.1111/sms.12813
Van Blarigan EL, Kenfield SA, Tantum L, Cadmus-Bertram LA, Carroll PR, Chan JM (2017) The Fitbit one physical activity tracker in men with prostate cancer: validation study. JMIR Cancer 3(1):e5. https://doi.org/10.2196/cancer.6935
Cella DF, Tulsky DS, Gray G, Sarafian B, Linn E, Bonomi A et al (1993) The functional assessment of cancer therapy scale: development and validation of the general measure. J Clin Oncol 11(3):570–579. https://doi.org/10.1200/JCO.1993.11.3.570
Webster K, Cella D, Yost K (2003) The functional assessment of chronic illness therapy (FACIT) measurement system: properties, applications, and interpretation. Health Qual Life Outcomes;1:79. https://doi.org/10.1186/1477-7525-1-79
Hopko DR, Bell JL, Armento ME, Robertson SM, Hunt MK, Wolf NJ et al (2008) The phenomenology and screening of clinical depression in cancer patients. J Psychosoc Oncol 26(1):31–51
Rogers LQ, Courneya KS, Robbins KT, Malone J, Seiz A, Koch L et al (2006) Physical activity and quality of life in head and neck cancer survivors. Support Care Cancer 14(10):1012–1019. https://doi.org/10.1007/s00520-006-0044-7
Hodges JL, Lehmann EL (1963) Estimates of location based on tank-tests. Ann Math Stat 34(2):598–0. https://doi.org/10.1214/aoms/1177704172
Button KS, Kounali D, Thomas L, Wiles NJ, Peters TJ, Welton NJ et al (2015) Minimal clinically important difference on the Beck Depression Inventory–II according to the patient’s perspective. Psychol Med 45(15):3269–3279. https://doi.org/10.1017/S0033291715001270
Yost KJ, Eton DT (2005) Combining distribution- and anchor-based approaches to determine minimally important differences: the FACIT experience. Eval Health Prof 28(2):172–191. https://doi.org/10.1177/0163278705275340
Denison E, Vist GE, Underland V, Berg RC. (2014) Interventions aimed at increasing the level of physical activity by including organised follow-up: a systematic review of effect. BMC Fam Pract 15:120. https://doi.org/10.1186/1471-2296-15-120
Isaacs AJ, Critchley JA, Tai SS, Buckingham K, Westley D, Harridge SD et al. (2007) Exercise Evaluation Randomised Trial (EXERT): a randomised trial comparing GP referral for leisure centre-based exercise, community-based walking and advice only. Health Technol Assess; 11(10):1–165, iii-iv.
Chung IY, Jung M, Lee SB, Lee JW, Park YR, Cho D et al (2019) An assessment of physical activity data collected via a smartphone app and a smart band in breast cancer survivors: observational study. J Med Internet Res 21(9):13463. https://doi.org/10.2196/13463
Demark-Wahnefried W, Clipp EC, Lipkus IM, Lobach D, Snyder DC, Sloane R et al (2007) Main outcomes of the FRESH START trial: a sequentially tailored, diet and exercise mailed print intervention among breast and prostate cancer survivors. J Clin Oncol 25(19):2709–2718. https://doi.org/10.1200/JCO.2007.10.7094
Hegberg NJ, Hayes JP, Hayes SM (2019) Exercise intervention in PTSD: a narrative review and rationale for implementation. Front Psychiatry;10:133. https://doi.org/10.3389/fpsyt.2019.00133
Acknowledgements
We thank Xuechan Li and Bercedis Peterson for their invaluable assistance in the development of this protocol. In addition, we acknowledge Mouna Abouamara, Maleka Ahmed, Ivy Altomare, Russell Anderson, Junzo Chino, Christopher R Kelsey, and Donette Vicente for supporting this study in their clinics.
Funding
This study was funded by institutional pilot funding from the Duke University School of Medicine.
Author information
Authors and Affiliations
Contributions
Conceptualization, BK, WEK, GB, and KIP; methodology, BK, EL, FM, MS, and GB; formal analysis and investigation, BK, EL, FM, DN, LS, TD, MS, CR, and LO; writing — original draft preparation, BK; writing — review and editing, EL, FM, DN, LS, TD, MS, CR, LO, WEK, GB, and KIP; funding acquisition, BK; supervision, BK.
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable.
Conflict of interest
BK receives unrestricted research funding from Janssen Pharmaceuticals, Merck, and Blue Earth Diagnostics, all unrelated to this project. She has been an Advisory Board Member of Myovant, Bayer, and Blue Earth Diagnostics, unrelated to this project. GB is a scientific advisor at WW and holds equity in Coeus Health which are unrelated to this project. All remaining authors declare no conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The findings herein have been presented in part at the following meetings: ASTRO 2017 (poster) ASCO Survivorship 2018 (poster).
Gary Bennett and Kathryn I. Pollak contributed equally to this work
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Koontz, B.F., Levine, E., McSherry, F. et al. Increasing physical activity in Cancer Survivors through a Text-messaging Exercise motivation Program (ICanSTEP). Support Care Cancer 29, 7339–7349 (2021). https://doi.org/10.1007/s00520-021-06281-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-021-06281-y